 |
 |
 |
| |
Standard Triple Therapy Controls HIV Better Than Raltegravir/Darunavir at 48 Weeks RADAR Study
|
| |
| |
7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur
Mark Mascolini
Darunavir/ritonavir plus tenofovir/emtricitabine (TDF/FTC) produced a better virologic response rate than darunavir/ritonavir plus twice-daily raltegravir in a 48-week randomized trial [1]. Bone but not renal changes were significantly worse with the TDF/FTC regimen through 48 weeks. Total cholesterol rose more with raltegravir/darunavir.
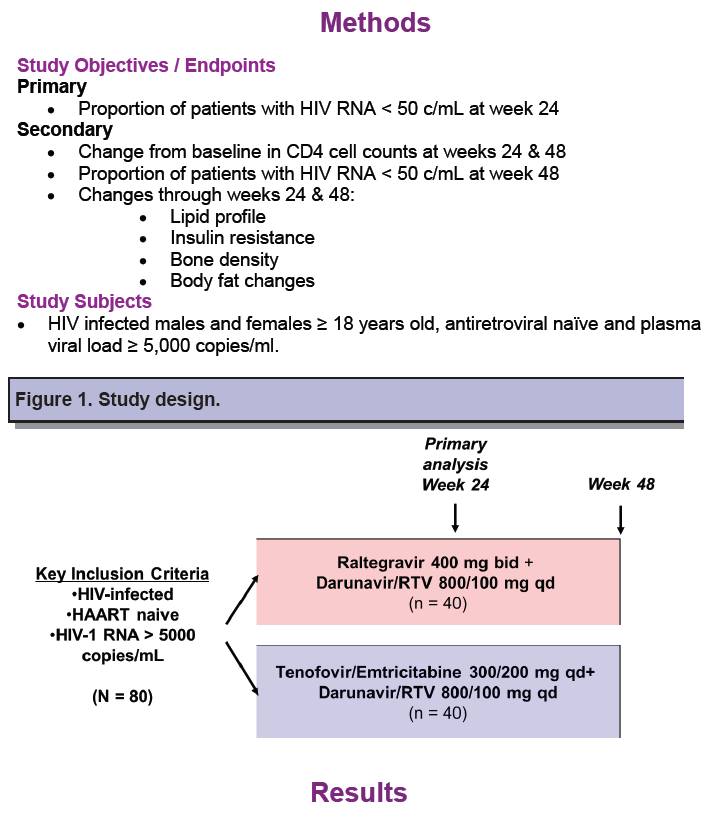
For several years, researchers have tested nucleoside-sparing first-line and maintenance regimens on the grounds that some newer antiretrovirals have better safety records than nucleosides, including TDF/FTC, the preferred first-line backbone in US antiretroviral guidelines. Clinical investigators in Texas and Pennsylvania mounted this open-label pilot study, RADAR, to compare efficacy and safety of TDF/FTC and raltegravir, both given with standard once-daily darunavir/ritonavir. The primary endpoint was the proportion of participants in each arm with a 24-week viral load below 48 copies by a noncompletion-equals-failure analysis.
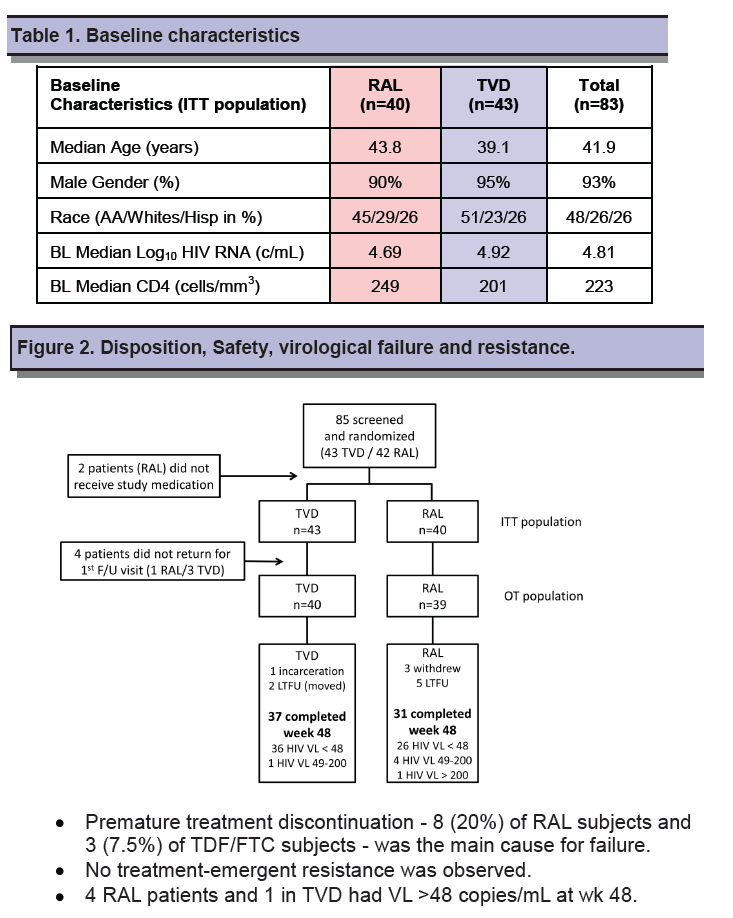
Study participants were antiretroviral-naive adults with a viral load of at least 5000 copies. RADAR investigators randomized 40 to the raltegravir arm and 43 to the TDF/FTC arm. Most study participants (93%) were men and almost half in each group were African American. Median age was higher in the raltegravir group (43.8 versus 39.1). Median pretreatment viral load was lower in the raltegravir arm (4.69 versus 4.92 log10 copies/mL), and median CD4 count was higher (249 versus 201).
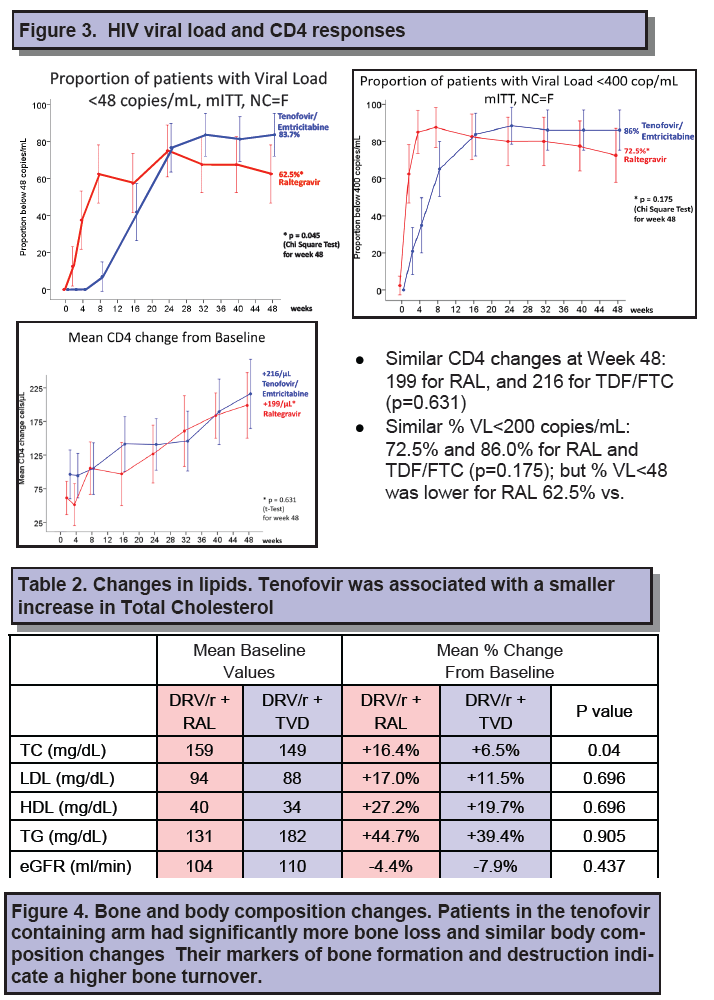
Thirty-one people randomized to raltegravir (77.5%) and 37 randomized to TDF/FTC (86%) completed 48 weeks of treatment. In the noncompletion-equals-failure analysis, 62.5% randomized to raltegravir and 83.7% randomized to TDF/FTC had a week-48 viral load below 48 copies, a significant difference (P = 0.045). Proportions with a 48-week viral load below 400 copies also favored TDF/FTC (86% versus 72.5%), though that difference lacked statistical significance (P = 0.175).
Premature discontinuation proved the main cause of failure--in 8 people (20%) randomized to raltegravir and 3 (7.0%) randomized to TDF/FTC. But virologic failure also played a role: 4 people (10%) assigned to raltegravir and 1 (2%) randomized to TDF/FTC had a viral load above 48 copies at week 48. The investigators speculated that the worse outcomes with raltegravir could reflect the need for twice-daily dosing or pharmacokinetic interactions between raltegravir and darunavir/ritonavir. CD4 gains through 48 weeks averaged 199 with raltegravir and 216 with TDF/FTC (P = 0.63).
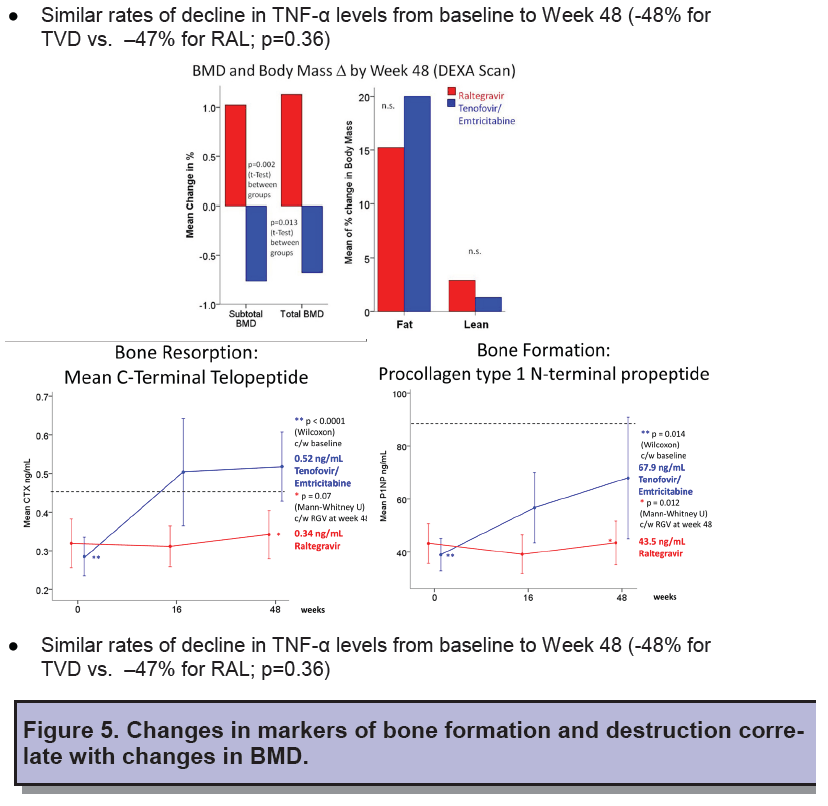
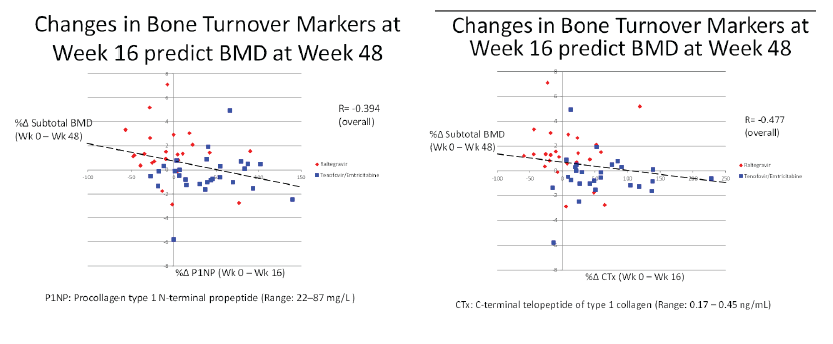
Total cholesterol rose more with raltegravir than TDF/FTC through 48 weeks (16.4% versus 6.5%, P = 0.04), but other lipid changes and estimated glomerular filtration rate were similar between study arms. Bone mineral density measured by DXA scan rose about 1% through 48 weeks in the raltegravir arm (P = 0.002) while falling between 0.5% and 1% in the TDF/FTC arm (P = 0 .013). Increasing levels of bone formation and resorption markers indicated higher bone turnover in the TDF/FTC group. Week-16 changes in bone turnover predicted bone mineral density loss at week 48.
The investigators noted that darunavir/ritonavir plus raltegravir yielded an even higher virologic failure rate in the single-arm ACTG 5262 trial [2]. Twenty-eight of 112 previously untreated participants (25%) had virologic failure by week 48. In an analysis adjusted for age and sex, virologic failure was associated with a pretreatment viral load above 100,000 copies and a lower pretreatment CD4 count. In a review of the meeting where these results were first presented, clinical researcher Paul Sax (not an ACTG 5262 investigator) suggested other reasons why this seemingly potent combination may perform poorly in practice: (1) lower darunavir concentrations with raltegravir, (2) selective nonadherence to the second daily raltegravir dose, and (3) a low barrier to resistance with raltegravir, although raltegravir-related mutations did not emerge in all ACTG 5262 failures or any RADAR failures [3]. Failure without resistance mutations often indicates poor adherence.
References
1. Bedimo R, Drechsler H, Cutrell J, Jain M, Tebas P, Maalouf N. RADAR study: week 48 safety and efficacy of raltegravir combined with boosted darunavir compared to tenofovir/emtricitabine combined with boosted darunavir in antiretroviral-naive patients: impact on bone health. 7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur. Abstract WEPE512.
2. Taiwo B, Zheng L, Gallien S, et al. Efficacy of a nucleoside-sparing regimen of darunavir/ritonavir plus raltegravir in treatment-naive HIV-1-infected patients (ACTG A5262). AIDS. 2011;25:2113-2122. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3515052/
3. Sax PE. CROI 2011. Online update. Search for ACTG 5262 Sax CROI 2011.
RADAR study: week 48 safety and efficacy of RAltegravir combined
with boosted DARunavir compared to tenofovir/emtricitabine
combined with boosted darunavir in antiretroviral-naive patients.
Impact on bone health
R. Bedimo1,2, H. Drechsler1,2, J. Cutrell2, M. Jain2, I. Farukhi1,2, R. Castanon1,2, P. Tebas3, N. Maalouf2
1VA North Texas Health Care System, Medicine, Dallas, United States, 2UT Southwestern Medical Center, Medicine, Dallas, United States;; 3University of Pennsylvania, Philadelphia, United States
CONCLUSIONS
- Patients on the NRTI-sparing regimen RAL + DRV/RTV achieved faster virologial suppression but over 48 weeks had more treatment discontinuations and less virological suppression. Our findings are consistent with the ones observed in ACTG 5262 and could be due to the twice a day administration or unknown PK interactions.
- Patients in the TVD arm had smaller increases in total cholesterol.
- Patients taking RAL had lower bone turnover and less bone loss than the ones on tenofovir. However this was not explained by differential changes in inflammatory markers. Nucleoside-sparing regimens might be associated with improved bone health.
- Early Changes in bone turnover at week 16 were predictive of changes in bone density at week48.
|
| |
|
 |
 |
|
|