| |
Effects of Antidiabetic and Antihyperlipidemic Agents on C-Reactive Protein
|
| |
| |
Download the PDF here
Mayo Clinic Proceedings March 2008
PARESH DANDONA, MBBS, DPHIL, FRCP
From the State University of New York at Buffalo Diabetes-Endocrinology
Center of Western New York, Buffalo.
Type 2 diabetes mellitus (DM) increases the risk of cardiovascular disease, a major cause of morbidity and mortality. Central to type 2 DM is insulin resistance, a proinflammatory, hypercoagulable state that predisposes patients to develop cardiovascular disease and that is associated with risk factors for atherosclerosis including dyslipidemia, hypertension, inflammation, and altered hemostasis. Atherosclerosis is recognized as a chronic inflammatory disease of the arteries. C-reactive protein (CRP) is an acute-phase response protein that is considered both a marker of inflammation and a predictor of cardiovascular events including myocardial infarction, stroke, peripheral arterial disease, and sudden cardiac death. Evidence indicates that CRP has a direct proatherogenic effect through up-regulation of angiotensin II type 1 receptors and through the stimulation of other proinflammatory factors. Patients with type 2 DM tend to have higher CRP concentrations than do those without it, suggesting an increased role of inflammation in the accelerated atherosclerosis seen in these patients. Reducing CRP concentrations through lifestyle changes or pharmacotherapeutics could have clinical benefit; long-term studies are needed to determine whether reductions in CRP concentrations translate into improved cardiovascular outcomes. Because glucose and lipid levels as well as CRP concentrations are often elevated in patients with type 2 DM, an agent that positively affects multiple cardiovascular risk factors would be most beneficial. This article reviews available data on antidiabetic and antihyperlipidemic agents that reduce CRP concentrations in addition to their primary effect of lowering glucose or lipid levels.
Type 2 diabetes mellitus (DM) is a progressive and complex metabolic disorder characterized by chronic hyperglycemia and by disturbances in carbohydrate, lipid, and protein metabolism due to insulin resistance. Insulin resistance is caused by impaired insulin secretion and/or insulin action. It is a proinflammatory, hypercoagulable state that predisposes patients to develop cardiovascular disease, a major cause of morbidity and mortality. It is also associated with risk factors for atherosclerosis, including altered hemostasis, dyslipidemia, hypertension, and inflammation.1
Atherosclerosis is an inflammatory disease of the arteries. C-reactive protein (CRP), an acute-phase response protein synthesized in the liver in response to interleukin (IL) 1, IL-6, and tumor necrosis factor α (TNF-α), is known to be present in atheromatous plaques and can be a clinical marker of atherosclerosis.2 An elevated CRP concentration is a predictor of cardiovascular events, including myocardial infarction (MI), stroke, peripheral arterial disease, cardiac dysrhythmias, and sudden cardiac death (Table 1). In fact, CRP concentration is a stronger predictor of cardiovascular disease than is low-density lipoprotein cholesterol (LDL-C),3 even among healthy people with low LDL-C levels.4 Evidence suggests that CRP contributes primarily to the progression rather than to the initiation of atherosclerosis; the mechanism of this effect likely occurs through up-regulation of angiotensin II type 1 receptors, the major receptor for the proinflammatory molecule angiotensin II.5 However, CRP has been shown to inhibit nitric oxide production, balancing the effect of angiotensin II in addition to promoting angiogenesis; these findings suggest that CRP has a role in the initiation of atherosclerosis.6
Patients with type 2 DM tend to have higher CRP concentrations than those without it, suggesting that inflammation could contribute to the accelerated atherosclerosis seen in patients with type 2 DM.5 In fact, CRP was significantly associated with an increased risk of MI, coronary artery bypass grafting/angioplasty, and stroke in men with type 2 DM (P=.01).7 Furthermore, a 5-year study of patients with type 2 DM found that CRP was a stronger predictor of death (relative risk [RR], 3.3) and cardiovascular death (RR, 5.4) than were other cardiovascular risk factors.8 In addition, CRP was independently associated with nonfatal cardiovascular events in patients with type 2 DM,9 and a long-term study revealed that patients with type 2 DM and CRP concentrations of more than 0.3 mg/dL (to convert to nanomolesper liter, multiply by 0.845) had a greater risk of coronary artery disease (RR, 1.72) than did those with CRP concentrations of 0.3 mg/dL or less, and that this increased risk was independent of other cardiovascular risk factors.10
Healthy people have an average CRP concentration of approximately 0.25 mg/dL, but the concentration can increase to more than 50 mg/dL during acute infection. Before the development of the high-sensitivity CRP (hsCRP) test, only gross elevations in CRP concentration (approximately 1-100 mg/dL) could be detected. The current hsCRP test can detect normal or slightly elevated CRP concentrations (0.05-1.00 mg/dL). Consequently, hsCRP measurements to evaluate low-level systemic inflammation are recommended as a clinical tool to help predict cardiovascular or cerebrovascular disease.11
Reducing CRP concentrations through weight loss, improved nutrition, increased exercise, and smoking cessation is clinically beneficial and associated with positive cardiovascular outcomes.3 In addition to lifestyle changes, data have shown that certain drugs, including antidiabetic and antihyperlipidemic agents, can reduce CRP concentrations; however, studies are needed to determine whether reductions in CRP concentrations resulting from pharmacologic interventions translate into improved cardiovascular outcomes. This article reviews data on the effect of antidiabetic and antihyperlipidemic agents on CRP concentrations in addition to their primary indication for either glucose control or lipid lowering.
ROLE OF C-REACTIVE PROTEIN IN THE PATHOGENESIS OF INFLAMMATION
Plasma CRP concentration is known to be a key index of systemic inflammation; however, its importance as a marker of future cardiovascular risk is still under debate. A study by Ridker et al4 showed that patients in the highest quintile of CRP concentration (>0.419 mg/dL) have a 2.3 times greater risk of cardiovascular events than those in the lowest quintile (≤0.049 mg/dL) and that this risk is greater than that predicted by plasma LDL-C concentrations in the highest quintile. Furthermore, the magnitude of the decrease in CRP concentration has been shown to be a better predictor of the response to statin therapy than the magnitude of the decrease in LDL-C levels.12 In contrast, Danesh et al13 have shown that patients in the highest tertile have an increased risk (by a factor of 1.36) of cardiovascular events.
A better understanding of the role of CRP, if any, in the pathogenesis of inflammation would allow its potential as a marker of cardiovascular disease to be assessed. Several studies have shown that CRP exerts a proinflammatory effect on endothelial cells in vitro. It increases the expression of monocyte chemoattractant protein 1 (MCP-1), intercellular adhesion molecule 1 (ICAM-1), and plasminogen activator inhibitor 1 (PAI-1) in these cells. These effects are mediated by the Fcγ receptors I and II, which have been suggested recently as the putative receptors for CRP.14 In animals with experimental MI, CRP increases the size of the infarct and is expressed de novo in the infarcted zone.15 The injection of derivatives of phosphocholine, which bind to CRP in rats with experimental MI, reduces the size of the infarct.16 A similar approach is currently being developed for treating experimental ischemic stroke.
Some have questioned the in vitro data showing the proinflammatory effect of CRP, raising the possibility of contamination with endotoxin.17 Others have countered that objection, finding no evidence of such contamination.18 The injection of CRP into healthy humans has been shown to induce systemic inflammation with an increase in the biochemical indices of inflammation. These findings suggest that CRP is not only a marker of inflammation but also a mediator of inflammation.
ANTIDIABETIC AGENTS AND CRP
An increasing body of evidence shows that treatment with antidiabetic agents substantially reduces CRP concentrations. Although insulin appears to be the most potent agent, oral antidiabetic agents, such as biguanides (metformin), thiazolidinediones, and sulfonylureas, have also been shown to reduce CRP concentrations. However, the mechanism of this effect remains unclear. Clinical trial data suggest that CRP concentrations may be influenced only moderately by glycemic control. After 6 months of intensive antidiabetic treatment, more than 50% of patients with type 2 DM maintained CRP concentrations of greater than 0.3 mg/dL (concentrations consistent with high risk), despite achieving glycemic control.19 Furthermore, agents that produce comparable levels of glycemic control have differential effects on CRP concentrations, suggesting that no direct association exists between glycemic control and CRP concentrations.
Insulin
Insulin has been shown to have anti-inflammatory and profibrinolytic effects after acute ST-segment elevation MI. For example, increases in CRP concentrations were significantly (P<.05) attenuated in the group receiving dextrose, insulin, and potassium infusions vs the control group who received infusion with saline and potassium. At 24 hours, the absolute mean ± SD increase in CRP concentrations was 0.33±0.08 mg/dL in the control group vs 0.19±0.05 mg/dL in the group receiving insulin. At 48 hours, the corresponding increase in CRP concentrations was 0.55±0.09 mg/dL vs 0.31±0.06 mg/dL.20 It is important to note that only 2 patients in each group had type 2 DM and that no significant difference in plasma glucose level was observed between the control and insulin groups at either baseline (146 mg/dL vs 132 mg/dL [to convert to millimoles per liter, multiply by 0.0555]) or 48 hours (131 mg/dL vs 124 mg/dL). Similarly, patients without type 2 DM undergoing coronary artery bypass grafting had elevated CRP and serum amyloid A (SAA) concentrations; treatment with insulin, glucose, and potassium resulted in a significant reduction in concentrations of CRP (by 40% compared with control; P<.04) and SAA (by 40% compared with control; P=.03) despite the fact that CRP concentrations during coronary artery bypass grafting are 30 times greater than during acute MI.21 More recently, a study confirmed that insulin infusion reduces CRP concentrations by 40% in patients undergoing coronary artery bypass grafting; maintaining comparable levels of normoglycemia by intravenous insulin bolus did not reduce CRP concentrations.22 These findings suggest that intravenous insulin infusion could be necessary to increase plasma insulin concentrations sufficiently to obtain the anti-inflammatory effect.
Similarly, in patients with type 2 DM or elevated blood glucose concentrations, the maintenance of tight glycemic control, via infusion with insulin-dextrose within 24 hours after acute MI, also attenuated the increase in CRP concentration on day 2 of the infusion.23 In this study, both the direct effect of insulin and the reduction in blood glucose concentrations helped suppress the inflammatory response. Similarly, patients with poorly controlled type 2 DM showed a significant reduction in CRP concentrations after 2 weeks of insulin treatment (P=.01); this decrease was particularly pronounced in patients whose weight was unchanged or increased after initiation of therapy.24
Because CRP concentrations have been shown to be related to the magnitude of myocardial damage in patients with acute MI,15, 25, 26 insulin-induced reductions in CRP concentrations could have clinical applications. A low-dose infusion of insulin reduced CRP concentrations and was cardioprotective. The cardioprotective effect was similar to that observed in rats and dogs with experimental MI given insulin at the time of reperfusion; the reduction in the infarct was associated with a reduction in apoptosis.27, 28 Furthermore, in a rat model of MI, injection of human CRP caused the infarct to enlarge by 40%,15 whereas administration of small molecules that bind CRP reduced the infarct.16 This CRP-based approach to infarct reduction offers promise as a clinical therapy.
In vitro studies have shown that insulin, at physiologic levels, inhibits the expression of ICAM-1, MCP-1, and nuclear factor κB (NF-κB), a proinflammatory transcription factor.29, 30 The rapid and impressive reduction in CRP (and SAA) concentrations after insulin infusion is similar to that observed in patients undergoing surgery who were given 8 mg of dexamethasone.31
Metformin
The insulin sensitizer metformin decreases fasting plasma glucose and glycated hemoglobin (A1c) levels in patients with type 2 DM. Tight glycemic control with metformin has also been shown to reduce the risk of cardiovascular complications,32 possibly by reducing inflammation, measured clinically by a reduction in CRP concentration.19, 33, 34 A small study of 12 patients with poorly controlled type 2 DM reported a significant reduction in CRP concentrations (by 33%; P<.01) when metformin was added to sulfonylurea therapy for 4 months; no appreciable effects on blood pressure or levels of LDL-C, high-density lipoprotein cholesterol (HDL-C), triglycerides, or PAI-1 were observed.33 In overweight patients with type 2 DM, metformin reduced the concentration of CRP but not that of another inflammatory marker, complement factor C3,34 suggesting that metformin regulates CRP specifically. Metformin could interact with factors that affect CRP synthesis or secretion, which are unrelated to interactions that improve insulin sensitivity and reduce chronic inflammation.34
A study was performed to assess whether the beneficial cardiovascular effects of metformin observed in patients with type 2 DM result from a reduction in inflammation. In contrast to results reported in patients with type 2 DM, metformin had no effect on CRP and TNF-α concentrations in patients with impaired glucose tolerance,35 suggesting that metformin likely reduces levels of inflammatory markers by reducing hyperglycemia, improving insulin sensitivity, and/or promoting weight loss in patients with type 2 DM.
Metformin could also exert its anti-inflammatory effect via macrophage migration inhibitory factor (MIF), a proinflammatory cytokine involved in innate and adaptive immunity. After 6 weeks of metformin treatment, patients who were obese showed a significant reduction in MIF concentration (from 230 ng/dL to 160 ng/dL [to convert to nanomoles per liter, multiply by 0.00081]; P<.05), which was independent of changes in body weight or levels of glucose, insulin, and free fatty acids.36 By reducing MIF concentrations, metformin could have an antiatherogenic effect.36
Peroxisome Proliferator-Activated Receptor γ Agonists
Members of the nuclear receptor family of peroxisome proliferator-activated receptors (PPARs) can be classified into 3 isoforms: PPAR-α, PPAR-γ, and PPAR-δ. These molecules are ligand-activated transcription factors that regulate genes involved in both lipid and glucose metabolism and are therefore implicated in metabolic disorders, such as dyslipidemia and type 2 DM, that predispose patients to develop atherosclerosis. Thiazolidinediones are PPAR-γ agonists used in patients with type 2 DM to enhance insulin sensitivity and decrease insulin resistance. Peroxisome proliferator-activated receptor γ agonists also have an anti-inflammatory effect, initially thought to be mediated only through PPAR-α. However, the PPAR-γ agonist troglitazone was shown to reduce CRP concentrations in patients who were obese, both those with (P<.01)37 and without (P<.05)38 type 2 DM. Although troglitazone was removed from the market, the current thiazolidinediones have also been shown to attenuate CRP concentrations and produce antiatherogenic effects.39
Multiple studies have shown that thiazolidinediones reduce CRP concentrations in people with and without type 2 DM, including those with additional cardiovascular risk factors such as obesity or hypertension (Table 2).40, 41, 42, 43, 44, 45, 46, 47, 48, 49 Significant reductions in CRP concentrations have been consistently observed after 6 to 26 weeks of treatment with rosiglitazone, compared with baseline,40, 41, 42, 43, 44 placebo,40, 41 or no treatment. In many cases,43, 44 these reductions in CRP concentrations occurred in patients receiving established treatments (including metformin42 or a statin40 therapy) that could have already reduced the CRP concentrations, suggesting an additive effect. Although a correlation was observed between the change in CRP concentrations and insulin resistance (measured by homeostasis model assessment) after rosiglitazone treatment in 2 studies,41, 44 no correlation was found in an independent study,42 suggesting that these events could be independent.
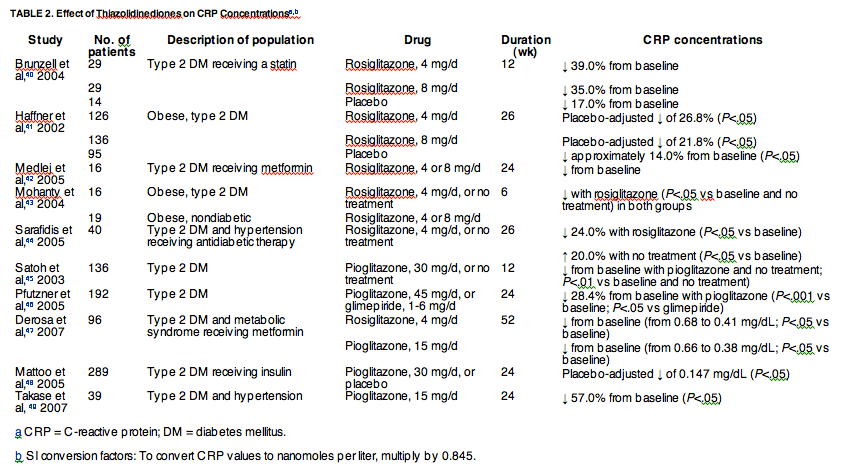
Like rosiglitazone, pioglitazone can produce marked reductions in CRP concentrations. In a 3-month study in patients with type 2 DM, CRP concentrations were significantly lower after treatment with pioglitazone than at baseline or in controls (Table 2).45 In addition, fasting plasma glucose, A1c, and LDL-C levels decreased (P<.05 for all) and adiponectin concentration increased (P<.01).45 When glimepiride and pioglitazone treatment were compared in patients with type 2 DM, both treatments significantly reduced A1c; however, only pioglitazone significantly reduced CRP concentrations relative to baseline.46 Results from the Prospective Pioglitazone Clinical Trial in Macrovascular Events (PROactive), a study of 5238 patients with type 2 DM, found that pioglitazone was effective for the prevention of cardiovascular events in high-risk patients (hazard ratio, 0.84; P=.03).50 However, it has yet to be determined whether the beneficial cardiovascular effect of pioglitazone correlates with reduced CRP concentrations.
Thiazolidinediones were recently reported to potentially increase the risk of cardiovascular events. Although 3 independent analyses found that rosiglitazone significantly increased the risk of MI and death,51, 52, 53 pioglitazone was reported to reduce significantly the risk of MI, stroke, and death relative to control.54
Adipose tissue macrophages contribute substantially to the expression of proinflammatory molecules, such as IL-6,55 which induce expression of CRP. By suppressing production of IL-6,56, 57, 58, 59 thiazolidinediones could reduce expression of CRP. In addition, the anti-inflammatory effect of thiazolidinediones could extend beyond suppression of CRP production to inhibition of CRP action. In vitro, rosiglitazone inhibits the effects of CRP on endothelial progenitor cell proliferation and promotes cell survival and function,60 potentially preserving vascular endothelial function. Rosiglitazone exerts an early and potent anti-inflammatory effect; within 3 days, NF-κB binding by mononuclear cells and plasma concentrations of MCP-1, ICAM-1, PAI-1, and SAA were suppressed.43 Furthermore, even when levels of plasma glucose, free fatty acids, or insulin were not reduced, rosiglitazone increased adiponectin levels and decreased resistin, CRP, and SAA levels.61 Given that significant dose-related weight gain occurred with rosiglitazone, the alterations in adiponectin and resistin levels and in CRP and SAA concentrations are likely related to its anti-inflammatory effect.61
Sulfonylureas
Studies have shown that sulfonylureas, which are insulin secretagogues, only modestly affect CRP concentrations in patients with type 2 DM.46, 62, 63 Glimepiride produced only a marginal (4%) reduction in CRP concentrations after 6 months (0.326 mg/dL to 0.313 mg/dL).46 Greater reductions in CRP concentrations have been observed with metformin and insulin than with sulfonylureas. Mean CRP concentrations were significantly lower with metformin than with glyburide treatment (0.56 mg/dL vs 0.83 mg/dL; P=.001) in patients with well-controlled type 2 DM and metabolic syndrome.62 Similarly, both insulin and glyburide reduced CRP concentrations from baseline in patients with type 2 DM; however, the reduction was significantly greater with insulin (54% vs 40%; P<.05).63
ANTIHYPERLIPIDEMIC AGENTS AND CRP
Patients with type 2 DM tend to have a characteristic dyslipidemia (ie, increased concentrations of LDL-C and decreased concentrations of HDL-C), likely responsible for their being 2 to 4 times more inclined to develop cardiovascular disease than those without type 2 DM.64 In fact, patients with type 2 DM are twice as likely as those without type 2 DM to have elevated triglyceride levels and decreased HDL-C concentrations.65 Even when effective glycemic control is achieved with antidiabetic treatment, dyslipidemia persists in many patients with type 2 DM. As a result, the American Diabetes Association recommends that patients with type 2 DM achieve an LDL-C target of less than 100 mg/dL (to convert to millimoles per liter, multiply by 0.0259) to reduce long-term cardiovascular risk. Recommended lipid-lowering agents for patients with type 2 DM include 3-hydroxy-3-methylglutaryl-coenzyme A reductase inhibitors (statins), nicotinic acid (niacin), fibrates, and bile acid sequestrants.66 Like the glucose-lowering therapies, many lipid-lowering agents affect CRP concentrations in addition to improving lipid profile.
Statins
Although they do not affect glucose levels, statins have a pivotal role in the management of type 2 DM and the metabolic syndrome. The predominant effect of statin treatment is the reduction of LDL-C and total cholesterol levels; these agents also reduce triglyceride levels and increase HDL-C levels. A retrospective analysis of well-controlled trials in patients with coronary artery disease showed that coronary artery disease risk was associated with elevated CRP concentrations and that statin therapy attenuated both the CRP elevation and the cardiovascular risk.67 Prompted by these findings, a number of prospective studies investigated the effects of statins on CRP concentrations (Table 3).12, 68, 69, 70, 71, 72, 73, 74, 75 These studies showed a consistent reduction in CRP concentrations with statins, both in a primary and a secondary prevention setting. In the Cholesterol And Recurrent Events (CARE) study,67, 72 the Air Force/Texas Coronary Atherosclerosis Prevention Study (AFCAPS/TexCAPS),12 and the Pravastatin or Atorvastatin Evaluation and Infection Therapy (PROVE-IT) study,71 statin treatment lowered CRP concentrations independently of its lipid-lowering effects and reduced the risk of cardiovascular events by approximately 30%, supporting CRP as an independent predictor of cardiovascular events. More intensive statin therapy resulted in greater reductions in CRP concentrations70, 71, 73, 74, 75; however, in the PROVE-IT study, even the most intensive regimen of atorvastatin (80 mg/d) resulted in only 43.9% of patients achieving both a CRP concentration of less than 0.2 mg/dL and anLDL-C level of less than 130 mg/dL; achieving both goals reduced the risk of a recurrent cardiovascular event by 35%.71 Reduction in CRP concentrations was significantly greater with 80 mg than with 10 mg of atorvastatin after 30 weeks.74 Clearly, statin therapy offers a benefit to patients with type 2 DM.
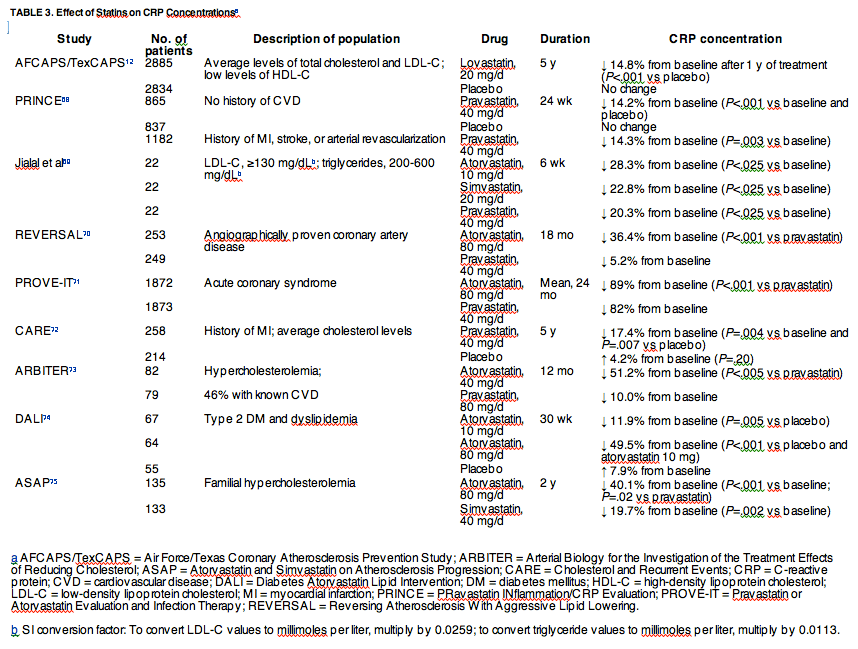
A prospective study showed that the ability of statins to lower CRP concentrations is a class effect; dosages of 40 mg of pravastatin, 20 mg of simvastatin, or 10 mg of atorvastatin similarly reduced total cholesterol, LDL-C, and triglyceride levels as well as CRP concentrations in patients with hyperlipidemia.69 However, the CRP effect with statins appears to be independent of its lipid effects; a significant correlation between change in CRP concentrations and LDL-C levels was observed in only 1 study,75 with no correlation seen in 5 others.12, 68, 69, 72, 74 Currently, a prospective randomized trial (Justification for the Use of Statins in Primary Prevention [JUPITER]) is investigating rosuvastatin for prevention of cardiovascular disease in patients with normal LDL-C levels and elevated CRP concentrations without heart disease or type 2 DM.76
The production of CRP in the liver is thought to be initiated via IL-6-induced geranylgeranylation of Ras-related C3 botulinum toxin substrate 1 (Rac-1). Activated Rac-1 subsequently phosphorylates signal transducer and activator of transcription 3 (STAT3), a transcription factor, thereby inducing expression of CRP. Statins inhibit the L-mevalonate pathway, which is responsible for protein geranylgeranylation. As a result, Rac-1 is prevented from phosphorylating STAT3.77 The CRP-lowering effect of statins is therefore thought to result from inhibition of these protein modifications.
Nicotinic Acid
Nicotinic acid (niacin) is used in conjunction with a restricted diet to reduce cholesterol levels. Niacin has a favorable effect on the lipid profile of patients with type 2 DM; at concentrations of approximately 3000 to 4000 mg/d, niacin markedly increases HDL-C levels while lowering levels of LDL-C, triglyceride, and apolipoprotein A-I. Although these effects target the dyslipidemia that is most marked and prevalent in patients with type 2 DM, the use of niacin is limited by its potential to increase fasting plasma glucose concentrations at high doses (greater than 3000 mg/d) and by the high prevalence of flushing.78 Currently, limited data suggest that niacin monotherapy lowers CRP concentrations.79 In patients with type 2 DM and dyslipidemia (47% of whom receive statin therapy), 1000 or 1500 mg/d of extended-release niacin for 16 weeks reduced CRP concentrations by 11% and 20%, respectively, compared with placebo.78 Glycemic control was minimally affected and correlated more closely with the higher dose of niacin.
Fibrates
Fibrates, including bezafibrate, fenofibrate, and gemfibrozil, are PPAR-α agonists used to correct dyslipidemia in patients with type 2 DM. Although fibrates primarily affect triglyceride metabolism, having the greatest effect on triglyceride and HDL-C levels,80 these agents also have shown beneficial effects beyond lipid lowering. Both primary and secondary prevention trials have shown that fibrates reduce coronary artery disease incidence. The 5-year Fenofibrate Intervention and Event Lowering in Diabetes (FIELD) study in 9795 patients with type 2 DM reported significant reductions in total cardiovascular disease events (from 13.9% to 12.5%; P=.04) and microvascular-associated complications (P=.002, for reduced albuminuria progression; P<.001, for reduced retinopathy requiring laser surgery) with fenofibrate treatment.81
The CRP-lowering effects of fibrates have been shown in differing patient populations with mixed/combined hyperlipidemia.82 Fibrates are as effective as other classes of lipid-lowering drugs, eg, statins, in lowering CRP concentrations. They also are associated with up to a 50% reduction in CRP levels.83, 84, 85, 86, 87, 88 Fenofibrate (250 mg/d for 4 weeks) lowered CRP concentrations in patients with mild hyperlipidemia, both those with and without coronary artery disease; however, the effect was greatest in those with coronary artery disease and was maintained for up to 6 weeks after treatment discontinuation.83 This study also showed that, in vitro, activators of PPAR-α inhibit the inflammatory response of aortic smooth-muscle cells, decreasing the concentrations of CRP in these cells. In patients with impaired glucose tolerance and high plasma concentrations of oxidized LDLs, CRP, fibrinogen, factor VII, and PAI-1 at study entry, treatment with fenofibrate for 30 days markedly reduced these concentrations, improved the lipid profile, and improved glucose metabolism compared with controls.89
Peroxisome proliferator-activated receptor α agonists exert their anti-inflammatory effects by antagonizing the NF-κB pathway. Fibrates induce the expression of inhibitor of κB, which acts to repress NF-κB, thereby preventing activation of genes involved in the inflammatory response, including IL6, which contributes to up-regulation of CRP. In vitro, fibrates do not affect inhibitor κBα protein degradation or inhibitor κB-kinase activity.90
Bile Acid Sequestrants
Bile acid sequestrants are primarily indicated for lowering cholesterol; these agents can significantly reduce LDL-C levels and modestly increase HDL-C levels compared with placebo.91, 92, 93, 94 Colesevelam hydrochloride, a specifically engineered bile acid sequestrant, can be used in monotherapy or in combination with other lipid-lowering agents to improve the lipid profile of patients with hypercholesterolemia.95 The nonsystemic nature of colesevelam could reduce the potential for drug-drug interactions via the cytochrome P450 pathway.96
Colesevelam is the only bile acid sequestrant investigated for its effects on CRP.97 According to a meta-analysis of 3 randomized, double-blind trials of patients with hypercholesterolemia, the addition of 3.75 g/d of colesevelam to established statin therapy (simvastatin, atorvastatin, or pravastatin) for 6 weeks significantly reduced CRP concentrations by 23% (P=.007).97 Similar reductions were reported with colesevelam in monotherapy; in patients with mild hypercholesterolemia, 3.75 g/d of colesevelam significantly reduced CRP concentrations compared with those at baseline and with placebo (15.9% and 18.7%, respectively; P<.03), in addition to lowering LDL-C levels (P<.001) after 6 weeks. No correlation was observed between LDL-C and CRP lowering, and no significant changes in plasma IL-6 or TNF-α concentrations were reported.98
Recent data have shown that colesevelam improves glycemic control in patients with type 2 DM inadequately controlled with existing metformin or sulfonylurea treatment.99 These preliminary results were substantiated in a clinical trial in patients with type 2 DM with inadequate glycemic control on insulin, whether administered alone or in combination with oral antidiabetic agents. In this study, treatment with 3.75 g/d of colesevelam for 16 weeks significantly reduced CRP concentrations (by 18.6%; P=.003) as well as A1c, fructosamine, and LDL-C levels (P<.001 for all).100 Similarly, reductions in CRP concentration were reported when colesevelam was combined with either metformin (14.0%)101 or sulfonylurea (11.0%).102
Ezetimibe
Ezetimibe can be used in monotherapy or in combination with a statin to reduce levels of LDL-C and total cholesterol by inhibiting cholesterol absorption from the intestines; however, this agent does not affect triglyceride levels.88 Although ezetimibe monotherapy has not been shown to lower CRP concentrations significantly,88, 103 it can result in significant reductions in CRP concentrations when combined with a statin.103, 104 In patients with hypercholesterolemia, simvastatin monotherapy (10-80 mg) reduced CRP concentrations by 18.2%; a combined regimen of simvastatin and 10 mg of ezetimibe reduced CRP concentrations by almost twice as much (34.8%; P<.01) at week 12, suggesting that ezetimibe enhances the CRP-lowering effect of statins.105
CONCLUSION
Increasingly prevalent, type 2 DM is a major health concern and is associated with substantial cardiovascular risk. C-reactive protein is a marker of inflammation and a strong predictor of cardiovascular risk. Interventions that lower CRP concentrations could reduce cardiovascular risk, especially in patients with type 2 DM. A single agent that could reduce CRP concentrations, while also improving glycemic control and the lipid profile, could be especially beneficial for these patients.
Of the antidiabetic and antihyperlipidemic agents, insulin causes the most pronounced and rapid reduction in CRP concentrations, ie, a 40% reduction within 24 hours.20, 21, 23 Among oral antidiabetic agents, accumulating evidence shows that metformin reduces CRP concentrations in patients with type 2 DM. However, this effect is probably dependent on improving glycemic control.
Thiazolidinediones markedly reduce CRP concentrations and improve glycemic control. Although sulfonylureas can decrease CRP concentrations, the reported reductions are small and could depend on decreasing blood glucose concentrations. Among the antihyperlipidemic agents, statins reduce CRP concentration independently of their LDL-C-lowering action. Fibrate therapy has shown variable effects on CRP concentrations depending on the patient population. The limited data regarding niacin suggest that this agent moderately reduces CRP concentrations. Colesevelam is the only bile acid sequestrant shown to substantially reduce CRP concentrations in patients with type 2 DM. Finally, ezetimibe monotherapy appears to have no effect on CRP concentrations; however, when combined with a statin, ezetimibe has the additive effect of lowering CRP concentrations. Long-term outcome studies are needed to determine whether lowering CRP concentrations in patients with type 2 DM will translate into a reduction in cardiovascular risk.
| |
| |
| |
|
|
|