 |
 |
 |
| |
Maraviroc (MVC) is Similar to Efavirenz (EFV) for Virologic Response (VR) Across Subgroups: MERIT 96-Week
|
| |
| |
Reported by Jules Levin
ICAAC Sept 15 2009 San Francisco
WD Hardy1, C Cohen2, P Ive3, AIM Hoepelman4, E van der Ryst5, J Heera6, H Fan6, J Goodrich6, M Westby5, H Mayer6
1Cedars-Sinai Medical Center, Geffen School of Medicine, UCLA, Los Angeles, CA, USA; 2Community Research Initiative New England, Boston, MA, USA; 3University of Witwatersrand, Johannesburg, South Africa; 4University Medical Center Utrecht, Utrecht, Netherlands; 5Pfizer Global R & D, Sandwich, UK; 6Pfizer Global R & D, New London, CT, USA
For further information, please contact Jayvant R Heera, Pfizer Global R&D, New London, CT, 860-732-2248, jayvant.heera@pfizer.com
AUTHOR DISCUSSION
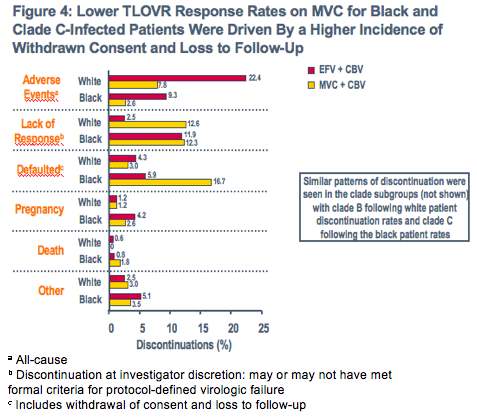
The lower occurrence of AE-related discontinuations among the highly overlapping groups of Black, African patients and clade C infections may be related to socioeconomic factors - including a lack of alternative treatment options - promoting a greater initial tolerance of adverse events
This lower rate of AE discontinuations on EFV may in turn be linked to the higher rate of subsequent EFV efficacy discontinuations in these groups relative to White and clade B-infected patients
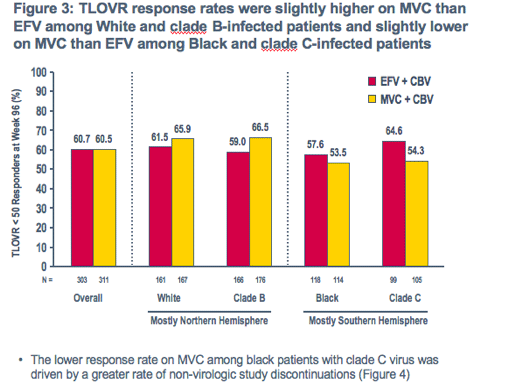
- Since MVC was better tolerated than EFV in all groups, efficacy-related discontinuations on MVC were not significantly impacted by differences in AE tolerance and remained similar across subgroups
The reasons for the higher rates of patient default among Black and clade C patients receiving MVC are unclear
Given the strong co-localization of these groups to a single country this may represent complex local influences of limited extrapolation to other settings
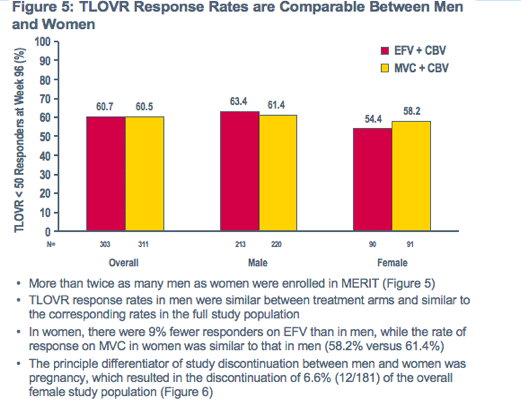
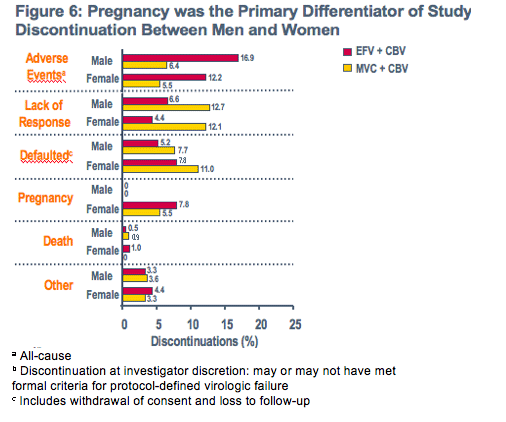
Slightly lower TLOVR responses in both treatment arms among women compared to men appears driven primarily by pregnancy-related discontinuations
The greater response difference between genders for patients receiving EFV may be an artifact of the smaller female sample set
AUTHOR CONCLUSIONS
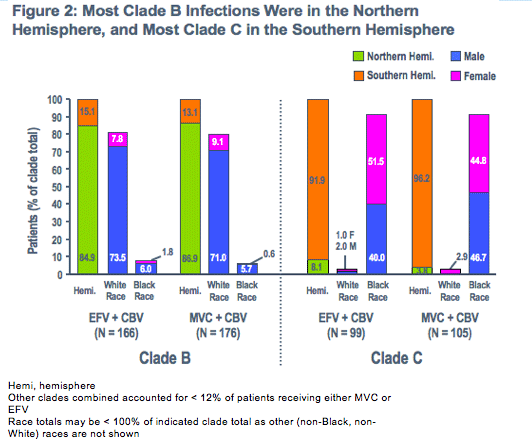
The three subgroups studied - race, gender and viral clade - proved to be highly interrelated
Three of the major strata (Black, female and clade C) strongly co-localized to a single country (South Africa), and the White, male and clade B strata also clustered together
MVC and EFV showed broadly similar TLOVR response rates across subgroups at week 96 in MERIT-ES, with such differences as were noted being attributable to non-virologic discontinuations with no apparent link to subgroup differences in the efficacy or tolerability of the drugs received
INTRODUCTION & ANALYSIS
The MERIT study evaluated maraviroc (MVC) 300 mg BID versus efavirenz (EFV) 600 mg QD, each with Combivir (CBV; zidovudine/lamivudine), in treatment-naïve subjects infected with CCR5-tropic HIV-1 (Figure 1)
- The primary analysis of MERIT occurred at Week 481
Viral tropism was determined at screening visit by the original Trofile assay (Monogram Biosciences), which is no longer clinically available for patient management
- A post-hoc reanalysis (MERIT-ES) was conducted at 48 and 96 weeks, which omitted patients who re-screened with non-R5 virus using the newer, enhanced sensitivity Trofile assay
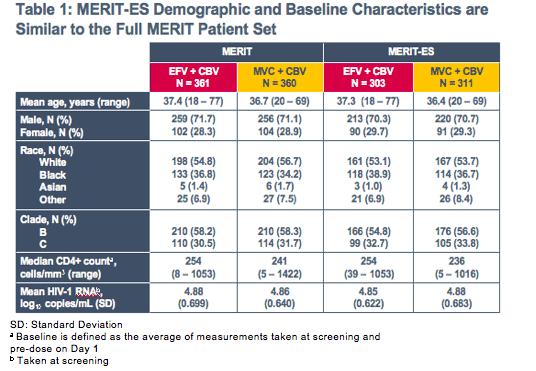
- Patient demographics and baseline characteristics in the MERIT-ES dataset are similar to the full MERIT dataset (Table 1)
In the Week 96 MERIT-ES analysis the proportion of responders by the Time to Loss of Virologic Response (TLOVR) algorithm (< 50 copies/mL) was similar between the EFV (60.7%) and the MVC (60.5%) treatment arms3
- The lower bound of the one-sided 97.5% confidence interval (CI) for the treatment difference using the TLOVR algorithm (-7.9%) was above -10%
- In addition, the significant difference in CD4+ cell count increases favoring MVC previously noted at week 481,2 was maintained through week 963
The current post-hoc analysis sought to evaluate week 96 virologic responses between MERIT-ES demographic subgroups and investigate the cause of any differences noted
METHODS
Patients in the MERIT-ES dataset (N = 614) were stratified by race, gender and viral clade; TLOVR response rates for suppression < 50 HIV-1 RNA copies/mL at week 96 were evaluated by the above three baseline variables using descriptive statistics
Discontinuation rates and causes were assessed by treatment for each subgroup stratum





|
| |
|
 |
 |
|
|