 |
 |
 |
| |
Efficacy and Safety of Rilpivirine-Based Regimens in Treatment-Experienced HIV-1 Infected Patients: A Prospective Cohort Study
|
| |
| |
Reported by Jules Levin
ICAAC 2013 Sept 10-13 Denver, CO
S. Gazaignes1,3, C. Yang2, C. Gatey1, K. Desseaux1,3, C. Lascoux1, M.Lafaurie1, H. Akpe1, M. Guionie1, W. Rozenbaum1, J-M. Molina1,3.
1Saint-Louis Hospital, Paris, France; 2University of Toronto, Toronto, Canada; 3 University Paris Diderot, Paris, France

ABSTRACT
Background: In treatment-naïve patients, Rilpivirine (RPV) has shown non-inferior antiviral activity versus Efavirenz but data in
treatment-experienced patients are limited. We assessed the efficacy and safety of RPV in treatment-experienced patients switching
to RPV-based regimen.
Methods: All ART-experienced HIV-1 infected patients with plasma HIV1-RNA (VL)<500 cp/ml who switched to RPV-based regimen
were enrolled in this prospective monocentric cohort study. Clinical and laboratory data were collected to assess safety and efficacy.
The primary endpoint was the proportion of patients with virologic success (plasma HIV-RNA <50 cp/ml) at M6 using the FDA
snapshot algorithm.
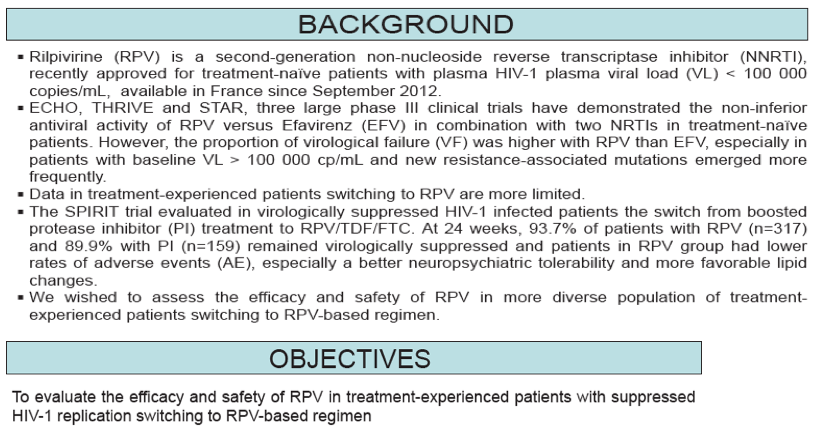
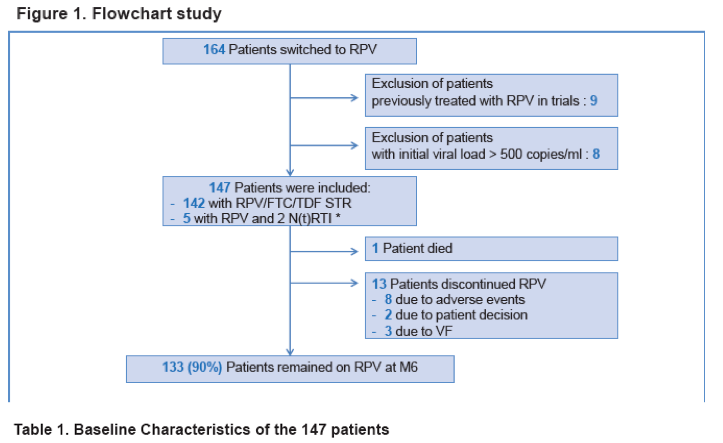
Results: Between Sept. 2012 and Dec. 2012, 147 patients (82% male, median age: 46 yrs, 60% MSM) switched to a RPV-based
regimen.
Patients have received ART for a median of 7 yrs and 88% had baseline plasma HIV-RNA <50 cp/ml. Median baseline CD4
count was 630/mm3.
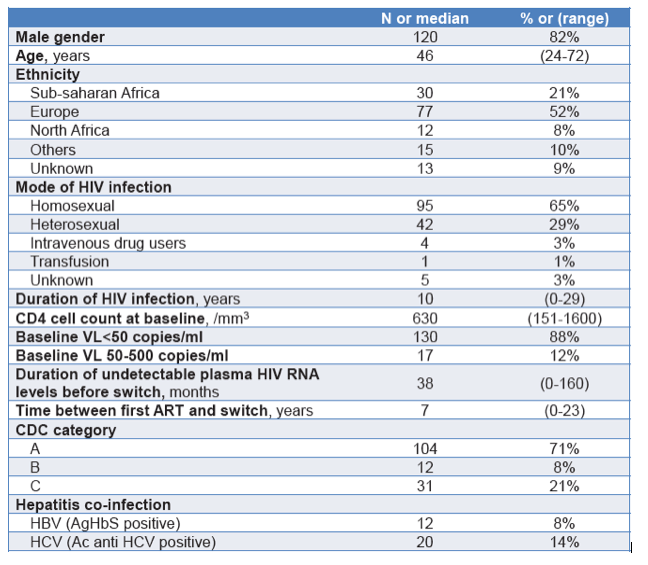
Before the switch, patients received NNRTIs (43%), protease inhibitors (51%) or integrase inhibitors (5%).
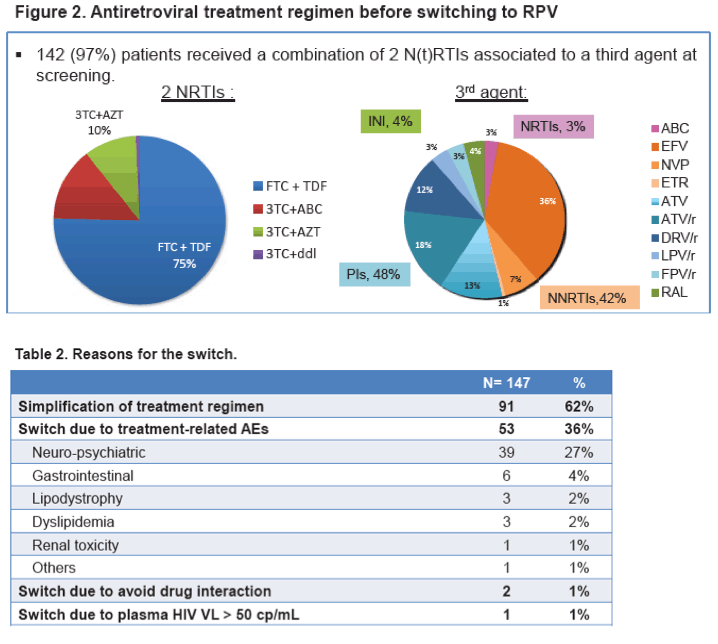
88 (60%) patients switched for simplification, 53 (36%) for adverse events (AE) and 6 (4%) for other reasons.
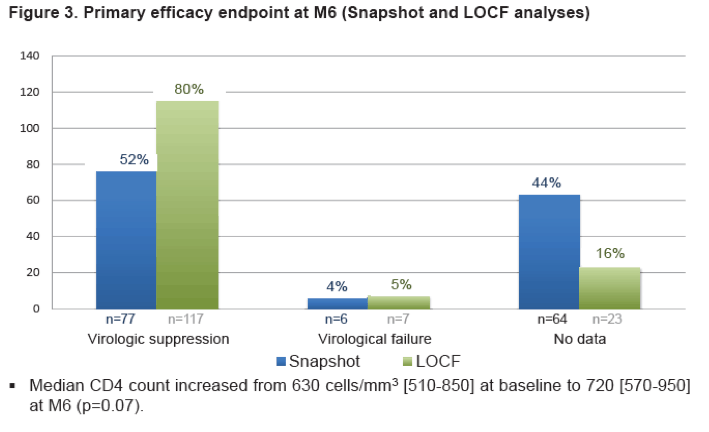
At M6 (database frozen at Jul. 2013) in the snapshot analysis, 77 (52%) patients were suppressed, 6 (4%) had virological failure (plasma HIV-RNA >50 cp/ml) and 64 (44%) had no data. In the LOCF analysis, 117 (80%) were suppressed, 7 (5%) had virological failure and 23 (16%) had no data.
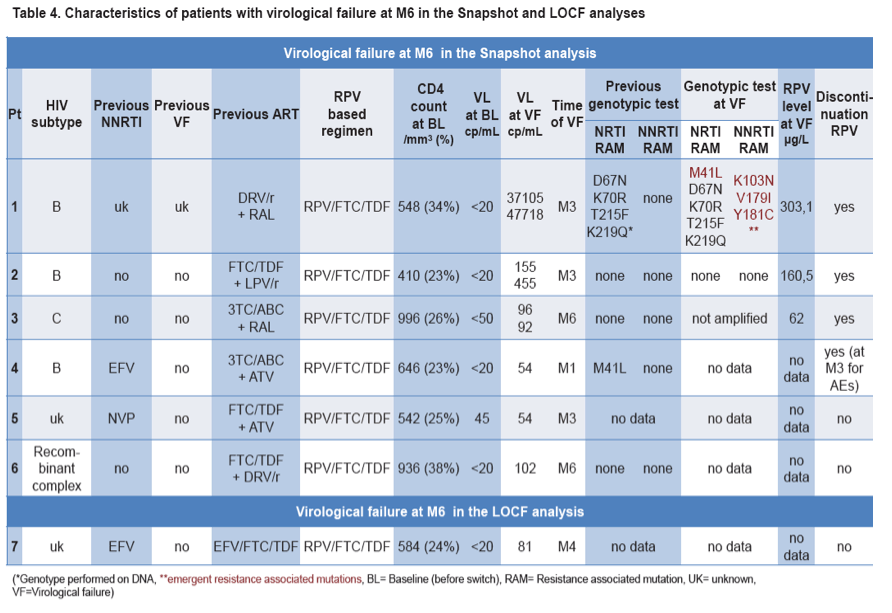
Genotypic resistance tests were performed in 2 of 7 failing patients and yielded emergent NNRTI and NRTI resistant mutations
in 1 patient.
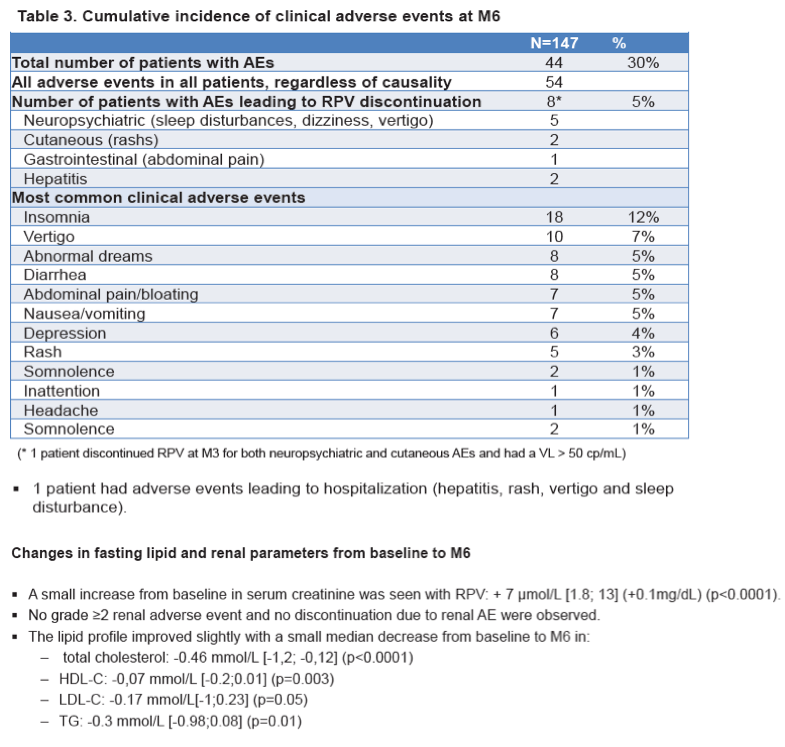
RPV was overall well tolerated and discontinuations due to AEs occurred in 8 cases (5%). The most common grade 2-4 AEs were neuropsychiatric (8%), followed by GI (5%) and rash (3%). Median increase from baseline in plasma creatinine was 7μmol/L (p<0.005).
The lipid profile improved slightly with a median decrease of total cholesterol, HDL, LDL, and TG by 0.46 mmol/L, 0.07 mmol/L, 0.17 mmol/L, and 0.28 mmol/L (p≤0.05) respectively.
Conclusion: Among well suppressed ART-experienced HIV-1 patients switching to RPV, virologic suppression was maintained in a
majority of patients with good tolerability.

|
| |
|
 |
 |
|
|