 |
 |
 |
| |
Changes In HBeAg Levels Among Patients With HBeAg-Positive Chronic Hepatitis B Treated With Peginterferon Alfa-2a [40KD] Enrolled In The S-Collate Cohort Study
|
| |
| |
Reported by Jules Levin
AASLD Nov 2 2010 Boston
Lou GQ,1 Marcellin P,2 Xie Y,3 Chen XP,4 Chen YP,5 Rothe V,6 Regep L,7 Martins EB,8 Jia J,9 Wei L10
1The Sixth People's Hospital of Hangzhou, Hangzhou, China; 2Service d'Hepatologie and Centre de Recherches Biologiques Beaujon (Inserm CRB3), University of Paris, Clichy, France; 3Beijing DiTan Hospital, Beijing, China;
4Guangdong General Hospital, Guangdong Academy of Medical Science, Guangzhou, China; 5The First Affiliated Hospital of Wenzhou Medical College, Wenzhou, China; 6IST GmbH, Mannheim, Germany; 7F.Hoffmann-La Roche Ltd, Basel, Switzerland;
8Genentech - A Member of the Roche Group, South San Francisco, California, USA; 9Beijing Friendship Hospital, Capital Medical University, Beijing, China; 10Peking University People's Hospital, Beijing, China
Presented at the 61st Annual Meeting of the American Association for the Study of Liver Disease (AASLD), 29 October - 2 November 2010, Boston, USA

Background
Approximately one-third of patients with hepatitis B e antigen (HBeAg)-positive chronic hepatitis B (CHB) will achieve HBeAg seroconversion (sustained immune control) 6 months after completing pegylated interferon therapy.1,2 Importantly, as sustained immune control is associated with increasing rates of hepatitis B surface antigen (HBsAg) clearance3 - the ultimate goal of therapy in CHB - durable HBeAg seroconversion has been demonstrated with pegylated interferon in HBeAg-positive patients even up to 5 years post-treatment4
Recently, there has been considerable interest in predicting, during pegylated interferon therapy, those patients likely to achieve sustained immune control post-treatment. The main focus has been on measurement of HBsAg, but several publications have also demonstrated that quantitative HBeAg (qHBeAg) levels either before or during treatment with pegylated interferon may be associated with post-treatment response.5-7 Indeed, Fried and colleagues showed that HBeAg quantification was a more appropriate marker than HBV DNA in monitoring the response to pegylated interferon alfa-2a treatment5
However, Fried concluded that clinical use of quantitative HBeAg to predict response is likely to be limited as a commercial assay for quantification of HBeAg is not currently available.5 Data on a range of parameters, including quantitative HBeAg, will be collected during the S-collate cohort study and, therefore, this non interventional study provides an opportunity to determine whether HBeAg is quantified routinely in the complex and varied setting of routine clinical practice

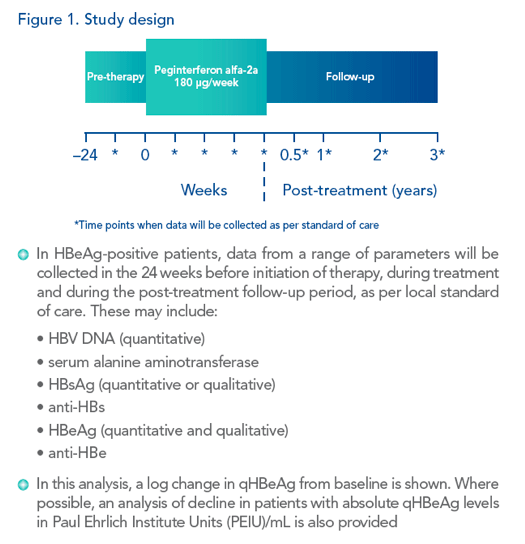
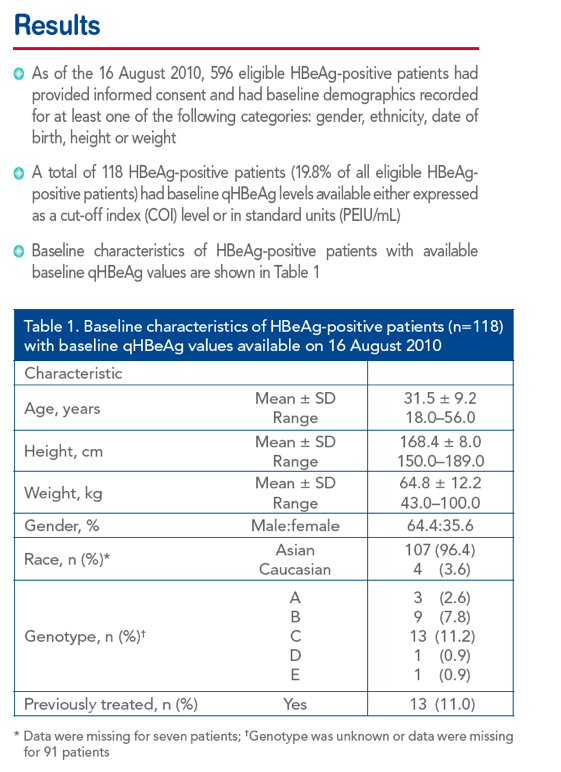

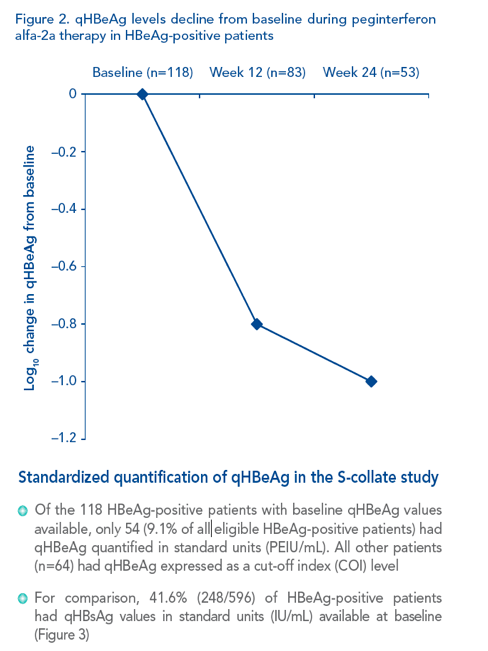
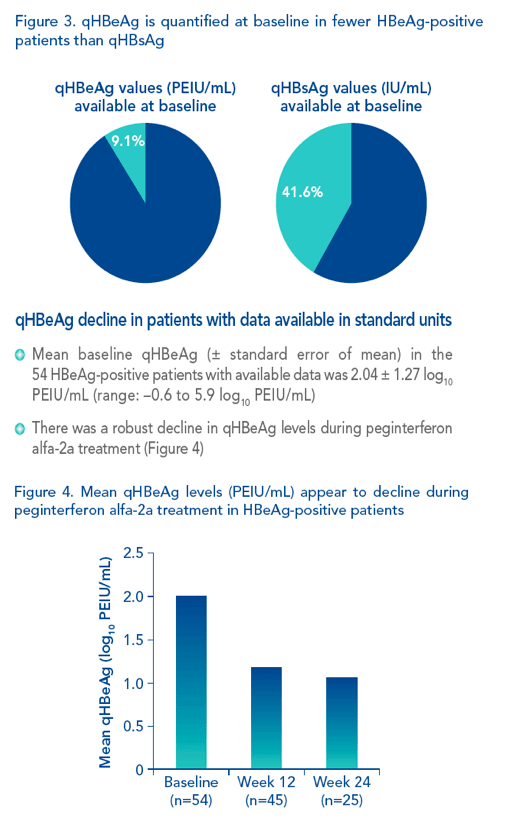
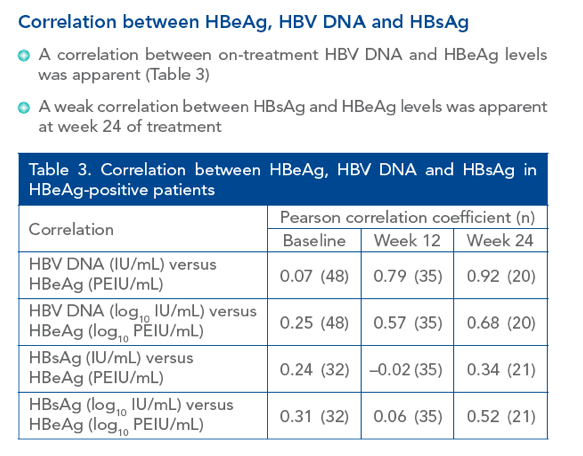

|
| |
|
 |
 |
|
|