 |
 |
 |
| |
Fasted Lipid Changes after Administration of Maraviroc or Efavirenz, both with Zidovudine and Lamivudine, to Treatment-Naive HIV-Infected Patients: 96-Week Results from MERIT
|
| |
| |
Reported by Jules Levin
ICAAC Sept 11-15 2009 San Francisco
A Lazzarin1, G Di Perri2, JG Sierra-Madero3, M Johnson4, J Aberg5, A MacInnes6, H Valdez7, J Heera8, N Rajicic7, J Goodrich8, H Mayer8
1 San Raffaele Scientific Institute, Milan, Italy; 2 Ospedale Amedeo di Savoia, Clinica, Torino, Italy; 3 Instituto Nacional de Ciencias Medicas y Nutricion Salvador Zubiran, Tlalpan, Mexico; 4 Royal Free Hospital, London, UK; 5 New York University School of Medicine, New York, NY, USA; 6 Pfizer Global R&D, St. Louis, MO, USA;
7 Pfizer, Inc, New York, NY, USA; 8 Pfizer Global R&D, New London, CT, USA
For further information, please contact: Jayvant R Heera, Pfizer Global R&D, New London, CT, 860-732-2248, jayvant.heera@pfizer.com
Increases from baseline in total cholesterol, HDL and LDL cholesterol, and triglycerides were greater for patients receiving efavirenz + CBV than for those receiving maraviroc + CBV over a 96-week period
A higher proportion of patients on efavirenz experienced lipid levels above those recommended by NCEP guidelines for intervention at both Week 48 and Week 96
These data confirm Week 48 findings that maraviroc has minimal impact on lipid profiles
Overall, maraviroc is lipid neutral and it may offer some advantages compared to efavirenz (for example, in those patients with elevated LDL cholesterol levels prior to treatment)
Whether maraviroc will be associated with decreased progression of atherosclerosis in humans that is independent of lipid effects, as seen in the mouse model,7 remains to be determined




Table 2: Lipid Profiles at Baseline Were Comparable Between Treatment Groups
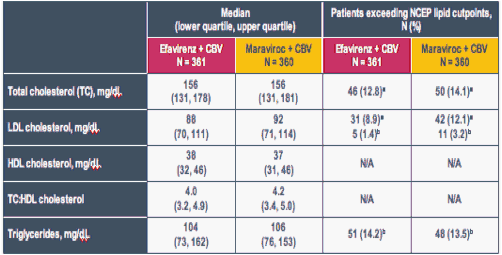
a Patients exceeding 'borderline-high' cutpoint (TC ≥ 200 mg/dL [≥ 5.2 mmol/L]; LDL ≥ 130 mg/dL [≥ 3.4 mmol/L])
b Patients exceeding 'high' cutpoint (LDL ≥ 160 mg/dL [≥ 4.1 mmol/L]; triglycerides ≥ 200 mg/dL [≥ 2.3 mmol/L])
Baseline values were missing for up to 13 patients in each treatment group
Three patients in efavirenz group and five in maraviroc group were receiving LDL-lowering therapy at baseline
To convert cholesterol to mmol/L, divide values by 38.7; to convert triglyceride values to mmol/L, divide by 88.6
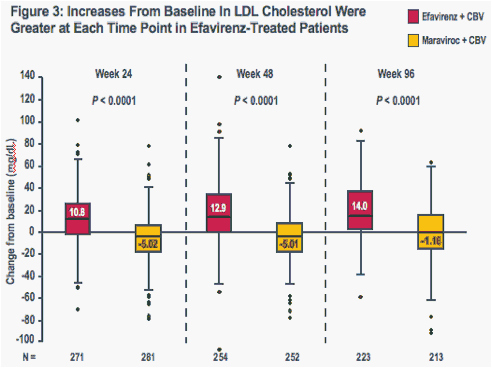
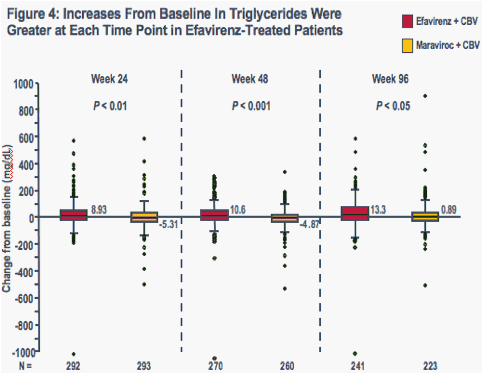
Increases in all evaluated lipid parameters were greater at each time point in the efavirenz group than in the maraviroc group (Figures 1-4)
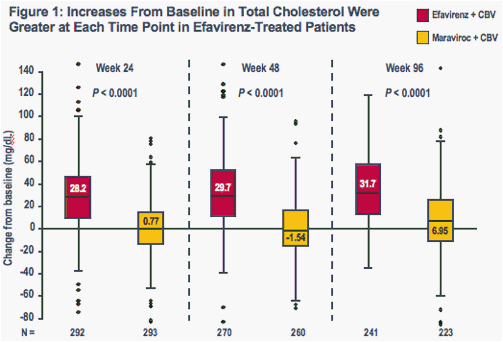
The boxes represent the interquartile range (IQR) from 25th to 75th percentile, while the horizontal black line in the box and accompanying value represents the median; the whiskers extend to the most extreme data point within 1.5x the IQR from the box
P-values are for comparisons of median values (Wilcoxon test
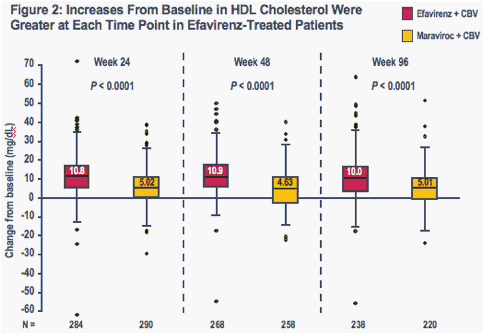
The boxes represent the interquartile range (IQR) from 25th to 75th percentile, while the horizontal black line in the box and accompanying value represents the median; the whiskers extend to the most extreme data point within 1.5x the IQR from the box
P-values are for comparisons of median values (Wilcoxon test
The boxes represent the interquartile range (IQR) from 25th to 75th percentile, while the horizontal black line in the box and accompanying value represents the median; the whiskers extend to the most extreme data point within 1.5x the IQR from the box
P-values are for comparisons of median values (Wilcoxon
The boxes represent the interquartile range (IQR) from 25th to 75th percentile, while the horizontal black line in the box and accompanying value represents the median; the whiskers extend to the most extreme data point within 1.5x the IQR from the box
P-values are for comparisons of median values (Wilcoxon
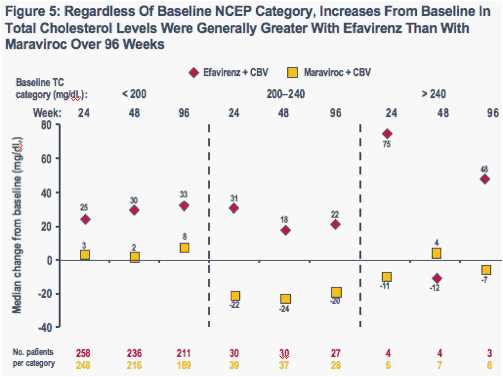
When analyzed by baseline NCEP cholesterol category, increases in total cholesterol (Figure 5) and LDL cholesterol (Figure 6) were generally greater at each time point in the efavirenz group than in the maraviroc group
-- With the exception of values at Week 48 among patients with baseline total cholesterol levels > 240 mg/dL (N = 11) and baseline LDL cholesterol levels ≥ 160 mg/dL (N = 14), increases were greater with efavirenz than maraviroc regardless of baseline NCEP cholesterol category
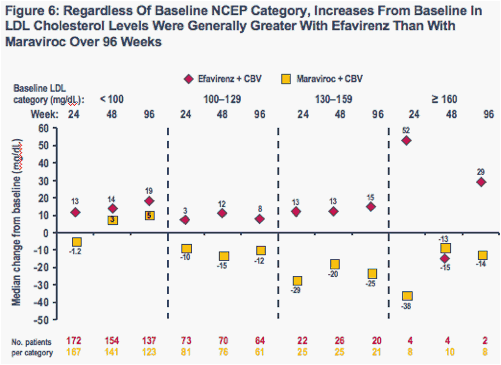
A greater percentage of patients receiving efavirenz therapy had LDL cholesterol levels ≥ 130 mg/dL at both Week 48 and Week 96, compared to those receiving maraviroc therapy (P = 0.0017) (Figure 7)
-- 5/317 (1.6%) patients receiving efavirenz versus 1/304 (0.3%) patients receiving maraviroc had LDL cholesterol levels that were ≥ 160 mg/dL at both Weeks 48 and 96
These analyses did not include patients whose lipid levels already exceeded the thresholds at baseline
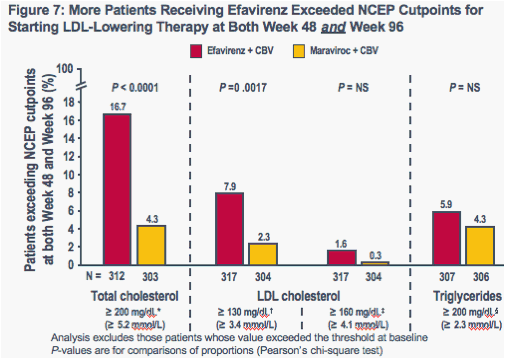
*Borderline high/high level; threshold at which the NCEP guidelines recommend considering LDL-lowering therapy in patients with ≥ 2 risk factors for CHD (e.g. hypertension, family history of premature CHD, older age, smoker) and a 10-20% 10-year risk for CHD; threshold at which the NCEP guidelines recommend considering LDL-lowering therapy in patients with ≥ 2 risk factors for CHD and a < 10% 10-year risk for CHD; § high/very high level

|
| |
|
 |
 |
|
|