 |
 |
 |
| |
Vicriviroc Long-term Safety and Efficacy:
96-Week Results from the Extension of VICTOR-E1 Study
|
| |
| |
Reported by Jules Levin
ICAAC Sept 11-15 2009
Michael C. McCarthy1, Jamal Suleiman2, Ricardo Diaz3, Jose V.R. Madruga4, Edwin DeJesus5, Barry S. Zingman6, Jihad Slim7, Natalie Case1, Lisa Dunkle1_
1Schering-Plough, Kenilworth, NJ; 2Brazilmed Assistencia Medica e Pesquisa, Sao Paolo, Brazil; 3Federal University of Sao Paolo, Brazil; 4Centro de Referencia e Treinamento em DST/AIDS, Sao Paolo, Brazil; 5Orlando Immunology Center, Orlando, FL; 6Montefiore Medical Center and Einstein-Montefiore Center for AIDS Research, Bronx, NY; 7St. Michael's Medical Center, Newark, NJ
AUTHOR CONCLUSIONS
VCV was generally well tolerated in this highly treatment-experienced population, with no apparent VCV-related toxicities.
Mild upper respiratory infections were the most commonly observed event of interest, consistent with findings in the double-blind phase (VICTOR-E1 study).
Sustained antiviral effect, including full virologic suppression, and durable CD4+ cell count increases were observed.
Resistance to VCV was infrequent and developed slowly, generally in subjects after prolonged therapy with suboptimal OBT, ie, fewer than 2 active drugs.
ABSTRACT
Background: This extension of VICTOR-E1 evaluated the continued safety and efficacy of vicriviroc (VCV) in treatment-experienced subjects.
Methods: After completing 48 weeks of blinded treatment with VCV 30 mg, VCV 20 mg, or placebo in a protease inhibitor/ritonavir (PI/r)-containing regimen, subjects were offered open-label VCV (30 mg in a PI/rcontaining regimen) for 48 more weeks. Adverse events (AEs) were assessed every 3 months. HIV RNA and CD4 counts were monitored.
Results: 85 of the 86 subjects who completed VICTOR-E1 enrolled in the extension. Mean duration of open-label therapy was 13 months (total 26 months). The only AEs in 35% of subjects were sinusitis, cough, and insomnia. One subject was discontinued upon simultaneous diagnosis of Hodgkin's lymphoma and Kaposi's sarcoma, which were not treatment-related. Another patient discontinued due to pregnancy. There were no treatment-emergent deaths, seizures, or hepatotoxic events.

Conclusions: VCV showed promising safety and tolerability with no clear VCV-related toxicity. Sustained antiviral effect and maintenance of improved CD4 counts were observed.
INTRODUCTION
Vicriviroc (VCV) is a next-generation extracellular inhibitor of HIV infection designed to block entry of infectious virions into uninfected CD4+ cells via antagonism of the CCR5 co-receptor (CCR5 antagonist).1
VCV plasma half-life of >24 hours allows for once-daily dosing2 and may be taken with or without food.3
VCV has demonstrated potent and durable antiretroviral activity in CCR5-tropic antiretroviral-experienced patients.4
In a randomized, placebo-controlled Phase 2b study (VICTOR-E1), vicriviroc 30 or 20 mg once daily plus ritonavir-boosted protease inhibitor (PI/r)-containing optimized background therapy (OBT) given to treatment-experienced HIV-infected patients with CCR5-tropic only virus demonstrated sustained superior virologic and immunologic efficacy compared with OBT alone.5
Vicriviroc (30 mg, QD) as part of a ritonavir-containing, PI-based regimen is now in Phase 3 clinical trials evaluating HIV-monoinfected and HIV/HCV-coinfected treatment-experienced patients; all patients have completed at least 48 weeks of treatment.
METHODS
This ongoing, open-label extension of VICTOR-E1 trial was designed to provide continued therapy with VCV and to collect safety data. Evidence for sustained viral suppression and CD4+ cell count improvement was monitored.5
After completing 48 weeks of blinded treatment in VICTOR-E1, subjects were offered open-label VCV 30 mg QD plus PI/r-containing OBT.
- Subjects who discontinued before Week 48 of the double-blind phase for reasons other than AEs were offered re-screening.
- OBT could be re-optimized at the discretion of site investigator.
Subjects returned every 3 ± 1 months for evaluation, including:
- Physical examination, vital signs, and clinical laboratory testing
- HIV-1 RNA level and CD4+ cell count measurements
- Treatment-emergent adverse events (AEs)
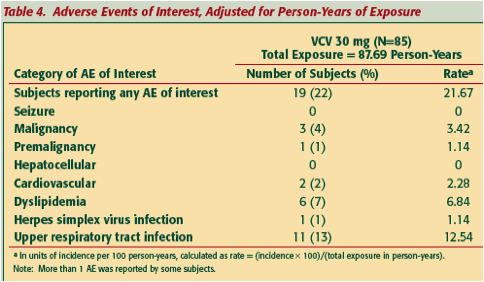
· Seizure, malignancy, premalignancy, hepatocellular AEs, dyslipidemia, cardiovascular AEs, herpes simplex virus infection, and upper respiratory tract infection were designated as AEs of interest.
Tropism testing (using the first-generation Trofile assay) and VCV susceptibility testing (using the PhenoSense Entry assay) were performed in all subjects at baseline (Week 48 of double-blind phase) except those whose HIV RNA was <500 copies/mL.
- Thereafter, tropism testing was performed every 6 months and at final visit.
- VCV susceptibility testing was to be performed at final visit.
- VCV resistance was assessed using maximal percent inhibition plateau value of <85% as cutoff for resistance.
Overall sensitivity scores (OSS), by combining genotypic and phenotypic resistance scores, for the OBT were calculated for subjects who experienced virologic failure during the open-label phase.
RESULTS
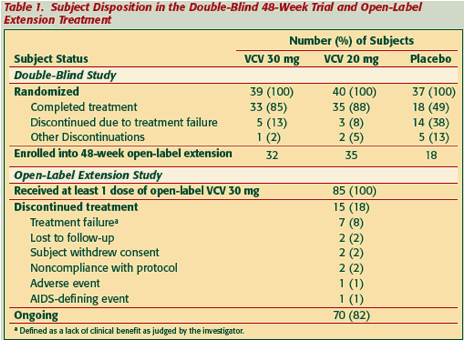
Subject Disposition and Baseline Characteristics
85 subjects were enrolled in the open-label treatment phase from the 48-week
double-blind treatment arms (Tables 1 and 2):
- 32 from VCV 30 mg QD arm
- 35 from VCV 20 mg QD arm
- 18 from placebo arm
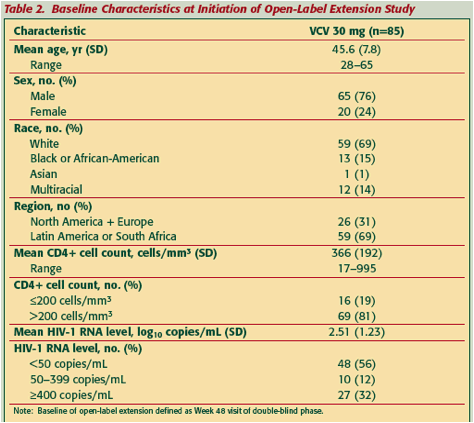
More than half of subjects began open-label treatment with an HIV-1 RNA level <50 copies/mL and two thirds <400 copies/mL (Table 2).
VCV Exposure
All subjects received 31 dose of open-label VCV 30 mg.
· Mean duration of open-label VCV treatment was 13 months.
· Total exposure was 88 person-years.
Virologic Efficacy
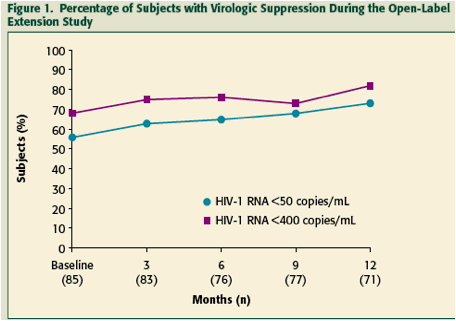
HIV-1 RNA levels were sustained during open-label treatment, with a mean log10 change from baseline of -0.32.
Increases in the proportion of subjects achieving undetectable viral loads were observed (Figure 1).
Immunologic Efficacy
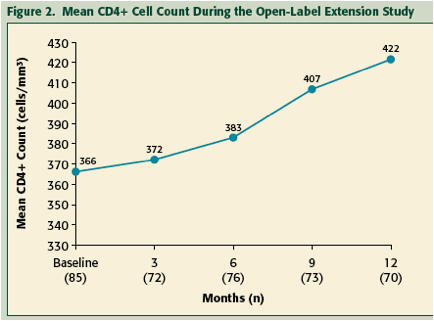
Further improvements in CD4+ cell counts were observed with prolonged VCV treatment, with a mean increase of 50 cells/mm3 from baseline by Month 12 (Figure 2).
Adverse Events
- 58 subjects reported one or more adverse event (AE) (Table 3).
- Most (93%) AEs were mild or moderate in severity.
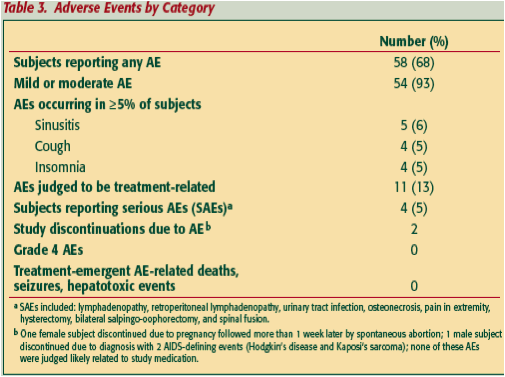
Adverse Events of Interest
Nineteen (22%) of subjects experienced an AE of interest on study (Table 4).
Laboratory Abnormalities
No Grade 3 or 4 changes from baseline occurred for any laboratory parameter measured, including liver function tests.
2 cases of new-onset diabetes mellitus occurred-considered possibly related to study drug.
- Mean change in fasting glucose level was not clinically significant over the entire study population.
- The OBTs of the two subjects experiencing new-onset diabetes both contained the PIs SQV and LPV/RTV-antiretrovirals associated with the development of insulin resistance.
- Glucose abnormalities have been reported in patients with extensive antiretroviral
exposure.
Mild elevations in serum lipids were observed in a small number of subjects.
- Many antiretrovirals are associated with lipid changes.
Viral Tropism
Of those subjects in whom viral tropism testing was possible (ie, having HIV-1 RNA >500 copies/mL)
- Immunologic decline requiring study discontinuation was not observed in 11 of the 14
subjects with detectable DM/X4 virus.
- Only 1 subject with documented R5-tropic virus at entry into the open-label extension
was later found to have DM/X4 virus; the subject was discontinued for treatment failure.
Interpretation of HIV tropism in this study is complex because the majority of subjects had achieved virologic suppression prior to entry into open-label treatment.
Antiretroviral Resistance
Treatment failure occurred in 11 (13%) subjects.
-- 7 of the 11 were protocol-defined virologic failures.
- 4 of the 11 were investigator-determined treatment failures.
- 8 of the 11 had one or fewer active drugs in their OBT.
- Treatment failure was the most common reason for discontinuation from the open-label extension (11%).
2 of the subjects with treatment failure already had documented resistance to VCV during the double-blind phase.
- Both subjects had OSS = 0 at entry to open-label phase, and neither subject achieved virologic suppression during either the blinded or open-label phases.
2 other subjects developed changes in VCV susceptibility during open-label phase consistent with resistance.
REFERENCES
1. Strizki JM, Tremblay C, Xu S, et al. Antimicrob Agents Chemother. 2005;49:4911-4919.
2. Seiberling M, Kraan M, Keung A, et al. 6th International Workshop on Clinical
Pharmacology of HIV Therapy, Quebec City, Que., Canada, Apr. 28-30, 2005.
Amsterdam: Virology Education, 2005. Abstract 6.4.
3. Sansone A, Keung A, Caceres M, et al. 45th ICAAC; December 16-19, 2005;
Washington, DC. Abstract A-1200.
4. Gulick RM, Su Z, Flexner C, et al. J Infect Dis. 2007;196:304-312.
5. Zingman B, Suleiman J, DeJesus E, et al. 15th CROI; February 3-6, 2008; Boston, MA. Abstract 39LB.
|
| |
|
 |
 |
|
|