 |
 |
 |
| |
BL-8030: A Novel, Potent, Selective, Orally Available Inhibitor of Hepatitis C Virus NS3/4A Protease
|
| |
| |
Reported by Jules Levin
EASL 2013 April 24-28 Amsterdam
Philippe Halfon1, Jerome Courcambeck1, Tony Whitaker2, Phillip M. Tharnish2, Tamara R. McBrayer2, Steven J. Coats2, Yaron Pereg3, Rinat Tabakman3, Johanna Schumann3, Meirav Matto3 and Raymond F. Schinazi4&
1Genoscience Pharma, Marseille, France; 2RFS Pharma LLC, Tucker, GA, USA; 3BioLineRx Ltd., Jerusalem, Israel; 4Center for AIDS Research, Laboratory of Biochemical Pharmacology, Department of Pediatrics, Emory University School of Medicine, and Veterans Affairs Medical Center, Decatur, GA, USA. & RFS is a founder and major shareholder of RFS Pharma, LLC
ABSTRACT
Background and aims: Hepatitis C Virus (HCV) NS3/4A protease inhibitors (PI) are likely to be a key component of future combination therapies for patients with chronic HCV infections. These agents have demonstrated dramatic antiviral effects, but tend to be genotype 1 specific and associated with unpleasant adverse effects. Furthermore, PIs have demonstrated a high risk for development of resistant virus. Based on structure-based drug design and NS3 molecular resistance mechanisms, a set of peptidomimetic NS3/4A protease inhibitors were synthesized. Here, we report the pharmacodynamic and pharmacokinetic profile of BL-8030 (formerly GNS-227), a low-nanomolar inhibitor of HCV.
Methods: The antiviral activity and selectivity of BL-8030 was assessed versus: 1) a Huh-7 cell based HCV replicon assay; 2) wild type and PI mutant enzymes and 3) a panel of human proteases to demonstrate specificity. Human cytochrome P450 enzyme inhibition and induction were also evaluated. The plasma pharmacokinetics (PK) and plasma to liver partition of BL-8030 following intravenous or oral administration in rats were evaluated.
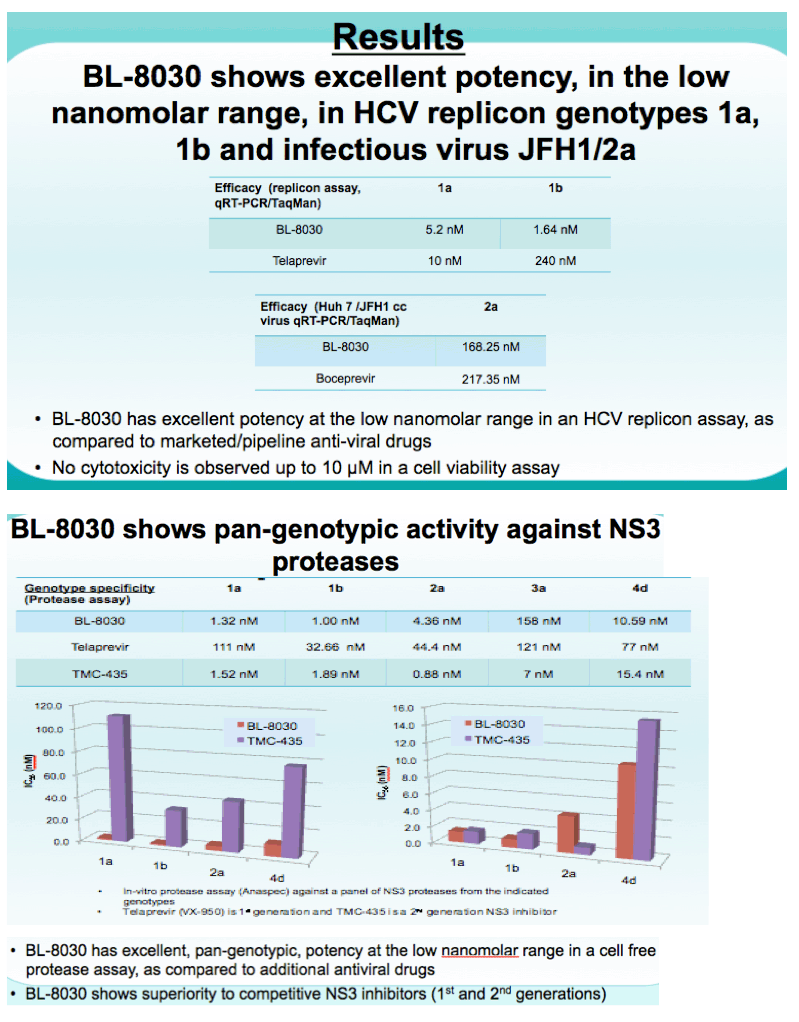
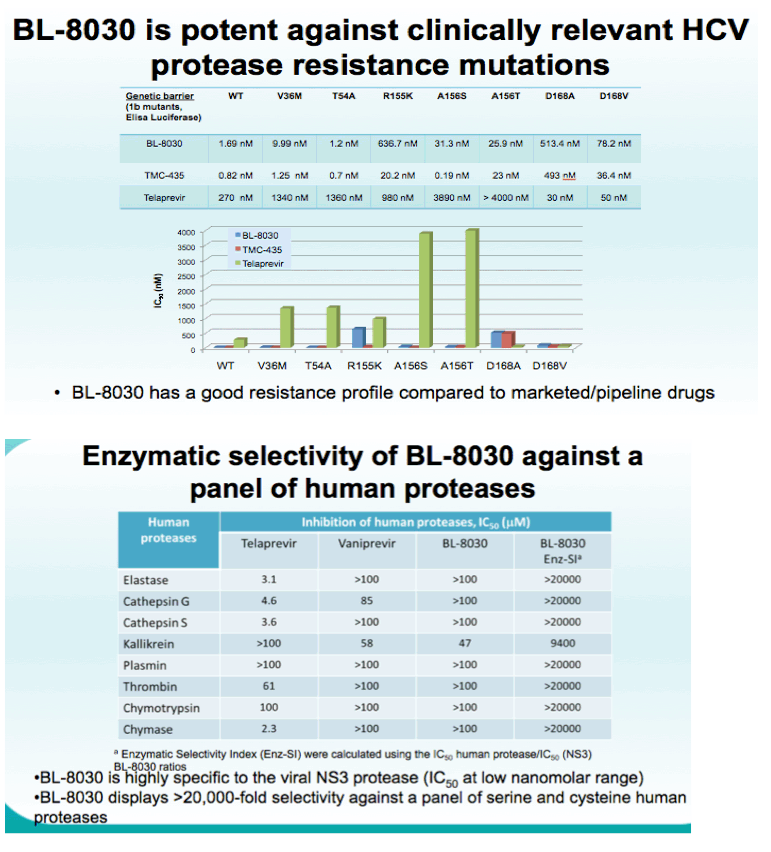
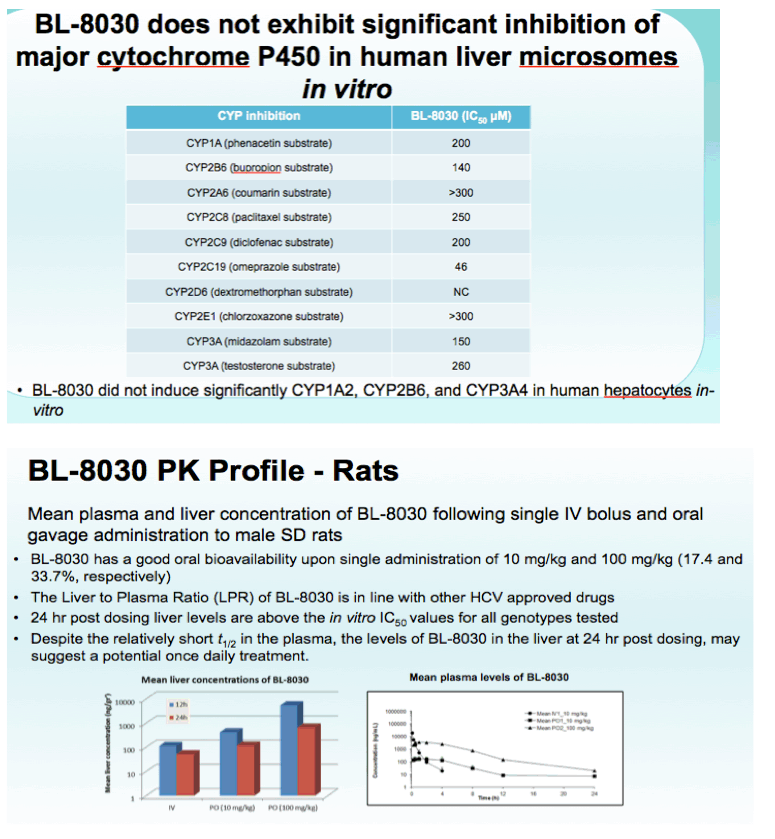
Results: BL-8030 demonstrated excellent potency, in the low nanomolar range, in HCV replicon genotypes 1a, 1b and 2a. Its pan-genotypic activity was tested on a panel of NS3 enzymes using a biochemical fluorimetric protease assay, in which BL-8030 demonstrated potent activity against genotypes 1a, 1b, 2a and 4. BL-8030 was also potent against the main, clinically relevant resistance mutations. BL-8030 showed several thousand-fold selectivity relative to a panel of human proteases. PK studies in rats indicated good oral bioavailability after single 10 and 100 mg/kg dosing (17.4 and 33.7%, respectively). Following oral administration, BL-8030 was distributed in the liver with a liver to plasma ratio ranging from 28.4 to 45.4 (24hr and 12hr post dosing). Importantly, BL-8030 levels in the liver 24 hours post dosing were above its EC50 suggesting the potential for once a day dosing in the clinic.
Conclusions: BL-8030 is a novel, highly potent and specific HCV NS3/4A protease inhibitor as demonstrated by cellular and biochemical assays and has the potential to prevent and treat emerging resistance variants. BL-8030 has a favorable pharmacological and safety profile with good oral bioavailability, liver distribution and the potential for once daily dosing.


|
| |
|
 |
 |
|
|