 |
 |
 |
| |
New HIV Rate High After iPrEx Men Stop Truvada PrEP (or Placebo): when taking Truvada PrEP it works
|
| |
| |
20th Conference on Retroviruses and Opportunistic Infections, March 3-6, 2013, Atlanta
Mark Mascolini
HIV incidence among gay/bisexual men taking tenofovir/emtricitabine (Truvada) preexposure prophylaxis (PrEP) during the iPrEx trial rebounded to high levels after they stopped daily dosing [1]. Post-trial incidence remained at the same high level in men randomized to placebo during the study.
But incidence "tended to be lower among the group formerly exposed to active PrEP," the iPrEx investigators determined, "indicating that infections averted during PrEP use were not offset by excess infections after PrEP use ended."
iPrEx randomized sexually active men and transgenders who have sex with men to daily Truvada or placebo and found an overall 43% reduction in adjusted HIV incidence with Truvada [2]. Protection from HIV reached 92% in participants with detectable study drug in plasma or blood cells.
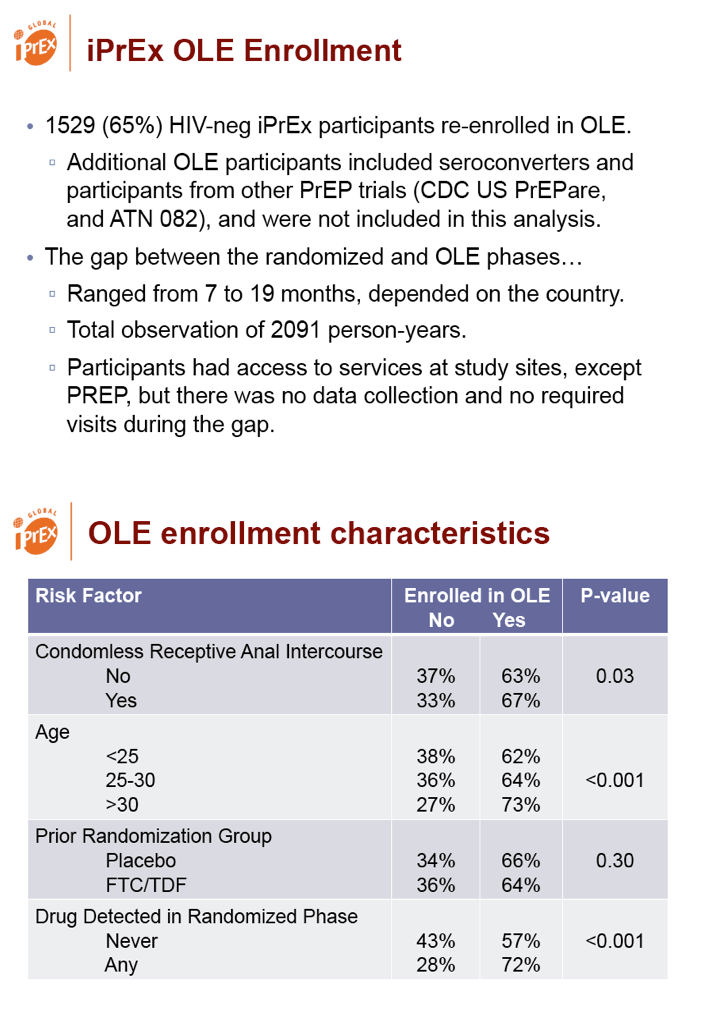
To see if HIV incidence changed after the randomized phase of iPrEx stopped, researchers monitored 1529 HIV negative participants for 7 to 19 months, before they started the iPrEx off-label extension (OLE) study (http://www.iprexole.com/). During that time participants had access to services at study sites but got no Truvada for PrEP. Taking active drug during iPrEx did not affect willingness to enroll in iPrEx OLE.
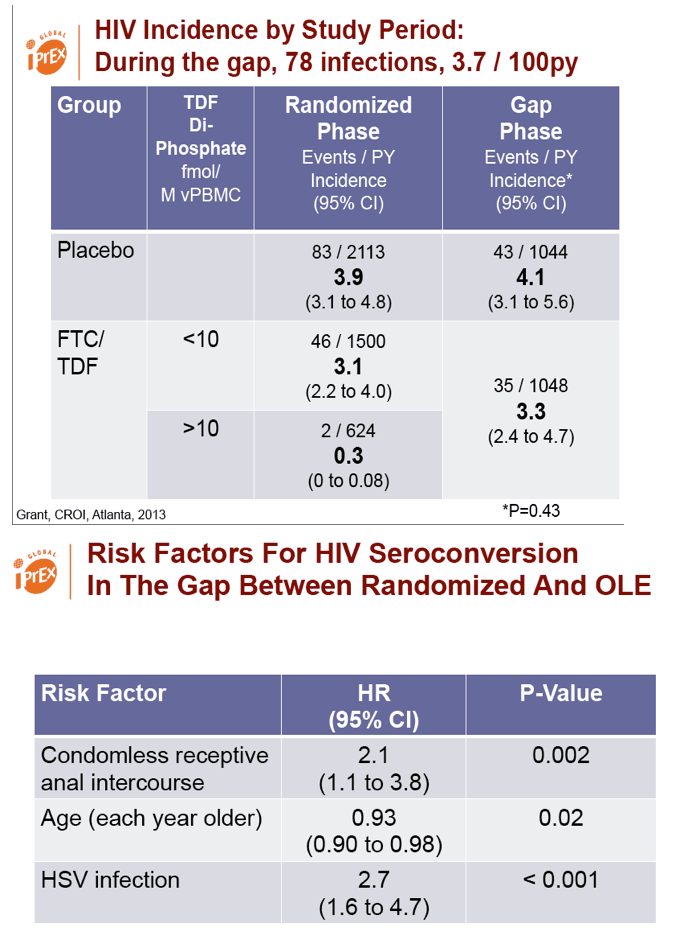
During 2091 person-years of follow-up in the no-PrEP gap between iPrEx and iPrEx OLE, there were 78 new HIV infections--43 in the placebo group and 35 in the Truvada group. Those numbers translated into incidence rates of 4.1 per 100 person-years (95% confidence interval [CI] 3.1 to 5.6) in the placebo group and 3.3 (95% CI 2.4 to 4.7) in the Truvada group, and that difference did not approach statistical significance (P = 0.43).
HIV incidence in both groups did not differ markedly from incidence in the placebo group during the randomized phase (3.9 per 100 person-years). Incidence among Truvada takers during the trial stood at 3.1 per 100 person-years in those with a tenofovir diphosphate level below 10 fmol/M blood cells and at 0.3 per 100 person-years in those with a tenofovir diphosphate level about that mark.
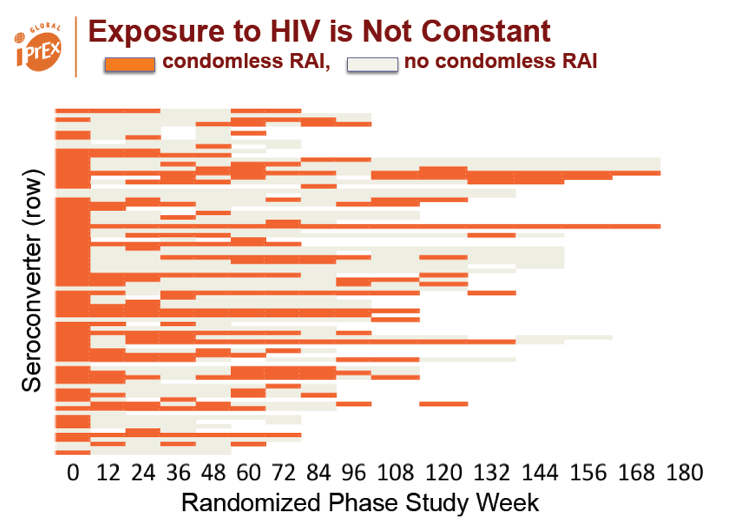
Each year of age lowered the risk of HIV infection 7% (hazard ratio [HR] 0.93, 95% CI 0.90 to 0.98, P = 0.02). Receptive anal intercourse without a condom doubled the risk (HR 2.1, 95% CI 1.1 to 3.8, P = 0.002), and HSV infection nearly tripled the risk (HR 2.7, 95% CI 1.6 to 4.7, P < 0.001).
The iPrEx investigators concluded that "overall HIV incidence continues to be high in the iPrEx cohort when PrEP is not available," but there is no "excess incidence" in the Truvada group after Truvada PrEP stops. The researchers will compare HIV incidence during iPrEx OLE with incidence seen in placebo users during the randomized phase and in all participants during the gap phase.
References
1. Grant R, Guanira J, Buchbinder S, et al. No excess in HIV incidence after stopping oral emtricitabine/tenofovir pre-exposure prophylaxis: the iPrEx trial. 20th Conference on Retroviruses and Opportunistic Infections. March 3-6, 2013. Atlanta. Abstract 27.
2. Grant RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587-2599. http://www.nejm.org/doi/full/10.1056/NEJMoa1011205



|
| |
|
 |
 |
|
|