 |
 |
 |
| |
Pharmacokinetic interaction between darunavir in combination with low-dose ritonavir and buprenorphine/naloxone
|
| |
| |
Reported by Jules Levin
ICCAC Sept 11 2009 San Francisco
Sekar V,1 Tomaka F,1 Meyvisch P,2 De Pauw M,2 Spinosa-Guzman S,2 Vangeneugden T,2 Hoetelmans R2
1Tibotec Inc., Yardley, PA, USA; 2Tibotec BVBA, Mechelen, Belgium
AUTHOR CONCLUSIONS
· These results demonstrate that when DRV/r 600/100mg bid was added to a stable, individualized BUP/NLX maintenance therapy, BUP exposure (Cmax, AUC24h) was comparable with that following BUP/NLX alone. Similar results are expected with the DRV/r qd dose.
· In contrast, norBUP exposure was increased (Cmax: 36%; AUC24h: 46%); the clinical relevance of the increased norBUP exposure in the presence of DRV/r is unknown.
· The combination of BUP/NLX and DRV/r was generally well tolerated.
· Based on these findings, no adjustment of BUP/NLX dosage is required during DRV/r coadministration. However, clinical monitoring is recommended as such maintenance therapy may need to be altered in some patients.
Introduction
· Substance abuse is a common comorbidity among HIV-infected patients, and untreated, can result in poor HIV treatment outcomes.1 The most effective treatment for opioid dependence is opioid maintenance therapy, which helps to reduce cravings and prevent the use of illicit substances.
· Buprenorphine (BUP) is a partial opioid receptor agonist, and naloxone (NLX) is an opioid antagonist. In combination, BUP and NLX are an effective maintenance pharmacotherapy for opioid dependence.2
· The HIV-1 protease inhibitor darunavir (DRV) in combination with low-dose ritonavir (DRV/r; RTV) is an approved treatment for
- treatment-naïve, HIV-1-infected adults (800/100mg qd) in the USA,3 Europe4 and other countries
- treatment-experienced, HIV-1-infected adults (600/100mg bid) in the USA,3 Europe4 and in many more countries
- treatment-experienced, HIV-1-infected pediatric patients aged ≥6 years (bid bodyweight-based dose) in the USA2 and the EU.
· Pharmacokinetic drug-drug interactions between BUP/NLX and DRV/r might be expected based on their hepatic metabolism. All of these drugs are metabolized by cytochrome P450 (CYP) 3A4.5,6 The main, and active, metabolite of BUP is norbuprenorphine (norBUP). RTV and DRV are inhibitors of CYP3A4 metabolism, with RTV being the most potent.5
· The primary objective of this study was to investigate the effect of multiple doses of DRV/r on the steady-state pharmacokinetics of BUP, norBUP and NLX. Secondary objectives included assessing the effect of BUP/NLX on the pharmacokinetics of DRV and RTV, evaluating pharmacodynamic effects (opiate withdrawal or excess), and determining the short-term safety and tolerability.
METHODS
Study design
· TMC114-TiDP3-C171 was a Phase I, open-label, add-on trial in HIV-negative healthy volunteers on stable, individualized BUP/NLX maintenance therapy.
· BUP/NLX maintenance therapy consisted of sublingual tablets (Suboxone, maximum dose 24/6mg qd), for at least 2 weeks prior to study start. Enrolled volunteers also received DRV/r 600/100mg bid for 7 days.
· Safety assessments were performed on an on-going basis. Signs and symptoms of opiate withdrawal and excess were assessed using physical examinations and questionnaires.
· The study protocol and amendments were reviewed and approved by the appropriate institutional review board health authorities, and the study was conducted in accordance with the Declaration of Helsinki. Written informed consent was obtained from all volunteers.
Pharmacokinetic analyses
· For full pharmacokinetic profiles, blood samples were collected for BUP, norBUP and NLX on Days -1 and 7, and for DRV and RTV on Day 7. Blood sampling times were predose and at 0.5, 1, 1.5, 2, 3, 4, 5, 6, 9, 12 and 24 hours post-dose.
· Plasma concentrations of BUP, norBUP, NLX, DRV and RTV, were determined using validated liquid chromatography-tandem mass spectrometry methods. The lower levels of quantification were 0.1ng/mL for BUP, norBUP and NLX, and 5.0ng/mL for DRV and RTV.
· Pharmacokinetic parameters included area under the plasma-concentration time curve from administration up to 24-hours post-dosing (AUC24h), predose (C0h), maximum (Cmax) and minimum (Cmin) plasma concentrations and time-to-reach maximum concentration (tmax).
· The least square means (LSM) of the primary pharmacokinetic parameters for each treatment group were estimated with a linear mixed-effects model, controlling for treatment as fixed effect, and volunteer as a random effect. The 90% confidence intervals (CIs) around the LSM ratios were calculated.
RESULTS
· Thirty-seven volunteers were screened; 19 were eligible, and two discontinued prior to study start. Seventeen volunteers on stable BUP/NLX received DRV/r, six discontinued (lost to follow-up), and 11 completed the study.
· Most volunteers were male (12/17, 71%), with a median age of 45 years (range: 21-53 years). All (except one) were Caucasian or Black (eight, 47% each), and all were smokers (except one).
· BUP/NLX maintenance therapy was either 16/4mg (n=5) or 8/2mg (n=12) qd.
Pharmacokinetics of BUP
· Steady-state conditions for BUP were achieved prior to sampling on Days -1 and 7 (data not shown).
· BUP mean plasma-concentration curves were comparable for BUP/NLX ± coadministration with DRV/r (Figure 1).
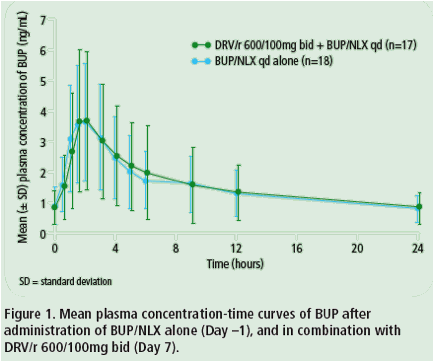
· Mean BUP C0h, Cmin, Cmax, AUC24h and median tmax values were similar when BUP/NLX was administered with or without DRV/r (Table 1).
· Based on the LSM ratios, mean values of C0h, Cmin, Cmax and AUC24h of BUP were decreased by 8%, 2%, 8% and 11%, respectively, in the presence of DRV/r (Table 1).
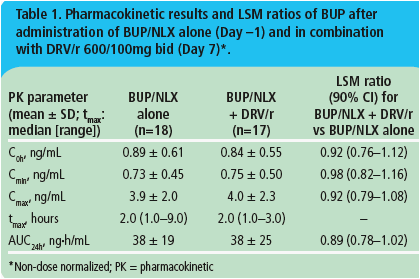
· Inter-individual variability (percent coefficient of variation [% CV]) for C0h, Cmin, Cmax, and AUC24h of BUP was comparable between treatments (50-69%). These high values partly reflect the different doses of BUP (8mg or 16mg qd). However, when the data were dose-normalized the % CV ranged from 38% to 51%.
Pharmacokinetics of norBUP
· Mean norBUP plasma concentrations were higher when BUP/NLX was coadministered with DRV/r (Figure 2).
· Mean norBUP C0h, Cmin, Cmax and AUC24h values were increased during coadministration of DRV/r, compared with BUP/NLX alone (Table 2).
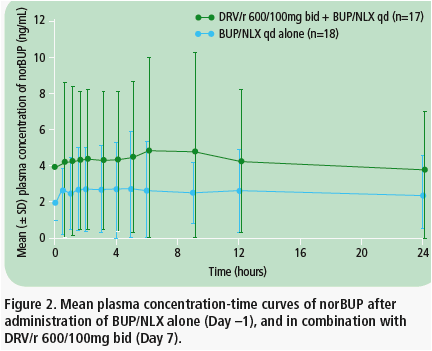
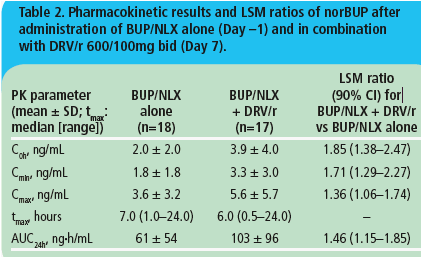
· Based on LSM ratios, mean values of norBUP C0h, Cmin, Cmax and AUC24h increased by 85%, 71%, 36% and 46%, respectively, with DRV/r, compared to BUP/NLX alone (Table 2).
· The inter-individual variability was comparable between treatments for C0h, Cmin, Cmax, and AUC24h, % CV: 87-102% for non-normalized data and 61-70% when the data were normalized for dose.
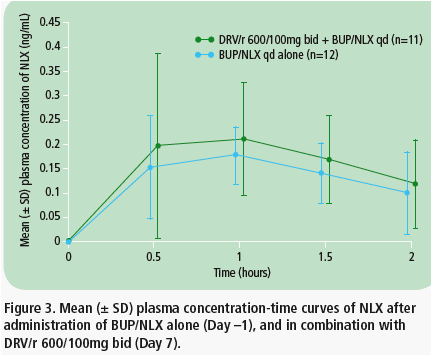
Pharmacokinetics of NLX
· Mean plasma concentration-time curves of NLX were comparable when BUP/NLX was administered ± DRV/r (Figure 3).
· DRV/r coadministration with BUP/NLX had no apparent influence on NLX pharmacokinetic parameters (data not shown).
· C0h and Cmax were below the lower limit of quantification.
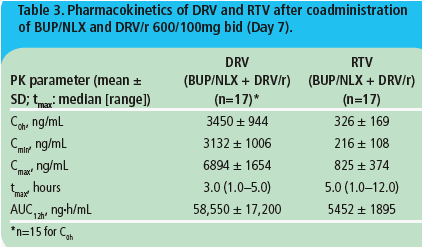
Pharmacokinetics of DRV and RTV
· The main pharmacokinetic parameters of DRV and RTV on Day 7 are shown in Table 3. Exposure to DRV and RTV in the presence of BUP/NLX was comparable to historic data.7,8
Safety and tolerability
· Fourteen (82%) volunteers reported at least one adverse event (AE) during the trial. Overall, the incidence of AEs was higher during coadministration of DRV/r and BUP/NLX, compared with BUP/NLX alone (Table 4).
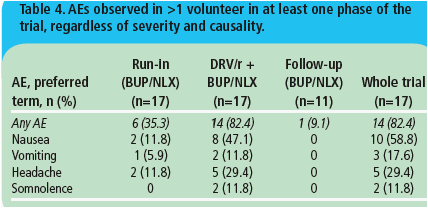
· All AEs were grade 1 in severity. No serious AEs or AEs leading to discontinuation were reported.
· Nausea was the most common AE during the trial, occuring in eight volunteers (47.1%) when BUP/NLX was coadministered with DRV/r, and in two volunteers (11.8%) during BUP/NLX alone.
· During coadministration, physical examinations and questionnaires showed that volunteers experienced either no or mild opiate withdrawal symptoms, and no-to-slight opiate excess symptoms.
· No BUP/NLX dose adjustments were necessary during combined treatment with BUP/NLX and DRV/r for any volunteer.
REFERENCES
1. Sullivan LE, Fiellin DA. Am J Addict 2004;13:1-20.
2. Orman JS, Keating GM. Drugs 2009;69:577-607.
3. PREZISTA (darunavir). Full Prescribing Information. Tibotec Inc. Revised February 2009 [accessed 26 August 2009]. Available from: http://www.prezista.com/prezista/documents/us_package_insert.pdf.
4. PREZISTA (darunavir). EPARs for authorised medicinal products for human use.14 July 2009 [accessed 26 August 2009]. Available from: http://www.emea.europa.eu/humandocs/Humans/EPAR/prezista/prezista.htm.
5. Back D, et al. Antiviral Ther 2008;13:1-13.
6. Suboxone product information. Revised 2007 [accessed 26 August 2009]. Available from: http://www.rxlist.com/suboxone-drug.htm.
7. Sekar V, et al. Antivir Ther 2008;13:563-9.
8. McKeage K, et al. Drugs 2009;69:477-503.
|
| |
|
 |
 |
|
|