| |
EASL Clinical Practice Guidelines: Management of chronic hepatitis B
|
| |
| |
Downlaod the PDF : EASL Clinical Practice Guidelines: Management of chronic hepatitis B
Journal of Hepatology Feb 2009
European Association for the Study of the Liver
Article Outline
⋅ 1. Introduction
⋅ 2. Context
⋅ 2.1. Epidemiology and public health burden
⋅ 2.2. Natural history
⋅ 3. Methodology
⋅ 4. Guidelines
⋅ 4.1. Pretherapeutic assessment of liver disease
⋅ 4.2. Goal of therapy
⋅ 4.3. End-points of therapy
⋅ 4.4. Definitions of response
⋅ 4.5. Results of current therapies
⋅ 4.6. Indications for treatment
⋅ 4.7. Predictors of response
⋅ 4.8. Treatment strategies: how-to-treat
⋅ 4.9. Treatment failure
⋅ 4.10. How to monitor treatment and stopping points
⋅ 4.10.1. Finite therapy with pegylated interferon alpha
⋅ 4.10.2. Finite treatment with NUCs in HBeAg-positive patients
⋅ 4.10.3. Long-term therapy with NUCs
⋅ 4.11. Treatment of patients with severe liver disease
⋅ 4.11.1. Treatment of patients with cirrhosis
⋅ 4.11.2. Treatment of patients with decompensated cirrhosis
⋅ 4.12. Prevention of recurrent hepatitis B after liver transplantation
⋅ 4.13. Treatment in special patient groups
⋅ 4.13.1. HIV co-infected patients
⋅ 4.13.2. HDV co-infected patients
⋅ 4.13.3. HCV co-infected patients
⋅ 4.13.4. Acute severe hepatitis
⋅ 4.13.5. Children
⋅ 4.13.6. Healthcare workers
⋅ 4.13.7. Pregnant women
⋅ 4.13.8. Pre-emptive therapy before immunosuppressive therapy or chemotherapy
⋅ 4.13.9. Dialysis and renal transplant patients
⋅ 4.13.10. Extrahepatic disease
⋅ 5. Unresolved issues and unmet needs
⋅ Conflicts of interest disclosure
⋅ References_
1. Introduction
Our understanding of the natural history of hepatitis B virus (HBV) infection and the potential for therapy of the resultant disease has improved. Several new and effective antiviral agents have been evaluated and licensed since the EASL International Consensus Conference on hepatitis B held in 2002 [1]. The objective of these EASL Clinical Practice Guidelines (CPGs) is to update recommendations for the optimal management of chronic hepatitis B (CHB). The CPGs do not focus on prevention and vaccination. Several difficulties remain in formulating treatments for CHB; thus areas of uncertainty exist. At the present time clinicians, patients and public health authorities must continue to make choices on the basis of evidence that is not fully matured.
2. Context
2.1. Epidemiology and public health burden
Approximately one third of the world's population has serological evidence of past or present infection with HBV and 350 million people are chronically infected. The spectrum of disease and natural history of chronic HBV infection is diverse and variable, ranging from a low viremic inactive carrier state to progressive chronic hepatitis, which may evolve to cirrhosis and hepatocellular carcinoma (HCC). HBV-related end stage liver disease or HCC are responsible for over 1 million deaths per year and currently represent 5-10% of cases of liver transplantation [2], [3], [4], [5]. Host and viral factors, as well as coinfection with other viruses, in particular hepatitis C virus (HCV), hepatitis D virus (HDV), or human immunodeficiency virus (HIV) together with other co-morbidities including alcohol abuse and overweight, can affect the natural course of HBV infection as well as the efficacy of antiviral strategies.
CHB may present either as hepatitis B e antigen (HBeAg)-positive or HBeAg-negative CHB. HBeAg-positive CHB is due to so-called "wild type" HBV. It typically represents the early phase of chronic HBV infection. HBeAg-negative CHB is due to replication of naturally occurring HBV variants with nucleotide substitutions in the precore and/or basic core promoter regions of the genome and represents a later phase of chronic HBV infection. The prevalence of the HBeAg-negative form of the disease has been increasing over the last decade as a result of HBV-infected population aging and represents the majority of cases in many areas, including Europe [6], [7], [8].
Morbidity and mortality in CHB are linked to persistence of viral replication and evolution to cirrhosis or HCC. Longitudinal studies of patients with CHB indicate that, after diagnosis, the 5-year cumulative incidence of developing cirrhosis ranges from 8 to 20%. The 5-year cumulative incidence of hepatic decompensation is approximately 20% with the 5-year probability of survival being approximately 80-86% in patients with compensated cirrhosis [4], [9], [10], [11], [12], [13]. Patients with decompensated cirrhosis have a poor prognosis with a 14-35% probability of survival at 5 years. The worldwide incidence of HCC has increased, mostly due to HBV and HCV infections; presently it constitutes the fifth most common cancer, representing around 5% of all cancers. The annual incidence of HBV-related HCC in patients with CHB is high, ranging from 2% to 5% when cirrhosis is established [13]. However, the incidence of HBV-related HCC appears to vary geographically and correlates with the underlying stage of liver disease.
Population movements and migration are currently changing the prevalence and incidence of the disease in several low endemicity countries in Europe and elsewhere. Substantial healthcare resources will be required for control of the worldwide burden of disease.
2.2. Natural history
Chronic hepatitis B is a dynamic process. The natural history of CHB can be schematically divided into five phases, which are not necessarily sequential.
(1)The "immune tolerant" phase is characterized by HBeAg positivity, high levels of HBV replication (reflected by high levels of serum HBV DNA), normal or low levels of aminotransferases, mild or no liver necroinflammation and no or slow progression of fibrosis [3], [5]. During this phase, the rate of spontaneous HBeAg loss is very low. This first phase is more frequent and more prolonged in subjects infected perinatally or in the first years of life. Because of high levels of viremia, these patients are highly contagious.
(2)The "immune reactive phase" is characterized by HBeAg positivity, a lower level of replication (as reflected by lower serum HBV DNA levels), increased or fluctuating levels of aminotransferases, moderate or severe liver necroinflammation and more rapid progression of fibrosis compared to the previous phase [3], [5]. It may last for several weeks to several years. In addition, the rate of spontaneous HBeAg loss is enhanced. This phase may occur after several years of immune tolerance and is more frequently reached in subjects infected during adulthood.
(3)The "inactive HBV carrier state" may follow seroconversion from HBeAg to anti-HBe antibodies. It is characterized by very low or undetectable serum HBV DNA levels and normal aminotransferases. As a result of immunological control of the infection, this state confers a favourable long-term outcome with a very low risk of cirrhosis or HCC in the majority of patients. HBsAg loss and seroconversion to anti-HBs antibodies may occur spontaneously in 1-3% of cases per year, usually after several years with persistently undetectable HBV DNA [14].
(4)"HBeAg-negative CHB" may follow seroconversion from HBeAg to anti-HBe antibodies during the immune reactive phase and represents a later phase in the natural history of CHB. It is characterized by periodic reactivation with a pattern of fluctuating levels of HBV DNA and aminotransferases and active hepatitis. These patients are HBeAg-negative, and harbour HBV variants with nucleotide substitutions in the precore and/or the basal core promoter regions unable to express or expressing low levels of HBeAg. HBeAg-negative CHB is associated with low rates of prolonged spontaneous disease remission. It is important and sometimes difficult to distinguish true inactive HBV carriers from patients with active HBeAg-negative CHB in whom phases of spontaneous remission may occur. The former patients have a good prognosis with a very low risk of complications, while the latter patients have active liver disease with a high risk of progression to advanced hepatic fibrosis, cirrhosis and subsequent complications such as decompensated cirrhosis and HCC. A careful assessment of the patient is needed and a minimal follow-up of one year with serum alanine aminotransferase (ALT) and HBV DNA levels every 3 months usually allows detection of fluctuations of activity in patients with active HBeAg-negative CHB [15].
(5)In the "HBsAg-negative phase" after HBsAg loss, low-level HBV replication may persist with detectable HBV DNA in the liver [16]. Generally, HBV DNA is not detectable in the serum while anti-HBc antibodies with or without anti-HBs are detectable. HBsAg loss is associated with improvement of the outcome with reduced risk of cirrhosis, decompensation and HCC. The clinical relevance of occult HBV infection (detectable HBV DNA in the liver with low-level [<200 international units (IU)/ml] HBV DNA in blood) is unclear [16]. Immunosuppression may lead to reactivation in these patients [17], [18].
3. Methodology
These EASL CPGs have been developed by a CPG Panel of experts chosen by the EASL Governing Board; the recommendations were peer-reviewed by external expert reviewers and approved by the EASL Governing Board. The CPGs have been based as far as possible on evidence from existing publications, and, if evidence was unavailable, the experts' personal experience and opinion. Manuscripts and abstracts of important meetings published prior to August 2008 have been evaluated. The evidence and recommendations in these guidelines have been graded according to the Grading of Recommendations Assessment Development and Evaluation (GRADE) system. The strength of recommendations thus reflects the quality of underlying evidence. The principles of the GRADE system have been enunciated. The quality of the evidence in these CPGs has been classified in one of three levels: high (A), moderate (B) or low (C). The GRADE system offers two grades of recommendation: strong (1) or weak (2) (Table 1). The CPGs thus consider the quality of evidence: the higher the quality of evidence, the more likely a strong recommendation is warranted; the greater the variability in values and preferences, or the greater the uncertainty, the more likely a weaker recommendation is warranted [19], [20], [21], [22], [23], [24], [25].
Table 1. Grading of evidence and recommendations (adapted from the GRADE system) [19], [20], [21], [22], [23], [24], [25]

The CPG Panel members considered the following questions:
⋅How should liver disease be assessed before therapy?
⋅What are the goals and end-points of treatment?
⋅What are the definitions of response?
⋅What is the optimal approach to first-line treatment?
⋅What are the predictors of response?
⋅What definitions of resistance should be applied and how should resistance be managed?
⋅How should treatment be monitored?
⋅When can treatment be stopped?
⋅How should special groups be treated?
⋅What are the current unresolved issues?
4. Guidelines
4.1. Pretherapeutic assessment of liver disease
As a first step, the causal relationship between HBV infection and liver disease has to be established and an assessment of the severity of liver disease needs to be performed. Not all patients with CHB have persistently elevated aminotransferases. Patients in the immune tolerant phase have persistently normal ALT levels and a proportion of patients with HBeAg-negative CHB may have intermittently normal ALT levels. Therefore appropriate, longitudinal long-term follow-up is crucial.
(1)The assessment of the severity of the liver disease should include: biochemical markers, including aspartate aminotransferase (AST) and ALT, gamma-glutamyl transpeptidase (GGT), alkaline phosphatase, prothrombin time and serum albumin; blood counts; and hepatic ultrasound (A1). Usually, ALT levels are higher than those of AST. However, when the disease progresses to cirrhosis, the ratio may be reversed. A progressive decline in serum albumin concentrations and prolongation of the prothrombin time, often accompanied by a drop in platelet counts, are characteristically observed after cirrhosis has developed.
(2)HBV DNA detection and HBV DNA level measurement is essential for the diagnosis, decision to treat and subsequent monitoring of patients (A1). Follow-up using real-time PCR quantification assays is strongly recommended because of their sensitivity, specificity, accuracy and broad dynamic range [26], [27], [28], [29] (A1). The World Health Organization (WHO) has defined an international standard for normalisation of expression of HBV DNA concentrations [30]. Serum HBV DNA levels should be expressed in IU/ml to ensure comparability; the same assay should be used in the same patient to evaluate antiviral efficacy (A1).
(3)Other causes of chronic liver disease should be systematically looked for including coinfection with HDV, HCV and/or HIV. Co-morbidities, including alcoholic, autoimmune, metabolic liver disease with steatosis or steato-hepatitis should be assessed (A1).
(4)A liver biopsy is recommended for determining the degree of necroinflammation and fibrosis in patients with either increased ALT or HBV DNA levels >2000 IU/ml (or both) since hepatic morphology can assist the decision to start treatment (A1). Biopsy is also useful for evaluating other possible causes of liver disease such as steatosis or steato-hepatitis. Although liver biopsy is an invasive procedure, the risk of severe complications is very low (1/4,000-10,000). It is important that the size of the needle biopsy specimen be large enough to precisely analyse the degree of liver injury and fibrosis [31] (A1). A liver biopsy is usually not required in patients with clinical evidence of cirrhosis or in those in whom treatment is indicated irrespective of the grade of activity or the stage of fibrosis (A1). There is growing interest in the use of non-invasive methods, including serum markers and transient elastography, to assess hepatic fibrosis to complement or avoid a liver biopsy [32], [33], [34], [35], [36].
4.2. Goal of therapy
The goal of therapy for hepatitis B is to improve quality of life and survival by preventing progression of the disease to cirrhosis, decompensated cirrhosis, end-stage liver disease, HCC and death. This goal can be achieved if HBV replication can be suppressed in a sustained manner, the accompanying reduction in histological activity of chronic hepatitis lessening the risk of cirrhosis and decreasing the risk of HCC in non-cirrhotic patients and probably also, but to a lesser extent, in cirrhotic patients [37] (B1). However, HBV infection cannot be completely eradicated due to the persistence of covalently closed circular DNA (cccDNA) in the nucleus of infected hepatocytes.
4.3. End-points of therapy
Therapy must reduce HBV DNA to as low a level as possible, ideally below the lower limit of detection of real-time PCR assays (10-15 IU/ml), to ensure a degree of virological suppression that will then lead to biochemical remission, histological improvement and prevention of complications. Interferon alpha or nucleoside/nucleotide analogue (NUC) therapy-induced HBV DNA reduction to low levels is associated with disease remission. Sustained HBV DNA reduction to undetectable levels is necessary to reduce the risk of resistance to NUCs. It also increases the chance of HBe seroconversion in HBeAg-positive patients and the possibility of HBsAg loss on the mid to long term in HBeAg-positive and HBeAg-negative patients. If real-time PCR is unavailable, HBV DNA should be measured by the most sensitive assay possible.
(1)In HBeAg-positive and HBeAg-negative patients, the ideal end-point of therapy is sustained HBsAg loss with or without seroconversion to anti-HBs. This is associated with a complete and definitive remission of the activity of chronic hepatitis B and an improved long-term outcome (A1).
(2)In HBeAg-positive patients, durable HBe seroconversion is a satisfactory end-point because it has been shown to be associated with improved prognosis (A1).
(3)In HBeAg-positive patients who do not achieve HBe seroconversion, and in HBeAg-negative patients, a maintained undetectable HBV DNA level on treatment with NUCs or a sustained undetectable HBV DNA level after interferon therapy is the next most desirable end-point (A1).
4.4. Definitions of response
Two different types of drugs can be used in the treatment of CHB: interferon alpha and nucleoside/nucleotide analogues referred to collectively as NUCs in this document. The definition of response to antiviral therapy varies according to the type of therapy.
(1)On interferon alpha therapy:
⋅Primary non-response is defined as less than 1 log10 IU/ml decrease in HBV DNA level from baseline at 3 months of therapy.
⋅Virological response is defined as an HBV DNA concentration of less than 2000 IU/ml at 24 weeks of therapy.
⋅Serological response is defined by HBe seroconversion in patients with HBeAg-positive CHB.
(2)On NUC therapy:
⋅Primary non-response is defined as less than 1 log10 IU/ml decrease in HBV DNA level from baseline at 3 months of therapy.
⋅Virological response is defined as undetectable HBV DNA by real-time PCR assay within 48 weeks of therapy.
⋅Partial virological response is defined as a decrease in HBV DNA of more than 1 log10 IU/ml but detectable HBV DNA by real-time PCR assay. A partial virological response should be assessed to modify therapy at 24 weeks of treatment for moderately potent drugs or drugs with a low genetic barrier to resistance (lamivudine and telbivudine) and at 48 weeks of treatment for highly potent drugs, drugs with a higher genetic barrier to resistance or drugs with a late emergence of resistance (entecavir, adefovir and tenofovir).
⋅Virological breakthrough is defined as a confirmed increase in HBV DNA level of more than 1 log10 IU/ml compared to the nadir (lowest value) HBV DNA level on therapy; it usually precedes a biochemical breakthrough, characterized by an increase in ALT levels. The main causes of virological breakthrough on NUC therapy are poor adherence to therapy and selection of drug-resistant HBV variants (resistance) (A1).
⋅HBV resistance to NUCs is characterized by selection of HBV variants with amino acid substitutions that confer reduced susceptibility to the administered NUC(s). Resistance may result in primary treatment failure or virological breakthrough on therapy (A1).
4.5. Results of current therapies
Seven drugs are now available for the treatment of chronic hepatitis B: they include conventional interferon alpha, pegylated interferon alpha and NUCs. NUCs for HBV therapy belong to three classes: L-nucleosides (lamivudine, telbivudine, and emtricitabine), deoxyguanosine analogues (entecavir) and acyclic nucleoside phosphonates (adefovir and tenofovir). Lamivudine, adefovir, entecavir, telbivudine and tenofovir have been approved in Europe for HBV treatment, and the combination of tenofovir and emtricitabine in one tablet has been licensed for the treatment of HIV infection.
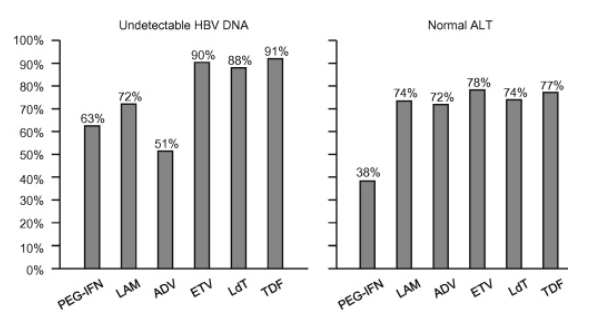
The efficacy of these drugs has been assessed in randomized controlled trials at one year (two years with telbivudine). Longer-term results (up to 5 years) are available for lamivudine, adefovir, entecavir, telbivudine and tenofovir in patient subgroups. Fig. 1, Fig. 2 show response rates with these drugs from different trials. These trials used different HBV DNA assays and they were not head-to-head comparisons for all the drugs.
(1)In HBeAg-positive patients, virological response rates at one year (undetectable HBV DNA, defined variously in the different trials and differently from the present guidelines) were 25%, 36-40%, 21%, 67%, 60% and 74% with pegylated interferon alpha-2a/2b, lamivudine, adefovir, entecavir, telbivudine and tenofovir, respectively (Fig. 1) [38], [39], [40], [41], [42], [43], [44]. HBe seroconversion rates were of the order of 30% with conventional and pegylated interferon alpha and approximately 20% for NUCs. HBe seroconversion rates increase with continued NUCs treatment, but are affected if resistance occurs (B1). Loss of HBsAg rates after one year were 3-4% with pegylated interferon alpha, 1% with lamivudine, 0% with adefovir, 2% with entecavir, 0% with telbivudine, and 3% with tenofovir.
(2)In HBeAg-negative patients, virological response rates at one year (undetectable HBV DNA, defined variously in the different trials and differently from the present guidelines) were 63%, 72%, 51%, 90%, 88% and 91% with pegylated interferon alpha-2a, lamivudine, adefovir, entecavir, telbivudine and tenofovir, respectively (Fig. 2) [41], [45], [46], [47], [48], [49]. Loss of HBsAg rates after one year were 3% with pegylated interferon alpha and 0% with lamivudine, adefovir, entecavir, telbivudine or tenofovir.
Fig. 1. Rates of HBe seroconversion, undetectable HBV DNA and normal ALT at one year of therapy with pegylated interferon alpha-2a (PEG-IFN), lamivudine (LAM), adefovir (ADV), entecavir (ETV), telbivudine (LdT) and tenofovir (TDF) in HBeAg-positive patients with CHB in randomized clinical trials. These trials used different HBV DNA assays and they were not head-to-head comparisons for all the drugs; thus, these numbers are only indicative and should be considered with caution.
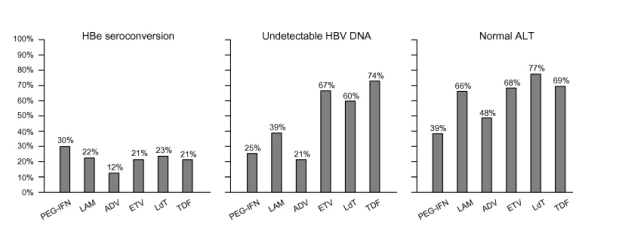
Fig. 2. Rates of undetectable HBV DNA and normal ALT at one year of therapy with pegylated interferon alpha-2a (PEG-IFN), lamivudine (LAM), adefovir (ADV), entecavir (ETV), telbivudine (LdT) and tenofovir (TDF) in HBeAg-negative patients with CHB in randomized clinical trials. These trials used different HBV DNA assays and they were not head-to-head comparisons for all the drugs; thus, these numbers are only indicative and should be considered with caution.
4.6. Indications for treatment
The indications for treatment are generally the same for both HBeAg-positive and HBeAg-negative CHB. This is based mainly on the combination of three criteria:
⋅Serum HBV DNA levels.
⋅Serum aminotransferase levels.
⋅Histological grade and stage.
Patients should be considered for treatment when HBV DNA levels are above 2000 IU/ml (i.e. approximately 10,000 copies/ml) and/or the serum ALT levels are above the upper limit of normal (ULN) for the laboratory, and liver biopsy (or non-invasive markers when validated in HBV-infected patients) shows moderate to severe active necroinflammation and/or fibrosis using a standardised scoring system (for example at least grade A2 or stage F2 by METAVIR scoring) (A1). Indications for treatment must also take into account age, health status, and availability of anti-viral agents in individual countries.
The following special groups of patients should be considered:
⋅Immunotolerant patients: most patients under 30 years of age with persistently normal ALT levels and a high HBV DNA level (usually above 107 IU/ml), without any suspicion of liver disease and without a family history of HCC or cirrhosis do not require immediate liver biopsy or therapy. Follow-up is mandatory (B1).
⋅Patients with mild CHB: patients with slightly elevated ALT (less than 2 times ULN) and mild histological lesions (less than A2F2 with METAVIR scoring) may not require therapy. Follow-up is mandatory (B1).
⋅Patients with compensated cirrhosis and detectable HBV DNA may be considered for treatment even if ALT levels are normal and/or HBV DNA levels are below 2000 IU/ml (i.e. approximately 10,000 copies/ml) (B1).
⋅Patients with decompensated cirrhosis require urgent antiviral treatment. Rapid and profound viral suppression and efficacious prevention of resistance are particularly needed in this group. Significant clinical improvement can be associated with control of viral replication, but patients with very advanced liver disease may not always benefit if treated at this late stage and should be considered for liver transplantation (A1).
4.7. Predictors of response
Certain general baseline and on-treatment predictors of subsequent response have been identified. Predictors of response for the existing antiviral therapies at various time points vary for different agents.
(1)For interferon alpha-based treatment:
⋅Pre-treatment factors predictive of HBe seroconversion are low viral load (HBV DNA below 107 IU/ml or 7 log10 IU/ml), high serum ALT levels (above 3 times ULN), and high activity scores on liver biopsy (at least A2) [50], [51], [52] (B2).
⋅During treatment, an HBV DNA decrease to less than 20,000 IU/ml at 12 weeks is associated with a 50% chance of HBe seroconversion in HBeAg-positive patients and with a 50% chance of sustained response in HBeAg-negative patients [53], [54].
⋅During treatment, HBeAg decrease at week 24 may predict HBe seroconversion [54], [55] (B2).
⋅Further studies are needed to determine the role of HBsAg quantitation to predict sustained virological response and HBsAg loss.
⋅HBV genotype A and B have been shown to be associated with a better response to interferon alpha than genotypes C and D [56]. However, the HBV genotype has a poor individual predictive value and currently, genotype alone should not override the choice of treatment (B2).
(2)For NUCs treatment:
⋅Pre-treatment factors predictive of HBe seroconversion are low viral load (HBV DNA below 107 IU/ml or 7 log10 IU/ml), high serum ALT levels (above 3 times ULN), high activity scores on liver biopsy (at least A2) [52].
⋅During treatment with lamivudine, adefovir or telbivudine, a virological response at 24 or 48 weeks (undetectable HBV DNA in a real-time PCR assay) is associated with a lower incidence of resistance, i.e. an improved chance of maintained virological response, and HBe seroconversion in HBeAg-positive patients [41], [46], [57] (B1).
⋅HBV genotype does not influence the response to any NUC.
4.8. Treatment strategies: how-to-treat
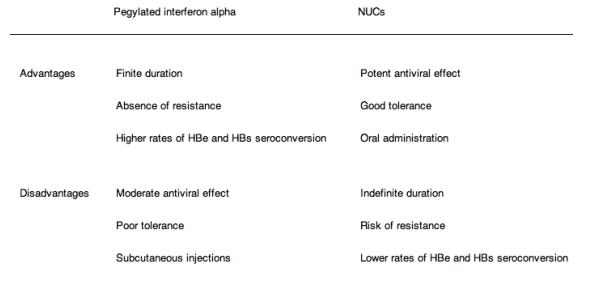
The main theoretical advantages of interferon alpha (conventional or pegylated) are the absence of resistance and the potential for immune-mediated containment of HBV infection with an opportunity to obtain a sustained virological response off-treatment and a chance of HBsAg loss in patients who achieve and maintain undetectable HBV DNA. Frequent side effects and subcutaneous injection are the main disadvantages of interferon alpha treatment. Interferon alpha is contraindicated in patients with decompensated HBV-related cirrhosis or autoimmune disease and in those with uncontrolled severe depression or psychosis (A1).
Entecavir and tenofovir are potent HBV inhibitors and they have a high barrier to resistance [38], [58], [59]. Thus they can be confidently used as first-line monotherapies (A1). The role of monotherapy with entecavir or tenofovir could be modified if higher rates of resistance become apparent with longer treatment duration.
Adefovir is more expensive than tenofovir, is less efficacious, and engenders higher rates of resistance (A1). Telbivudine is a potent inhibitor of HBV but, due to a low genetic barrier to resistance, a high incidence of resistance has been observed in patients with high baseline levels of replication and in those with detectable HBV DNA after 24 weeks of therapy [41] (A1). Lamivudine is an inexpensive agent, but engenders very high rates of resistance with monotherapy [60], [61] (A1).
Several treatment options exist for individual patients, making rational choices for first- and second-line treatment sometimes difficult. Two different treatment strategies are applicable in both HBeAg-positive and HBeAg-negative CHB patients: treatment of finite duration with pegylated interferon alpha or NUCs and long-term treatment with NUCs.
(1)Treatment of finite duration with pegylated interferon alpha or NUCs. This strategy is intended to achieve a sustained virological response off-treatment (A1).
⋅Finite-duration treatment with pegylated interferon alpha: a 48-week course of pegylated interferon alpha is mainly recommended for HBeAg-positive patients with the best chance of HBe seroconversion. It can also be used for HBeAg-negative patients who have the best chance of a sustained response off-treatment. In both groups, these are patients with high baseline ALT (>3 times ULN) and HBV DNA less than 2_106 IU/ml (approximately 107 copies/ml) or 6.3 log10 IU/ml at baseline. Full information about the advantages, adverse events and inconveniences of pegylated interferon alpha versus NUCs (Table 2) should be provided so the patient can participate in the decision. (B2)
Table 2. Main respective advantages and disadvantages of pegylated interferon alpha and NUCs in the treatment of CHB
The combination of pegylated interferon alpha with lamivudine showed a higher on-treatment response but did not show a higher rate of sustained response. There is limited information on the efficacy and safety of combination of pegylated interferon alpha with other NUCs and presently this type of combination is not recommended.
⋅Finite-duration treatment with NUCs is achievable for HBeAg-positive patients who develop HBe seroconversion on treatment. However, duration is unpredictable prior to therapy as it depends on when HBe seroconversion occurs. HBe seroconversion is more frequent in patients with high baseline ALT (>3 times ULN) and HBV DNA less than 2_106 IU/ml (approximately 107 copies/ml) or 6.3 log10 IU/ml at baseline (A1). An attempt at finite treatment should use the most potent agents with the highest barrier to resistance (entecavir or tenofovir) to rapidly reduce levels of viremia to undetectable levels and avoid rebounds due to HBV resistance (A1). Telbivudine might be used in patients with good predictors of response (HBV DNA <2_106 IU/ml, i.e. approximately 107 copies/ml, or 6.3 log10 IU/ml at baseline) with verification of HBV DNA suppression below detection in real-time PCR assay at 24 weeks. Once HBe seroconversion occurs on NUC, treatment should be prolonged for an additional 6 to (preferentially) 12 months; a durable response (persistence of anti-HBe antibodies off-treatment) can be expected in 80% of these patients (B1).
(2)Long-term treatment with NUCs. This strategy is necessary for patients who cannot achieve a sustained virological response off-treatment and require extended therapy, i.e. for HBeAg-positive patients who do not develop HBe seroconversion and in HBeAg-negative patients. This strategy is also recommended in patients with cirrhosis irrespective of HBeAg status or HBe seroconversion on treatment (A1).
The most potent drugs with the optimal resistance profile, i.e. tenofovir or entecavir, should be used as first-line monotherapies (A1). It is optimal to maintain HBV DNA suppression to undetectable HBV DNA in real-time PCR, whatever the drug used (B1). The long-term effects, safety and tolerability of entecavir and tenofovir (i.e. after five to ten years) are still unknown.
There are as yet no data to indicate an advantage of de novo combination treatment with NUCs in naive patients receiving either entecavir or tenofovir (C1). Therapeutic trials are in progress. Some experts recommend a de novo combination therapy approach to prevent potential resistance in patients with a high likelihood of developing resistance (high baseline HBV DNA levels) or in whom the occurrence of viral resistance would be life-threatening due to the underlying condition (cirrhosis). However, the long-term safety of the combination of NUCs, and in particular of the combination of entecavir and tenofovir is unknown and this approach is costly (B2). Tenofovir plus lamivudine, or tenofovir plus emtricitabine in one tablet, may be considered de novo for these patients (C1).
4.9. Treatment failure
It is important to distinguish between primary non-response (less than 1 log10 drop of HBV DNA at 12 weeks), partial virological response (detectable HBV DNA on real-time PCR assay during continuous therapy) and virological breakthrough due to antiviral drug resistance [29], [62].
(1)Primary non-response. Primary non-response seems to be more frequent with adefovir (approximately 10-20%) than with other NUCs because of suboptimal dosing. A rapid switch to tenofovir or entecavir is recommended (B1). Primary non-response is rarely observed with lamivudine, telbivudine, entecavir or tenofovir. In patients with primary non-response, it is important to check for compliance. In a compliant patient with a primary non-response, identification of possible HBV resistance mutations can formulate a rescue strategy that must reasonably be based on an early change to a more potent drug that is active against the resistant HBV variant (B1).
(2)Partial virological response. Partial virological response may be encountered with all available NUCs. It is important to check for compliance. In patients receiving lamivudine, adefovir or telbivudine with a partial virological response at week 24, two strategies can be used: change to a more potent drug (entecavir or tenofovir) or addition of a more potent drug that does not share cross-resistance (add tenofovir to lamivudine or telbivudine, or add entecavir to adefovir) (A1). In patients receiving entecavir or tenofovir with a partial virological response at week 48, some experts would suggest adding the other drug in order to prevent resistance in the long term (C1). The long-term safety of tenofovir and entecavir in combination is however unknown.
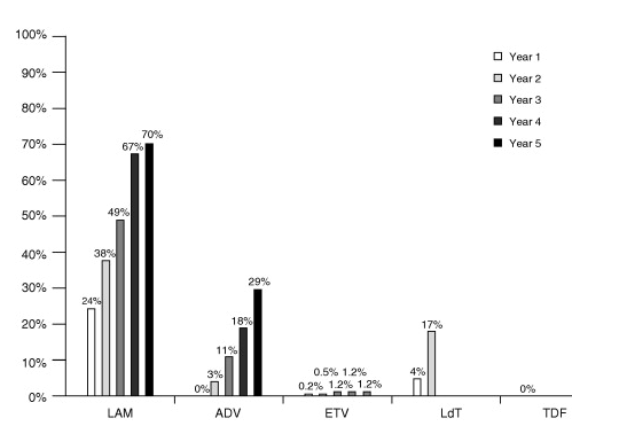
(3)Virological breakthrough. Virological breakthrough in compliant patients is related to viral resistance. Rates of resistance at up to 5 years of administration are shown for the different NUCs in Fig. 3. Resistance is associated with prior treatment with NUCs (i.e., lamuvidine, adefovir, telbivudine, emtricitabine) or, in treatment-naive patients, with high baseline HBV DNA levels, a slow decline in HBV DNA and partial virological response during treatment. Resistance should be identified as early as possible before clinical breakthrough (increased ALT) by means of HBV DNA monitoring, and if possible identification of the pattern of resistance mutations should be used to adapt therapeutic strategies. Indeed, clinical and virological studies have demonstrated the benefit of an early treatment adaptation, as soon as viral load increases [52], [63] (A1).
Fig. 3. Cumulative incidence of HBV resistance to lamivudine (LAM), adefovir (ADV), entecavir (ETV), telbivudine (LdT) and tenofovir (TDF) in published pivotal trials in NUC-naive patients. For method of calculation, see ref. [29]. These trials included different populations, used different exclusion criteria and different follow-up endpoints.
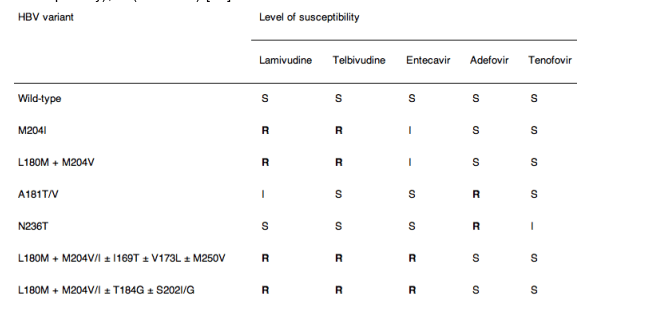
In case of resistance, an appropriate rescue therapy should be initiated with the most effective antiviral effect and the minimal risk to induce multiple drug-resistant strains. Therefore, adding-on a second drug without cross-resistance is the only efficient strategy. Table 3 shows cross-resistance data for the most frequent resistant HBV variants [64]. The safety of some combinations in the long term is unknown.
⋅ Lamivudine resistance: add tenofovir (add adefovir if tenofovir not yet available) (B1).
⋅ Adefovir resistance: it is recommended to switch to tenofovir if available and add a second drug without cross-resistance. If an N236T substitution is present, add lamivudine, entecavir or telbivudine or switch to tenofovir plus emtricitabine (in one tablet) (C1). If an A181T/V substitution is present, add entecavir (the safety of the tenofovir-entecavir combination is unknown) or switch to tenofovir plus emtricitabine (B1).
⋅ Telbivudine resistance: add tenofovir (add adefovir if tenofovir not yet available). The long-term safety of these combinations is unknown (C1).
⋅ Entecavir resistance: Add tenofovir (the safety of this combination is unknown) (C1).
⋅ Tenofovir resistance: resistance to tenofovir has not been described so far. It is recommended that genotyping and phenotyping be done by an expert laboratory to determine the cross-resistance profile. Entecavir, telbivudine, lamivudine or emtricitabine could be added (the safety of these combinations is unknown) (B1).
Table 3. Cross-resistance data for the most frequent resistant HBV variants. The amino-acid substitution profiles are shown in the left column and the level of susceptibility is given for each drug: S (sensitive), I (intermediate/reduced susceptibility), R (resistant) [64]
l should be assessed at weeks 12 and 24 to verify primary response.
⋅In HBeAg-positive patients, HBeAg and anti-HBe antibodies should be checked at weeks 24 and 48 and 24 weeks post-treatment. HBe seroconversion together with ALT normalisation and serum HBV DNA below 2000 IU/ml (approximately 10,000 copies/ml), i.e. 3.3 log10 IU/ml, is the desired outcome (A1). Undetectable serum HBV DNA by real-time PCR during follow-up is the optimal outcome since it is associated with a high chance of HBsAg loss. HBeAg-positive patients who develop HBe seroconversion with pegylated interferon or NUCs require long follow-up because of the possibility of HBe seroreversion or HBeAg-negative chronic hepatitis B. HBsAg should be checked at 6-month intervals after HBe seroconversion if HBV DNA is undetectable. Quantitative HBsAg assay is still a research tool. In case of a primary non-response, i.e. failure to achieve a 1 log10 reduction from baseline at 12 weeks, interferon treatment should be stopped and replaced by a NUC (B1).
⋅HBeAg-negative patients should be similarly monitored for efficacy and safety through 48 weeks of treatment. A virological response with HBV DNA <2000 IU/ml (approximately 10,000 copies/ml), i.e. 3.3 log10 IU/ml, is generally associated with remission of the liver disease. Undetectable HBV DNA in real-time PCR is the ideal desired off-treatment sustained response with a high probability of HBsAg loss in the longer term. HBsAg should be checked at 6-month intervals if HBV DNA is undetectable (B1).
All patients treated with pegylated interferon alpha should be monitored for the known adverse effects of interferon.
4.10.2. Finite treatment with NUCs in HBeAg-positive patients
The objective of finite treatment with NUCs is HBe seroconversion. HBV DNA should be measured every 12 weeks. HBV DNA suppression to undetectable levels in real-time PCR and subsequent HBe seroconversion is associated with biochemical and histological responses. Studies have suggested that NUC therapy can be stopped 24 to 48 weeks after HBe seroconversion (B1). HBsAg should be checked at 6-month intervals after HBe seroconversion. HBsAg loss is however rarely observed after NUC therapy.
4.10.3. Long-term therapy with NUCs
HBV DNA levels should be monitored at week 12 to ascertain virological response and then every 12 to 24 weeks. HBV DNA reduction to undetectable levels by real-time PCR (i.e. below 10-15 IU/ml) should ideally be achieved to avoid resistance. HBV DNA monitoring is thus critical to detect treatment failure (A1). In HBeAg-positive patients, HBeAg and subsequently anti-HBe antibodies once HBeAg is negative should be measured at intervals of 6 to 12 months.
NUCs are cleared by the kidneys, and appropriate dosing adjustments are recommended for patients with reduced creatinine clearance (A1). Drug concentrations are comparable in patients with varying degrees of hepatic impairment but this has not been fully studied. Exacerbations of hepatitis B may occur and require more intensive monitoring (monthly in the first three months) in patients with cirrhosis. The onset of complications in these patients requires urgent management (B1). Renal impairment has rarely been reported in patients with HIV infection receiving anti-HBV drugs, or in patients receiving nephrotoxic drugs and treated with tenofovir or adefovir 10mg/day and appropriate monitoring for nephrotoxicity and dose adjustments is necessary.
Decreases in bone mineral density have rarely been reported in HIV-positive patients treated with tenofovir (B2). Long-term study is needed. Long-term monitoring for carcinogenesis with entecavir is ongoing. Myopathy has rarely been reported in CHB patients treated with telbivudine. Peripheral neuropathy has been observed in patients treated with pegylated interferon and telbivudine; this combination should be avoided (B1).
4.11. Treatment of patients with severe liver disease
4.11.1. Treatment of patients with cirrhosis
Treatment of patients with cirrhosis should not be based on ALT levels, as these may be normal in advanced disease. Interferon alpha increases the risk of sepsis and decompensation in patients with advanced cirrhosis. However, interferon can be used for the treatment of well compensated cirrhosis [65] (A1). The use of potent NUCs with very low risk of resistance, i.e. tenofovir or entecavir, is particularly relevant in this group of patients (B1). Close monitoring of HBV DNA levels is important and resistance must be prevented by adding a second drug without cross-resistance if HBV DNA is not undetectable at week 48 of therapy. If lamivudine has to be prescribed (because of local policy), it should be used in combination with adefovir or preferably tenofovir (B1).
Hepatic decompensation may occur with exacerbations of disease that must be distinguished from non-compliance and resistance [40]. Thus patients with cirrhosis require long-term therapy, with careful monitoring for resistance and flares. Clinical studies indicate that prolonged and adequate suppression of HBV DNA may stabilize patients and delay or even obviate need for transplantation [37], [66] (B1). Partial regression of fibrosis has been reported.
4.11.2. Treatment of patients with decompensated cirrhosis
Patients with decompensated cirrhosis should be treated in specialized liver units, as the application of antiviral therapy is complex, and these patients may be candidates for liver transplantation. End-stage liver disease should be treated as a matter of urgency. Treatment is indicated even if HBV DNA level is low in order to prevent recurrent reactivation. Potent NUCs with good resistance profiles (entecavir or tenofovir) should be used. However, there are little data for the safety of these agents in decompensated cirrhosis (B1). Patients may show slow clinical improvement over a period of 3-6 months. However some patients with advanced hepatic disease with a high Child-Pugh or MELD score may have progressed beyond the point of no return, and may not benefit, thus requiring transplantation if possible [67]. In that situation, treatment with NUCs will decrease the risk of HBV recurrence in the graft.
4.12. Prevention of recurrent hepatitis B after liver transplantation
Recurrent HBV infection in the transplanted liver has previously been a major problem. Pre-transplant therapy with a potent NUC with a high barrier to resistance is recommended for all HBsAg-positive patients undergoing liver transplantation for HBV-related end-stage liver disease or HCC, to achieve the lowest possible level of HBV DNA before transplantation [68], [69], [70] (A1). To date, lamivudine and/or adefovir have been given post-transplant in combination with hepatitis B immunoglobulin (HBIg). This regimen has reduced the risk of graft infection to less than 10%. Adefovir has been successfully added for lamivudine resistance. Shorter courses and lower doses of HBIg and other forms of prophylaxis, including adefovir in combination with lamivudine and entecavir, are being studied. Efficacy and safety data with newer, more potent NUCs with lower rates of resistance, i.e. entecavir and tenofovir, have not been published but these agents should be considered, as profound suppression and low rates of resistance are advantageous (B1). Antiviral therapy for prophylaxis of recurrent hepatitis B probably requires life-long continuation of treatment (B1).
4.13. Treatment in special patient groups
4.13.1. HIV co-infected patients
HIV-positive patients with CHB are at increased risk of cirrhosis [71], [72], [73], [74], [75], [76]. Treatment of HIV may lead to flares of hepatitis B due to immune restitution. The indications for therapy are the same as in HIV-negative patients, based on HBV DNA levels, serum ALT levels and histological lesions [77]. In agreement with recent HIV guidelines, it is recommended that most coinfected patients be simultaneously treated for both HIV and HBV de novo [78]. Tenofovir and emtricitabine (FTC) together, plus a third agent active against HIV, are indicated [79] (A1). In a small number of patients, HBV may have to be treated before HIV; adefovir and telbivudine, which are not proven to be active against HIV, should be preferred. Lamivudine, entecavir and tenofovir have activity against both HIV and HBV and are contraindicated as single agents for hepatitis B in coinfected patients (A1). However, if these drugs with a low barrier to resistance do not reach the goal of undetectable HBV DNA, treatment of HIV infection should be envisaged.
4.13.2. HDV co-infected patients
Active co-infection with HDV is confirmed by the presence of detectable HDV RNA, immuno-histochemical staining for HDV antigen, or IgM anti-HDV. Interferon alpha (conventional or pegylated) is the only drug effective on HDV replication [80], [81], [82], [83], [84], [85]. The efficacy of interferon alpha therapy should be assessed at 24 weeks by measuring HDV RNA levels. More than one year of therapy may be necessary, but is of unproven efficacy [86] (B2). A proportion of patients become HDV RNA-negative or even HBsAg-negative, with accompanying improvement in histology. NUC monotherapy does not appear to impact HDV replication and related disease.
4.13.3. HCV co-infected patients
HBV DNA level is often low or is undetectable and HCV is responsible for the activity of chronic hepatitis in most patients, although this is variable. Thus patients should receive pegylated interferon alpha with ribavirin as for HCV [87] (B1). Sustained virological response (SVR) rates for HCV are broadly comparable with HCV monoinfected patients [88], [89], [90], [91]. There is a potential risk of HBV reactivation during or after clearance of HCV that must then be treated with NUCs (B1).
4.13.4. Acute severe hepatitis
More than 95-99% of adults with acute HBV infection will recover spontaneously and seroconvert to anti-HBs without anti-viral therapy. However, some patients with fulminant hepatitis or severe protracted subacute hepatic necrosis may benefit from NUC treatment. Support for such a strategy may be found in a small number of reports with lamivudine but the efficacy is unproven (B1). As for chronic hepatitis, more potent drugs with a high barrier to resistance, i.e. entecavir or tenofovir, should be used. The duration of treatment is not established. However, continuation of anti-viral therapy for at least 3 months after seroconversion to anti-HBs or at least 6 months after HBe seroconversion without HBsAg loss is recommended (B2). Sometimes, the distinction between true acute hepatitis B and reactivation of chronic hepatitis B may be difficult and may require liver biopsy. However, in both cases NUC treatment is the treatment of choice [92], [93], [94].
4.13.5. Children
Chronic hepatitis B causes benign disease in most children. Only conventional interferon alpha, lamivudine and adefovir have been evaluated for safety and efficacy comparable to adults [95], [96], [97], [98]. There are ongoing studies of other NUCs in children to better define treatment strategies for children.
4.13.6. Healthcare workers
Healthcare workers, especially surgeons, involved in exposure-prone procedures who are HBsAg-positive with HBV DNA 2000 IU/ml or 3.3 log10 IU/ml should be treated with a potent antiviral agent with a high barrier to resistance (i.e. entecavir or tenofovir), to reduce levels of HBV DNA ideally to undetectable and at least to <2000 IU/ml before resuming exposure-prone procedures (B1). The long-term safety, efficacy, complications and economic implications of such a policy in different countries are unknown [99].
4.13.7. Pregnant women
Lamivudine, adefovir and entecavir are listed by the FDA as pregnancy category C drugs, and telbivudine and tenofovir as category B drugs. These classifications are based on the risk of teratogenicity in preclinical evaluation. There is a considerable body of safety data in pregnant HIV-positive women who have received tenofovir and/or lamivudine or emtricitabine [100]. Recent reports suggest that lamivudine therapy during the last trimester of pregnancy in pregnant HBsAg-positive women with high levels of viremia reduces the risk of intra-uterine and perinatal transmission of HBV if given in addition to passive and active vaccination by HBIg and HBV vaccination [101]. Tenofovir or tenofovir with emtricitabine or entecavir could be considered. Although apparently safe, these protocols require further confirmation (B2). HBV-infected women should be monitored closely after delivery as exacerbations of chronic hepatitis B may occur [102].
4.13.8. Pre-emptive therapy before immunosuppressive therapy or chemotherapy
In HBV carriers receiving chemotherapy or immunosuppressive therapy, the risk of reactivation is high, particularly if rituximab is given alone or in combination with steroids [103]. All candidates for chemotherapy and immunosuppressive therapy should be screened for HBsAg and anti-HBc antibodies prior to initiation of treatment [104], [105]. Vaccination against HBV in seronegative patients is highly recommended.
HBsAg-positive candidates for chemo- and immunosuppressive therapy should be tested for HBV DNA levels and receive pre-emptive NUC administration during therapy (regardless of HBV DNA levels) and for 12 months after cessation of therapy. Most experience with pre-emptive treatment has been with lamivudine, which may suffice for patients with low HBV DNA levels and a low risk of resistance [103], [106], [107], [108]. It is however recommended that patients, especially those with a high HBV DNA level, be protected with a NUC with high antiviral potency and a high barrier to resistance, i.e. entecavir or tenofovir (A1).
HBsAg-negative patients with positive anti-HBc antibodies and undetectable HBV DNA in the serum who receive chemotherapy and/or immunosuppression should be followed carefully by means of ALT and HBV DNA testing and treated with NUC therapy upon confirmation of HBV reactivation before ALT elevation. NUC prophylaxis is also recommended in patients receiving bone marrow transplantation from a non-immune donor.
Recipients of anti-HBc-positive liver grafts should receive NUC prophylaxis combined with HBIg (A1). The optimal duration of combined prophylaxis is not known.
4.13.9. Dialysis and renal transplant patients
Most data in this group are available for lamivudine; the dose of lamivudine should be adapted for renal failure [109] (A1). There are reports of worsening of renal graft function in patients treated with adefovir. Entecavir may be the optimal choice of drug for patients undergoing renal transplantation. Tenofovir should be used with caution in renal impairment (B1).
4.13.10. Extrahepatic disease
HBsAg-positive patients with extra-hepatic manifestations and active HBV replication may respond to antiviral therapy. Lamivudine has been most widely used to date. Entecavir and tenofovir are expected to have enhanced efficacy in this group and the indications and management do not differ from patients without extra-hepatic manifestations. Plasmapheresis can be useful in addition to NUC therapy in special cases (C2).
5. Unresolved issues and unmet needs
(1)Improve knowledge of the natural history, in particular of immunotolerant patients, with long-term follow-up of cohorts: experimental studies to provide more definite prognostic information, and biomarkers to determine prognosis and indications for treatment.
(2)Develop and assess new therapeutic approaches, particularly immunomodulatory therapies to enhance loss of HBeAg and HBsAg and subsequent seroconversion.
(3)Assess the role of indirect markers (serum and biophysical) to assess the severity of liver disease and for the follow-up of treated and untreated patients.
(4)Assess the role of HBV genotype to determine prognosis and response to therapy and the risk of resistance.
(5)Assess the efficacy of different durations (24 weeks to 2 years) and lower doses of pegylated interferon alpha.
(6)Assess long-term efficacy and safety and resistance to new analogues (entecavir, telbivudine and tenofovir).
(7)Better define monitoring algorithms: timing of HBV DNA measurement with the new generation of NUCs with a high genetic barrier to resistance; role of genotypic resistance assays in adapting therapy.
(8)Assess the role of combination therapy with two NUCs to reduce resistance.
(9)Assess the efficacy of the combination of pegylated interferon alpha with potent NUCs (entecavir or tenofovir) to increase HBe and HBs seroconversion rates.
(10)Develop new drugs to manage multidrug resistant HBV resistant to both lineages of current NUCs.
(11)Assess long-term impact of therapy on the prevention of cirrhosis and its complications and HCC.
(12)Develop effective and optimum treatment for HDV coinfection.
|
|
| |
| |
|
|
|