 |
 |
 |
| |
Dual therapy with lopinavir/ritonavir (LPV/r) and lamivudine (3TC) is non-inferior to standard triple drug therapy in naive HIV-1 infected subjects: 48-week results of the GARDEL study
|
| |
| |
Reported by Jules Levin
EACS - 14th European AIDSConference Oct 16-19 2013 Brussels, Belgium
Pedro Cahn on behalf of the GARDEL study group

EXTENSION
305 entered the extension Phase of the study,
66 declined
Most frequent reasons :
Most frequent reasons :
· Study not approved in Peru (22 )
· Virological failure at week 48 ( 14 ) ( failures at week 48 are 15 but one patient by mistake entered to extension phase and was approved to continue due to <50 copies at next visit )
· Patient decision ( 7 )
· Medical decision ( 6 )


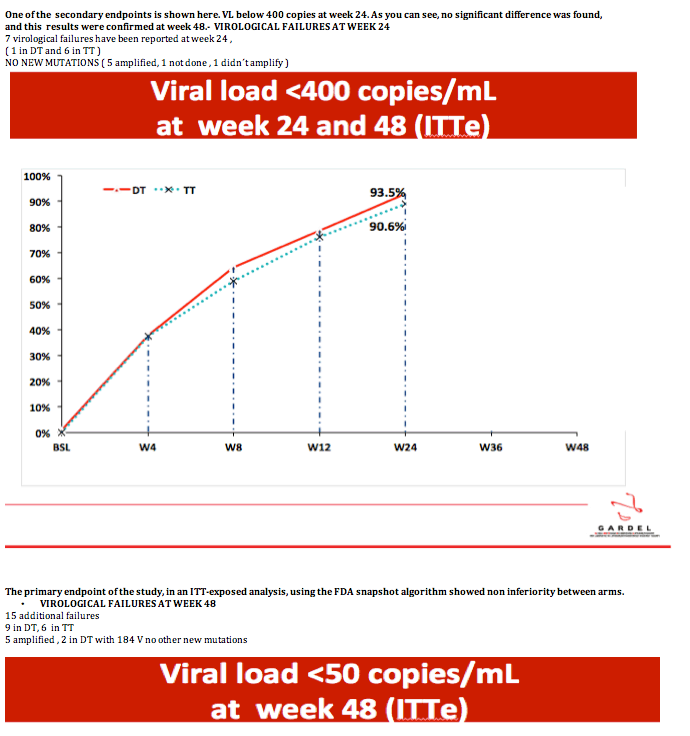
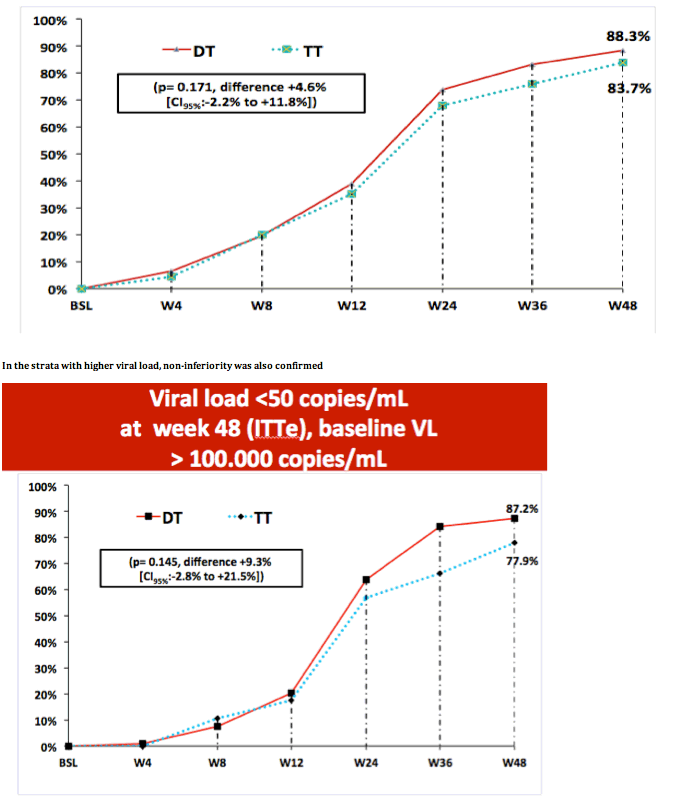
One of the secondary endpoints is shown here. VL below 400 copies at week 24. As you can see, no significant difference was found, and this results were confirmed at week 48.-
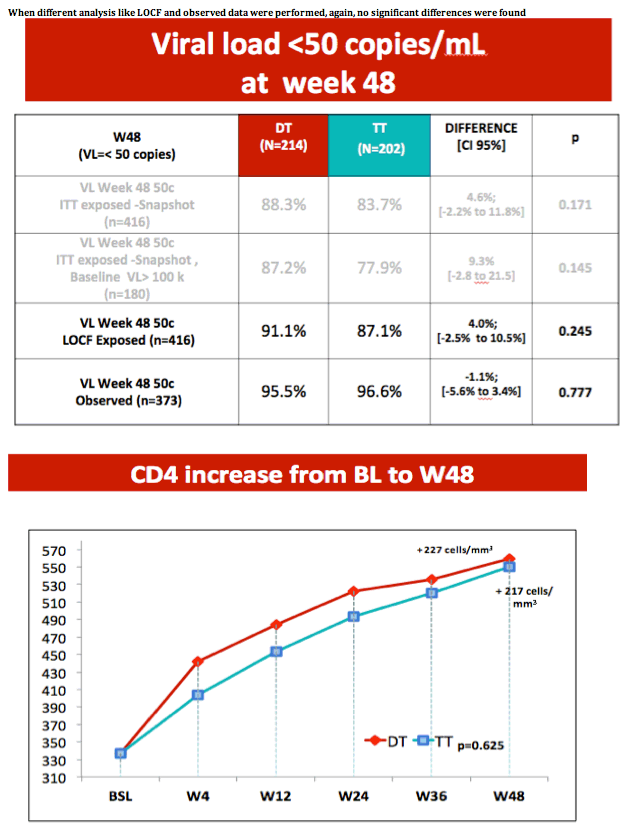
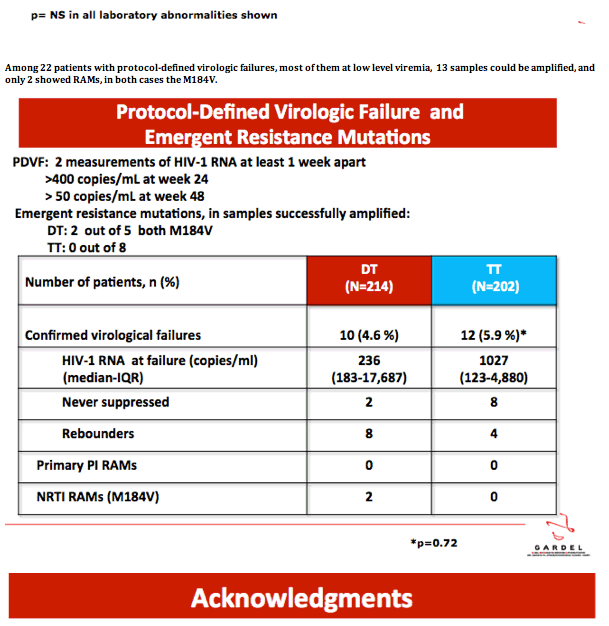
· VIROLOGICAL FAILURES AT WEEK 24
7 virological failures have been reported at week 24 ,
( 1 in DT and 6 in TT )
NO NEW MUTATIONS ( 5 amplified, 1 not done , 1 didn´t amplify )

|
| |
|
 |
 |
|
|