 |
 |
 |
| |
BIT225, a Novel Assembly Inhibitor, Cuts HIV Load in Monocyte Reservoir
|
| |
| |
7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur
Mark Mascolini
BIT225, a novel antiretroviral that inhibits viral assembly by blocking Vpu ion channel activity, reduced HIV dissemination from monocytes in the first trial of this agent in HIV-positive people [1].
Biotron scientists developed BIT225 to disrupt HIV assembly in macrophages, early targets of HIV infection that become a persistent viral reservoir. BIT225 reduces viral release from monocyte-derived macrophages by stifling ion channel activity of the HIV protein Vpu [2]. A phase 1 trial demonstrated the tolerability of BIT225 in healthy volunteers. BIT044 is a phase 1b/2a trial of BIT225 in HIV-positive adults in Thailand.
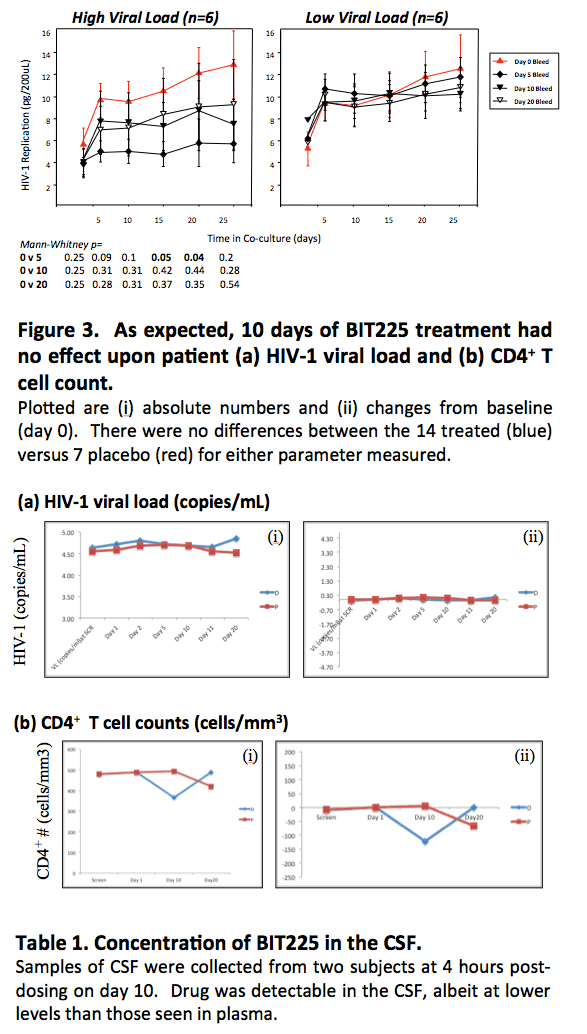
The trial took a new approach to assess HIV activity in CD14 monocytes during 10 days of BIT225 monotherapy by measuring HIV output from cells isolated from BIT225-treated people. The Biotron team did not directly measure viral load in study participants, anticipating that falling viral loads would be unlikely in a brief monotherapy trial of an agent that targets HIV in macrophages.
The investigators randomized 14 antiretroviral-naive adults to BIT225 and 7 to placebo. Participants had a viral load above 5000 copies and a CD4 count above 350. The investigators collected blood on days 0, 5, 10, and 20, isolating CD14 monocytes by magnetic bead sorting at each time point. The Biotron team combined isolated CD14 monocytes with MT4 cells and cocultured the cells ex vivo for 25 days. They determined HIV replication in coculture supernatants by p24 assay after 5, 10, 15, 20, and 25 days of culture.
There were 7 men and 7 women in the BIT225 arm and 4 men and 3 women in the placebo arm. Age averaged 30.4 in the BIT225 group and 27 in the placebo group. Median (range) viral load stood at 27,997 copies (3560 to 276,930) in people randomized to BIT225 and 20,521 copies (6109 to 81,829) in those randomized to placebo. Respective median (range) CD4 counts were 441 (299 to 835) and 482 (261 to 617).
Two people withdrew from the BIT225 group because of grade 1 and 2 headache, nausea, and vomiting. All placebo patients completed the trial. Twelve of 14 people randomized to BIT225 complained of headache, 9 complained of nausea, and 5 had vomiting. Respective numbers in the 7-person placebo group were 2, 2, and 1. One person interrupted BIT225 for 2 days because of palpitations. After the trial Biotron developed "an optimized capsule formulation" of BIT225 and tested it in healthy volunteers.
In the 7 participants receiving placebo, the amount of virus in CD14 monocytes remained constant throughout the trial. In the 12 treated participants receiving BIT255, the amount of virus in cocultured monocytes declined from day 0 to day 5 and remained lower throughout 20 days of treatment. The researchers divided the 12 BIT225-treated people into two groups determined by median viral load. The 6 participants with a viral load above the median 4.43 log10 copies had significantly less virus in cocultured monocytes on day 5 and later than on day 0.
The investigators also stored CD14-positive and negative T cells for single-copy HIV RNA and DNA assays. As they anticipated, BIT225 had no effect on viral load or CD4 count through 10 days of treatment.
In 2 participants who gave cerebrospinal fluid (CSF) samples 4 hours after dosing on day 10, BIT225 concentrations were detectable in CSF, though at lower levels than in plasma (87.8 versus 3550 ng/mL in one patient, 24.4 versus 1620 ng/mL in the other patient).
The investigators concluded that BIT225 has a significant impact on viral burden in monocytes of people with high viral loads. They proposed that "by targeting these cells and preventing (re)seeding of the myeloid reservoirs, BIT225 has a potential role in the eradication strategy of HIV-1."
References
1. Wilkinson J, Luscombe C, Ewart G, et al. BIT225, a novel Inhibitor of HIV-1 release from HIV-1 reservoirs of the myeloid lineage. 7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur. Abstract MOLBPE11.
2. Khoury G, Ewart G, Luscombe C, Miller M, Wilkinson J. Antiviral efficacy of the novel compound BIT225 against HIV-1 release from human macrophages. Antimicrob Agents Chemother. 2010;54:835-845. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2812169/
BIT225, a Novel Inhibitor of HIV-1 Release from HIV-1 Reservoirs of the Myeloid Lineage
John Wilkinson*1, Carolyn Luscombe1, Gary Ewart1, Stephen Kerr2, Nattaya Tanliang3, Winai Ratanasuwan3, Robert Murphy4, and Michelle Miller1
1Biotron Limited, Sydney, Australia 2HIV-NAT, Bangkok, Thailand 3Siriraj Hospital, Bangkok, Thailand and 4Northwestern University, Chicago, IL, US




|
| |
|
 |
 |
|
|