 |
 |
 |
| |
Dual Maintenance Therapy with Raltegravir 400 mg BID with Atazanavir 400 mg qD in Patients with no Prior PI Resistance and Intolerance to Other ARV Regimens: Preliminary Report
|
| |
| |
Reported by Jules Levin
ICAAC Sept 12 2009 San Francisco
PJ Ruane, B Alas and PR Wolfe; Light Source Medical Group, Los Angeles, California
from Jules: a concern with nuke-sparing is that there is no evidence that deleting nukes from therapy is safe regarding neurologic disorders. Two studies recently reported a few patients experienced neurologic disorders in these two studies of PI monotherapy:
Unexpectedly high failure rate in LPV/r monotherapy arm, involving CNS and involving low nadir CD4
Unexpectedly high failure rate in LPV/r monotherapy arm, involving CNS, and association with low nadir CD4 count in the MOST study. Reported by Jules Levin ...
www.natap.org/2009/CROI/croi_191.htm
A randomized multicenter study to compare the efficacy of a monotherapy of darunavir to a triple therapy with 2 nucleosides analogues combined to darunavir/r in HIV infected patients with full viral suppression: MONOI ANRS 136- (07/22/09)
AUTHOR DISCUSSION
RAL/ATV was well tolerated and effective in maintaining viral control in
this chronically virologically suppressed population.
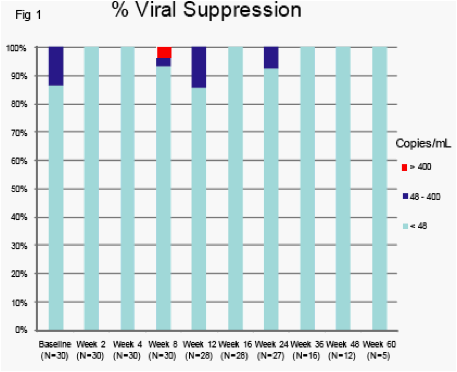
Single blips < 100 copies/mL by TaqMan assay were noted in 4 subjects at baseline and in 7 subjects after enrollment; the significance of this is unclear1.
C24 plasma ATV levels were variable; lower drug levels did not correlate with virological outcome or blips.
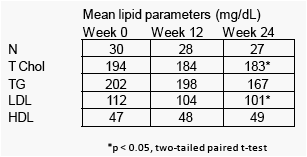
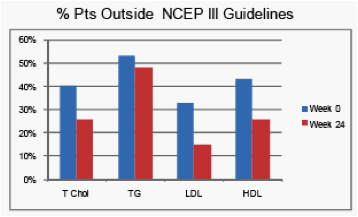
Week 24 serum T Chol and LDL significantly improved from baseline.
Switch from qD to BID in 14 study subjects was not associated with RAL compliance issues.
ABSTRACT (updated)
RAL is a new potent integrase inhibitor; ATV 400mg qD is a potent PI with excellent activity. Data from SwitchMrk suggests RAL performs best with an active antiviral
background. The type and requisite potency of the background regimen to RAL is not established.
Methods: Single center 30 pt, prospective 48 week single arm switch study
Results: 30 patients (median age 47, 100% MSM, 79% white), with consistent long-term viral suppression but intolerance (lipodystrophy, gastrointestinal, hyperlipidemia, other) to elements of current therapy were switched from current regimen (25/30 on prior PI therapy, 19/30 with prior ATV exposure) to RAL 400mg BID and ATV 400mg q day. 2/30 pts discontinued after week 8 and before week 12 (1 viral rebound on concomitant phenytoin, 1 elevated serum creatinine (sCr) that resolved off RAL/ATV). One pt died after 36 weeks. 27 remain on RAL/ATV, with median follow up of 36 weeks. At week 24, 27/27 pts had VL < 400 copies. 7 pts had blips < 100 copies with subsequent return to < 48
copies. CD4 counts were unchanged, lipids were improved and the regimen was well tolerated. Mean plasma ATV level (N=18) was 62.3 ng/mL (95% CI: 41.0-84.7ng/mL).
Conclusions: RAL 400 mg BID/ATV 400 mg qD provides a well-tolerated and effective suppressive maintenance regimen.
INTRODUCTION
While 3-drug regimens remain standard of care, concerns exist regarding the safety of multi-drug regimens taken for a lifetime. Problems with nucleoside analogue therapy prompted successful trials with ritonavir (RTV) boosted PI monotherapy. The GI and lipid tolerability of RTV remains a challenge. Clinical trials have shown raltegravir (RAL) to have potent activity when patients have
few active background drugs; it has a superior lipid profile compared with EFV and LPV/RTV.
Atazanavir (ATV) 400 mg qD without RTV is a potent, well tolerated PI with few GI side effects and lipid disturbances. We hypothesized that RAL/ATV would be a well tolerated and effective regimen for fully suppressed patients experiencing adverse events on their current ARVs.
METHODS
Prospective, observational study conducted at a single research site.
Patients meeting inclusion criteria were changed to RAL/ATV and followed at weeks 2, 4, 8, 16, 24, 36 and 48 weeks after switch.
Viral loads (VL), CD4 cell counts, lipid panels and safety labs were collected.
Medication adherence monitored by self report and raltegravir pill counts at each visit.
Enrollment Criteria:
Age > 18 yr, on stable ARV for at least 2 months with VL < 400 copies/mL for > 4 months and < 50 copies/mL at screening with any CD4 count.
No history of PI resistance or antiretroviral failure while receiving a PI.
Intolerance to current regimen with side effects including, but not limited to, GI, neurological, metabolic, body shape changes and/or dyslipidemia.
No prior RAL treatment nor contraindications to ATV; adequate renal and hepatic function.
Procedures:
Patients discontinued their current regimen and changed to a regimen of study-provided RAL 400 mg BID with or without food and independently procured ATV 400 mg qD with food.
Viral load at study outset was performed by Quest Diagnostics using the Roche Amplicor v.1.5 assay (sensitivity 50 copies/mL); 3 months after study initiation (∼May 2008), Quest adopted the Ampliprep /COBAS TaqMan assay v1.0 (sensitivity 48 copies/mL).
One study visit was scheduled to obtain C24 trough ATV levels with the two prior doses timed 24 hours apart. (ATV levels measured by S. Louie Pharm D., USC, Los Angeles, CA).

RESULTS
27/30 patients remain on study, median duration of 36 weeks (August 2009)
· Subject 006 rebounded at Week 8 to 844 copies, confirmed; noted to be receiving phenytoin. Genotype/phenotype testing demonstrated no PI resistance. C24 hour trough plasma ATV level was 0.
· Subject 007, an African-American man with a prior history of membranous GN, had a rise in sCr from 1.32 mg/dL to 2.25 mg/dL at 6 weeks. His sCr returned to baseline after he resumed his prior regimen.
· Subject 015 died of newly diagnosed lung carcinoma after week 36; his VL was < 48 copies/mL through week 36.
Primary Virological Endpoint (Week 24) % < 400 copies/mL (ITT M=F): 93% (27/29)
Secondary Virological Endpoint (Week 24) % < 48 copies/mL (ITT M=F): 83% (24/29)
Virological results (Fig 1): A "blip" (48-400 copies/mL) was measured in 4 subjects at baseline (range 51-91 copies/mL) and in 7 subjects (range 48-83 copies/mL) up to Week 24. All blip results were measured by TaqMan.
CD4 Results: mean CD4 cell count did not change.
ATZ PK (N=18): Mean plasma C24 ATV level was 62.3 ng/mL (95% CI: 41.0-84.7 ng/mL); median was 62.8 ng/mL (range 0-158 ng/mL). ATV levels in 4 subjects were < 20 ng/mL, 2 of these had levels = 0). Viral loads in these 4 subjects were < 48 copies/mL at all times.
Compliance: 90% of subjects showed > 85% compliance as assessed by RAL pill counts
REFERENCES
1Lima et al, J Acquir Immune Defic Syndr. Volume 51, Number 1, May 1, 2009 : Increased reporting of detectable plasma HIV-1 RNA levels at the critical threshold of 50 copies per milliliter with the TaqMan assay in comparison to the Amplicor assay.
|
| |
|
 |
 |
|
|