 |
 |
 |
| |
Entecavir Versus Tenofovir in Treatment-Naive
Chronic Hepatitis B Patients: Third Year Results of a Real World Study
|
| |
| |
Reported by Jules Levin
IDSA Oct 2-6 2013, San Francisco, CA

Program Abstract
Background:
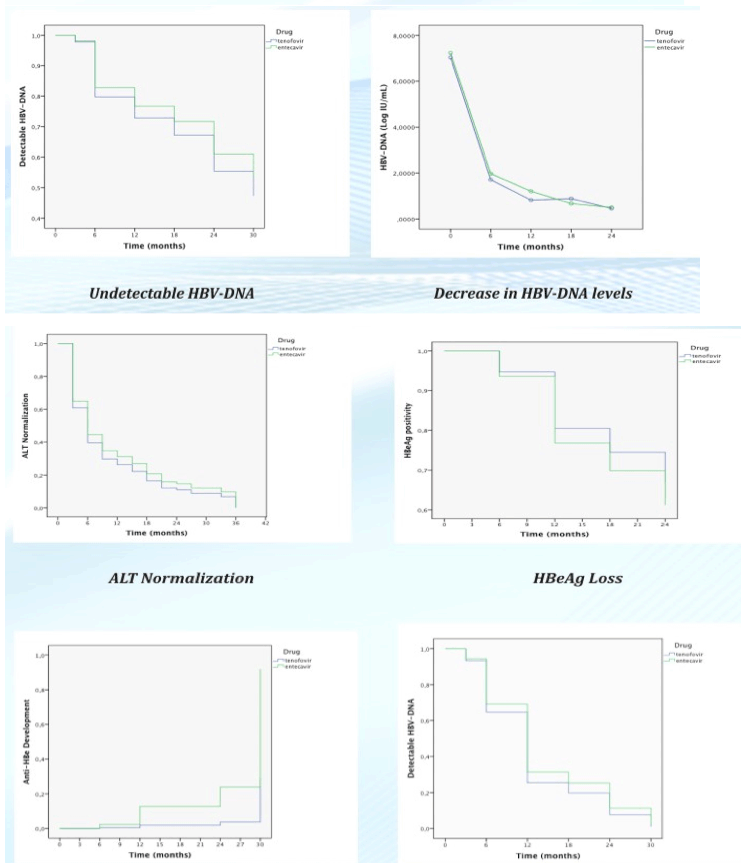
Entecavir and tenofovir are oral antivirals effective in the treatment of chronic hepatitis B with high genetic barriers against resistance. In this study, our aim was to evaluate and compare third year efficacies of these agents in a real-life cohort study.
Methods:
The Realist study is an observational clinical study enrolling treatment-naive chronic hepatitis B (CHB) patients from 11 different centers. Five hundred patients are enrolled in the study since November 2008. In this study demographic data and treatment responses of patients prescribed entecavir or tenofovir in Realist study were compared.
Results:
One hundred thirty four and 130 patients had been prescribed entecavir and tenofovir, respectively. Baseline HBV DNA and ALT levels, fibrosis scores were similar in both groups. 28% and 35% of patients in the entecavir and tenofovir groups had HBeAg positivity respectively and no significant difference was observed between groups. Mean treatment duration was 32±11months in entecavir and 27±13 months in tenofovir group.There was no significant difference between groups in terms of undetectable HBV DNA levels and normal ALT after 3 year of treatment in HBeAg negative and positive CHB patients (p:0.84). HBeAg seroconversion rate after third year seemed to be higher in entecavir group (38% with Entecavir, 20% with Tenofovir)(p:0.04).
Conclusion:
In real-world clinical practice, entecavir and tenofovir provided comparable efficacy. HBeAg seroconversion rates at third year seemed better with entecavir. However it was also observed that HBeAg seroconversion rates were increasing with time in both groups and were approaching to each other.



|
| |
|
 |
 |
|
|