 |
 |
 |
| |
PI Use, Female Gender, HCV, Detectable Sub-50 Viral load Predict Rebound From Under 50 Copies in Italian Group
|
| |
| |
14th European AIDS Conference. October 16-19, 2013. Brussels
Mark Mascolini
Three quarters of a 1065-person Italian group maintained a viral load with an undetectable polymerase chain reaction (PCR) signal for 2 years of follow-up [1]. Five factors predicted rebound above 50 copies: female gender, HCV coinfection, viral suppression for fewer than 2 years, protease inhibitor (PI) use, and a detectable sub-50 load at baseline.
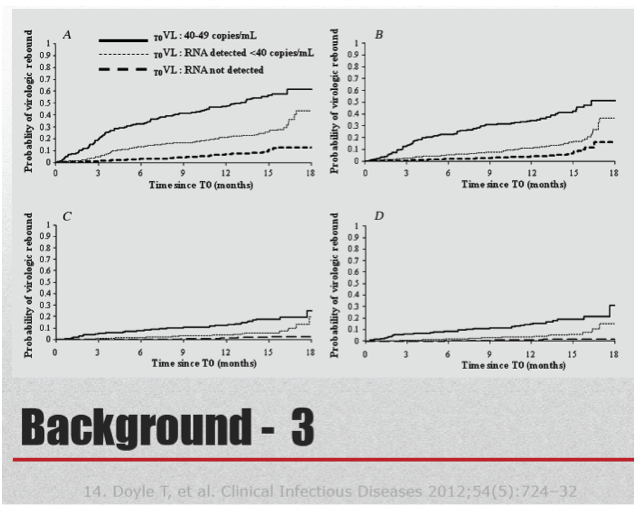
Current viral load assays can detect HIV RNA levels down to 50, 40, or 20 copies. Low-level viremia correlates with HIV DNA load in peripheral blood mononuclear cells [2] and may be associated with immune activation and microbial translocation. In a 1957-person cohort in France, people with no PCR signal on a viral load test had a 42% lower risk of viral blips and a 56% lower risk of virologic failure [3].
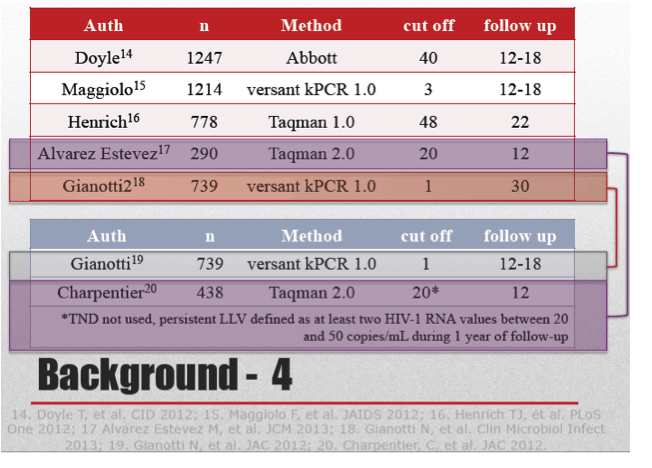
University of Torino investigators conducted this retrospective study to assess factors associated with a no-signal or "target not detected" (TND) viral load and to identify variables associated with rebound from an undetectable viral load. They defined rebound as consecutive viral loads above 50 copies or a last viral load above 50 copies in people who stopped returning for clinic visits.
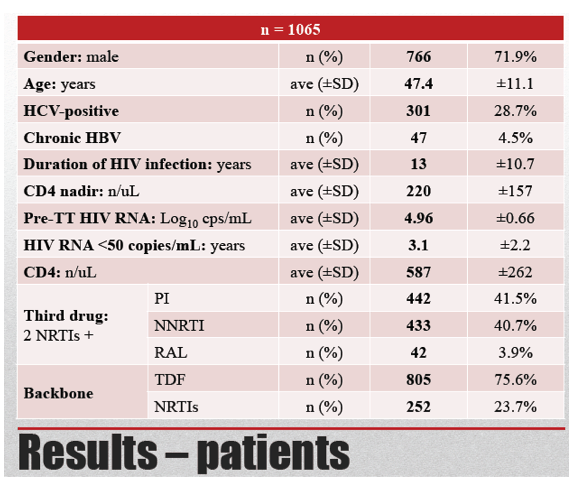
This retrospective analysis involved 1065 adults with two consecutive sub-50-copy viral loads seen from early 2010 through early 2012. Researchers divided them into three groups--those with a viral load between 20 and 50 copies, those with fewer than 20 copies, and those with TND. Age averaged 47.4 years, 766 people (72%) were men, and 301 (28%) tested positive for HCV. The group averaged 13 years in HIV infection duration, and pretreatment viral load stood around 91,000 copies. Nadir and current CD4 count averaged 220 and 587.
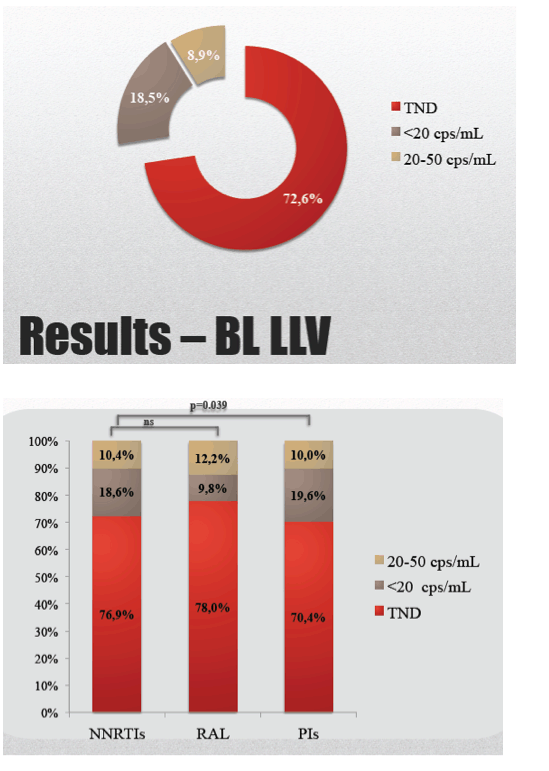
While 41.5% of the study group were taking a PI regimen, 40.7% were taking a nonnucleoside, and 3.9% were taking raltegravir. Almost three quarters of study participants (72.6%) had a TND viral load result, while 18.5% had fewer than 20 copies and 8.9% had 20 to 50 copies. By treatment group, 70.4% taking a PI, 76.9% taking nonnucleoside, and 78.0% taking raltegravir were TND. The difference between the PI group and the nonnucleoside group was statistically significant (P = 0.039).
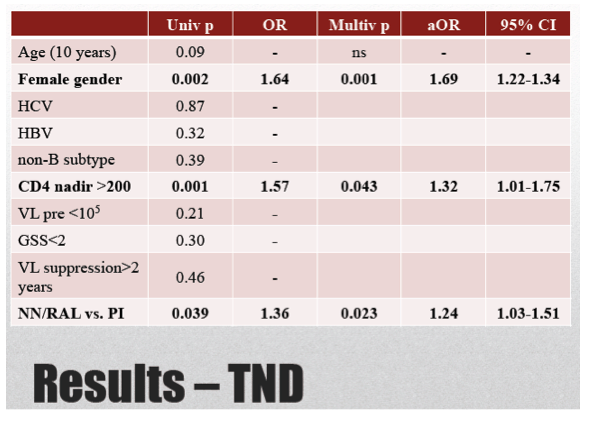
A Cox proportional hazards model identified three independent predictors of TND at the following adjusted odds ratios (aOR) and 95% confidence intervals (CI):
-- Female gender: aOR 1.69, 95% CI 1.22 to 1.32, P = 0.001
-- CD4 nadir above 200: aOR 1.32, 95% CI 1.01 to 1.75, P = 0.043
-- Nonnucleoside or raltegravir vs PI: aOR 1.24, 95% CI 1.03 to 1.51, P = 0.023
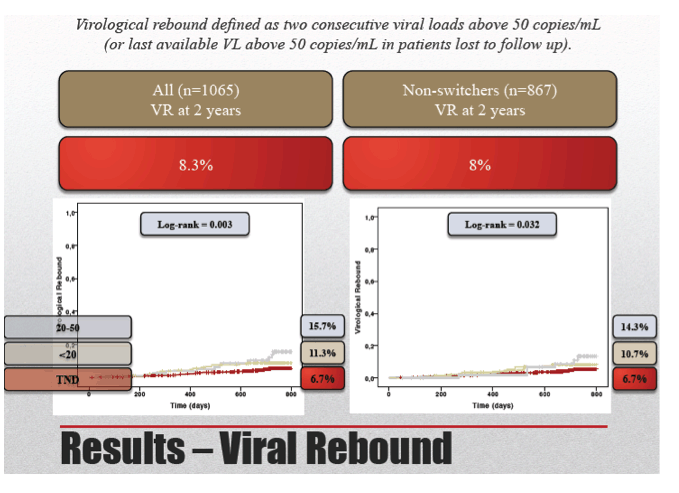
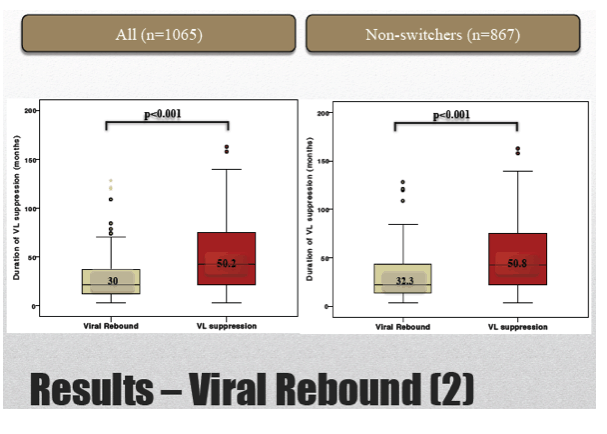
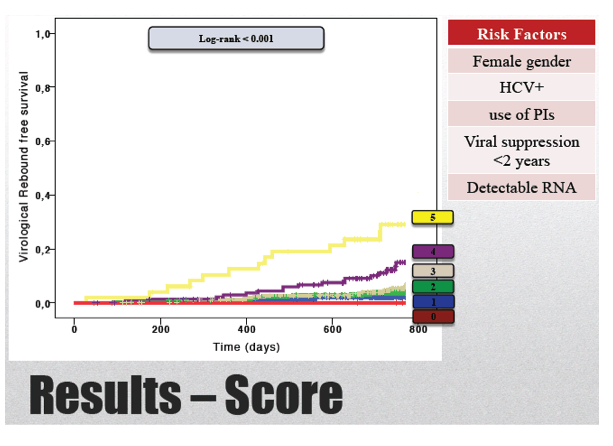
Of the 1065 study group members, 88 (8.3%) had a viral load rebound after 2 years of follow-up. Rebound rates were 6.7% in the TND group, 11.3% in the sub-20-copy group, and 15.7% in the 20-to-50-copy group. Median duration of virologic response was 50.2 months in those who maintained suppression and 30 months in rebounders (P < 0.001).
Limiting the analysis to 867 people who did not switch antiretrovirals during 2 years of follow-up, the researchers recorded an overall rebound rate of 8%, including 6.7% with TND, 10.7% with under 20 copies, and 14.3% with 20 to 50 copies. Among nonswitchers who maintained viral suppression, median duration of suppression was 50.8 months, compared with 32.3 months in rebounders (P < 0.001).
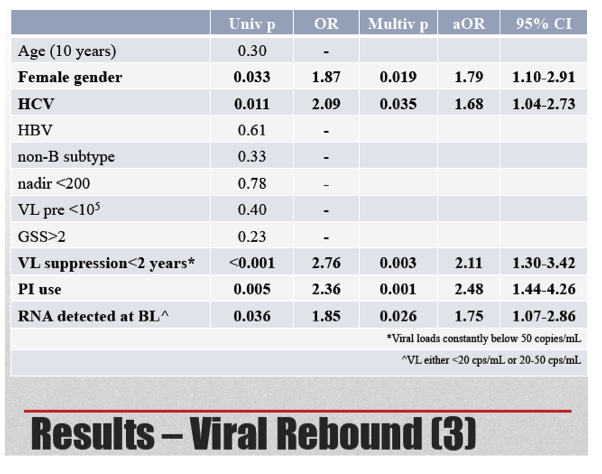
The Cox model identified five independent predictors of viral rebound:
-- Female gender: aOR 1.79, 95% CI 1.10 to 2.91, P = 0.019
-- HCV coinfection: aOR 1.68, 95% CI 1.04 to 2.73, P = 0.035
-- Viral suppression (<50 copies) less than 2 years: aOR 2.11, 95% CI 1.30 to 3.42, P = 0.003
-- PI vs nonnucleoside or raltegravir: aOR 2.48, 95% CI 1.44 to 4.26, P = 0.001
-- Viral load detectable at <50 copies vs TND at baseline: aOR 1.75, 95% CI 1.07 to 2.86, P = 0.026
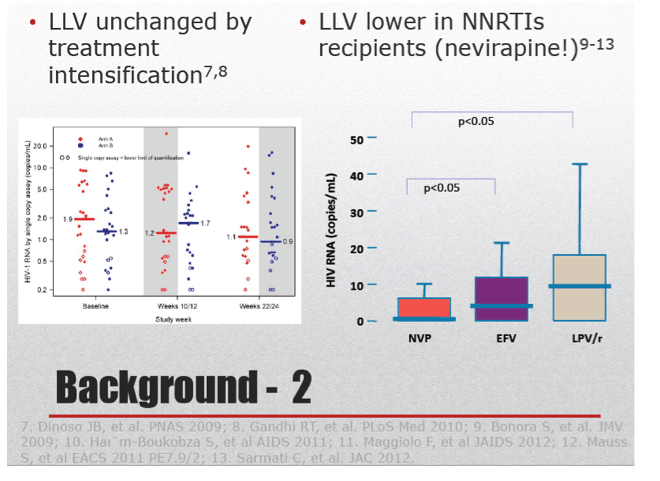
The researchers noted that their analysis is limited by its retrospective nature and by lack of formal adherence evaluation. They acknowledge that their findings may have limited clinical application because interventions such as treatment intensification may not push a viral load of 30 copies, for example, into the TND bracket. They suggested that switching to a nonnucleoside may help a person achieve TND status and that this strategy could be studied. Previous research found that sub-50 viral loads were significantly lower in people taking nevirapine than efavirenz or lopinavir/ritonavir.
References
1. Calcagno A, Ghisetti V, Lo Re S, et al. Level of HIV viremia below 50 copies/ml predicts virologic rebound in HAART-treated patients. 14th European AIDS Conference. October 16-19, 2013. Brussels. Abstract PS7/4.
2. Chun TW, Murray D, Justement JS, et al. Relationship between residual plasma viremia and the size of HIV proviral DNA reservoirs in infected individuals receiving effective antiretroviral therapy. J Infect Dis. 2011;204:135-138.
3. Pugliese P, Delpierre C, Cuzin L, et al. An undetectable polymerase chain reaction signal in routine HIV plasma viral load monitoring is associated with better virological outcomes in patients receiving highly active antiretroviral therapy. HIV Medicine. 2013;14:509-515.
-------------------------------------------------
PROGRAM ABSTRACT
Level of HIV Viremia below 50 Copies/ml Predicts Virologic Rebound in HAART-treated Patients
A. Calcagno1, V. Ghisetti2, S. Lo Re1, I. Motta1, T. Allice2, L. Marinaro1, M.C. Tettoni1, L. Trentini1, C. Bramato1, G. Orofino3, M. Bonasso1, B. Salassa1, G. Di Perri1, S. Bonora1
1Unit of Infectious Diseases, Department of Medical Sciences, University of Torino, Torino, Italy, 2Laboratory of Microbiology and Molecular Biology, Ospedale Amedeo di Savoia, ASLTO2, Torino, Italy, 3Unit of Infectious Diseases, Divisione A, Ospedale Amedeo di Savoia, ASLTO2, Torino, Italy
Objectives: To evaluate the predictivity of low level viremia (LLV) and other clinical features as a risk factors for virologic rebound in HAART-treated patients with HIV-RNA < 50 copies/ml.
Methods: HIV-infected patients on HAART with two consecutive HIV-RNA < 50 copies/ml in the first semester of 2010 were included. HIV-RNA was measured through real-time PCR Taqman 2.0 and stratified as "Target not detected" (TND), < 20 copies/mL ("< 20") and 20-50 copies/mL ("20-50"); virologic rebound was defined as two consecutive viral loads >50 copies/mL or last value >50 copies/mL in patients lost-to-follow up. A binomial logistic regression and a Cox proportional hazard model were used for multivariate analysis.
Results: 1065 patients (71.5% male; 28.5% HCV-positive) were included; mean age was 47.4 years (±11). Nadir and current CD4 cell count were 220/uL (±157) and 587/uL (±261), respectively.
At baseline, 782 patients (73.4%) showed TND 198 (18.6%) < 20 and 85 (8%) 20-50: female gender (0.003), CD4 nadir >200/uL (0.004) and NNRTIs or raltegravir use (0.04) were independent factors associated with TND.
In patients not changing HAART (n=867) 69 subjects (8%) showed virologic rebound.
Independent predictors of virologic rebound were previous HIV-RNA < 50 copies/mL for less than 2 years (0.001), detectable (< 20 or 20-50) HIV RNA at baseline (0.034), HCV infection (0.038), female gender (0.024) and PI use (0.005).
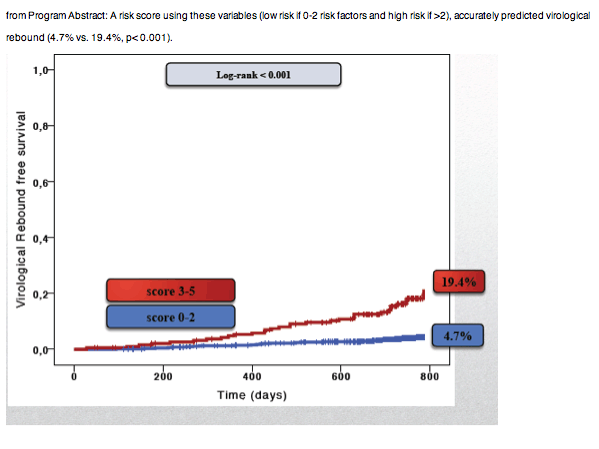
A risk score using these variables (low risk if 0-2 risk factors and high risk if >2), accurately predicted virological rebound (4.7% vs. 19.4%, p< 0.001).
Conclusion: In our study, level of viremia < 50 copies/ml, according to a commercial kit with 20 copies/mL cut-off, predicted virological rebound at 2 years of follow-up. A score including LLV along with gender, HCV-status, duration of previous HIV-RNA < 50 copies/ml and third-drug class may identify stable patients at increased risk of virologic rebound.





|
| |
|
 |
 |
|
|