 |
 |
 |
| |
24 Week Safety and Efficacy from IMPAACT P1066: A Phase I/II, Multicenter, Open-Label, Noncomparative Study to Evaluate Raltegravir (RAL) in HIV-1 Infected Youth
|
| |
| |
Reported by Jules Levin
ICAAC Sept 11-15 2009
San Francisco
S. Nachman1, P. Samson2, L. Frenkel3*, E. Acosta4, H. Teppler5, T. Fenton2, E. Handelsman6, C. Worrell7, B. Graham8, A. Wiznia9
1SUNY Stony Brook, NY, 2Harvard School Public Health, MA, 3U. Washington, WA, 4U Alabama, AL, 5Merck & Co. Inc., PA, 6NIAID, MD, 7NICHD, MD, 8Frontier Science Research Foundation Inc., NY, 9Jacobi Med Center, NY

ABSTRACT
Background: We report 24-week (wk) antiretroviral activity of RAL and all available safety data in 12-18 year-old participants.
Methods: HIV+ youth enrolled in either Stage 1 (dose finding: intensive PK and safety) or Stage 2 (extended follow-up) cohorts. Entry criteria included HIV RNA>1000 copies/mL, naïve to integrase inhibitors, and excluded individuals with HBV or HCV. Other ARV were optimized at entry or within 10 days of entry. Based on Cohort I, Stage 1 data, a dose of 400mg BID was chosen. Stage 1
participants not receiving 400mg BID were switched to this dose. Success was defined as achieving and maintaining >1-log drops from baseline or viral loads of ≤400 copies/ml and on-study treatment (RAL, 400mg BID) at wk 24. Any participant taken off-treatment prior to wk 24 was considered a "failure". All participants were included in the safety analyses.
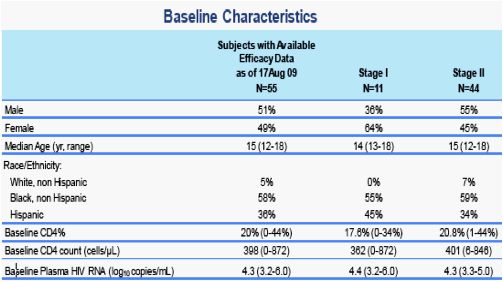
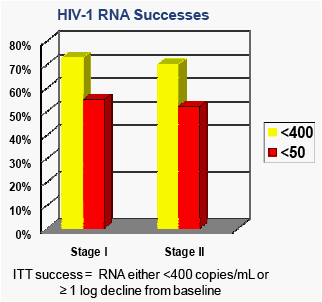
Results: 69 participants were accrued: Stage 1: n=22; Stage 2: n=47. Baseline demographics: Male: 53%; Race: 63% Black/African American, 33% White, 4% Others. Median: weight=56kgs; age=15yrs; log10RNA=4.3 CD4=397 (20%). Efficacy (Intent to Treat) data is available on 43 participants. At 24 wks, 74% achieved success, 95%CI (59, 86%); 56% achieved ≤50 copies/mL, 95%CI (40, 71%). Grade 3+ toxicities were seen in 14 of 69 participants; 4/69 were judged to be possibly related to RAL (2 Grade 3+ ANC, 1 elevated GGT, 1 Grade 3+ behavioral).
Conclusions: In HIV infected youth ages 12 -18, RAL appears generally safe and well tolerated through wk 24, and achieved efficacy rates comparable to those in treatment experienced adults.
BACKGROUND
· RAL is a potent inhibitor of HIV integrase and blocks viral replication
· Data in adults show an excellent safety profile as well as potent efficacy in experienced adults who add RAL to a new optimized regimen and when used as part of primary regimen in therapy naïve adults [1,2,3]
· Prior data from P1066 established a dose of 400 mg taken twice a day as the dose for further study in HIV+ youth aged 12-18 years of age [4,5]
· This report provides 24 week efficacy of RAL given with an optimized background regimen (optimized at or within 10 days of entry) in HIV+ therapy experienced youth, as well as safety data
METHODS
· These analyses include subjects who received RAL tablets at 400mg BID starting at study entry up to week 24, and have available data (as of 17 Aug 2009)
· The primary analysis was an Intent-to-Treat (ITT) analysis where "SUCCESS" is defined as having achieved and maintained at least a 1-log decline in viral RNA from baseline OR <400 copies/mL and continued on-study treatment (raltegravir 400mg BID) through week 24
· Any subject taken off-treatment (discontinued) prior to week 24 or who had a RAL dose adjustment not due to the reason of weight change is considered a "FAILURE"
· A secondary, more stringent definition of virologic success, required that participants achieve and maintain a viral RNA of <50 copies/mL
· Safety data includes all available data as of 17 Aug 2009
RESULTS


REFERENCES:
1. Steigbigel RT, et al. Raltegravir with optimized background therapy for resistant HIV-1 infection. N Engl J Med 2008;359:339-354.
2. Cooper, DA, et al. Subgroup and resistance Analyses of raltegravir for resistant HIV-1 infection. N Engl J Med 2008:359: 355-365.
3. Markowitz M. et al. Sustained antiretroviral efficacy of raltegravir as part of combination ART in treatmentnaive HIV-1 infected patients: 96 weeks. XVII International AIDS Conference; 2008 Aug 3-8. Mexico City, Mexico, 2008.
4. Nachman, S., et al. Raltegravir (RAL) pharmacokinetics and safety in adolescents: preliminary results from IMPAACT P1066 [ Poster]. ICAAC/IDSA Annual Joint Meeting 2008; 2008 Oct 25-28, Washington, D.C., 2008.
5. Wiznia, A., et al. Safety and Efficacy of Raltegravir (RAL) in Pediatric HIV Infection. Preliminary Analysis from IMPAACT P1066 [ Poster]. CROI 2009; 2009 Feb 8-11, Montreal, Canada, 2009.
|
| |
|
 |
 |
|
|