| |
Frailty & aging: African Americans, Hispanics, Caucasians (2 MACS Studies)
|
| |
| |
"the odds of FP conversion increased with older age, non-Hispanic black race/ethnicity, cigarette smoking, hepatitis C infection, depressive symptoms, history of diabetes, and kidney disease, whereas the opposite was true for education to college or beyond" study 2 below
Frailty Rate in HIV+ mid-50 Women Matches Rate in 70-Year-Olds Without HIV.......All women in both groups were Hispanic or African American, and ethnicity rates were similar in the two groups (60% Hispanic, 40% African American).....Bronx-Lebanon Hospital Center and other centers recorded an 11% frailty prevalence in the HIV group, while no women in the HIV-negative comparison group met frailty criteria......Four of the five frailty factors were more prevalent in women with HIV than in the HIV-negative group: weight loss (18% versus 4%), low energy expenditure (3% versus 0%), grip strength weakness (33% versus 23%), and exhaustion (25% versus 7%).......http://www.natap.org/2011/IAS/IAS_22.htm
Frailty, Inflammation and Mortality among Aging HIV-Infected and At-Risk Injection Drug Users......we observed a significant association of frailty with both HIV and mortality risk among aging injection drug users (IDUs).....< high school education....depressive symptoms.....African-American......http://www.natap.org/2014/CROI/croi_61.htm
FRAILTY AND PRE-FRAILTY IN A CONTEMPORARY COHORT OF HIV-INFECTED INDIVIDUALS IN THE SUN STUDY......http://www.natap.org/2012/CROI/croi_41.htm
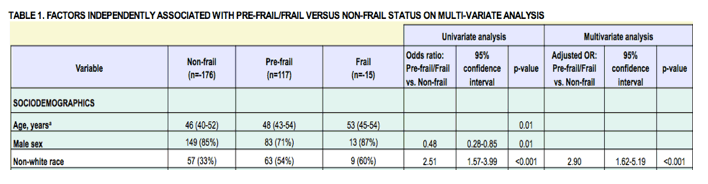
Geriatric Syndromes are Common Among Older HIV-Infected Adults: in this group over age 50, 50% of participants had frailty or pre-frailty ay higher frequencies than HIV-negatives".......http://www.natap.org/2014/CROI/croi_66.htm
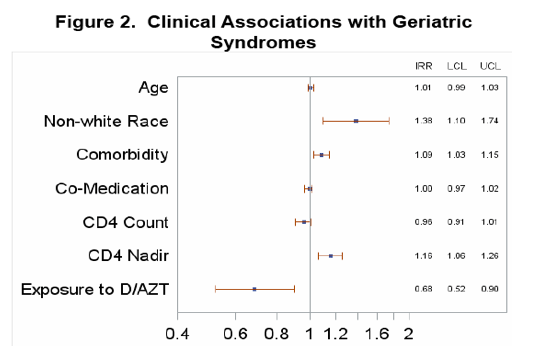
http://www.natap.org/2013/CROI/croi_45.htm
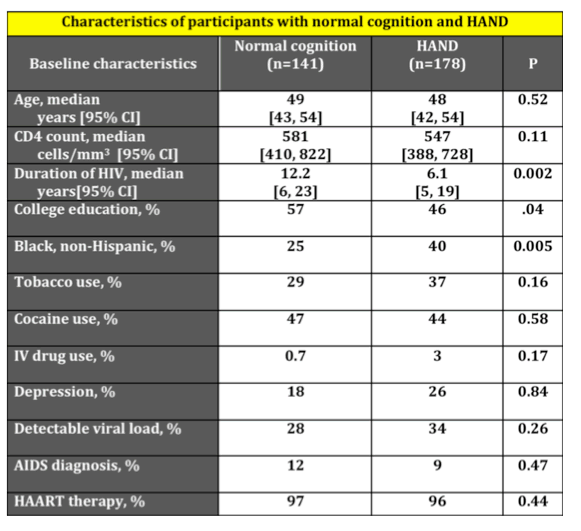
Inflammatory Markers Linked to Frailty in HIV-Positive MACS Men.......http://www.natap.org/2012/AGE/AGE_16.htm
New Research Finds HIV causes rapid aging in key infection-fighting cells: "Successful ART treatment does not fully reconstitute the CD31-CD4+ na�ve T-cell subset"......http://www.natap.org/2011/HIV/012711_01.htm
------------------------------------
MACS Aging studies
In the present study, a low CD4 T-cell count was an independent significant predictor of the FRP in HIV-positive men, adjusting for AIDS, and also in the absence of having a wasting syndrome
Relationship Between a Frailty-Related Phenotype and Progressive Deterioration of the Immune System in HIV-Infected Men: CD4 predicted frailty in MACS, viral load associated.....http://www.natap.org/2012/HIV/101012_05.htm
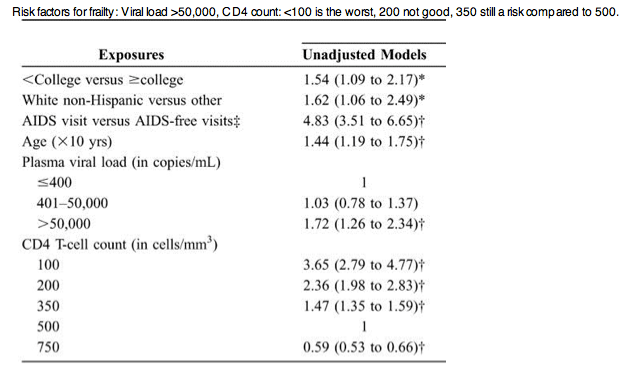
Supported by The Multicenter AIDS Cohort Study......CD4 T-cell count predicted the development of a FRP among HIV-infected men, independent of HAART use. This suggests that compromise of the immune system in HIV-infected individuals contributes to the systemic physiologic dysfunction of frailty....viral load >50,000 associated with frailty
Frailty is a late-stage clinical syndrome that in older adults is hypothesized to result from dysregulation or failure of many physiological systems
Frailty was 7.6% in 1994-1995, 5.2% in 1996-99, and 4.5% in 2000-2005, when approximately 80% of the men were under treatment, almost all of which was HAART. The median number of CD4 T cells increased from 323 to 494 cells per cubic millimeter and the median age from 41 to 48 years......In univariate analysis (unadjusted models), having an AIDS-defining illness was significantly associated with an increased risk of FRP, as were older age, white non-Hispanic ethnicity, and low education (< college) (Table 3).......the odds ratio for the association of FRP with AIDS was still significantly elevated at 4.23 [95% confidence interval (CI), 3.00 to 5.96; P < 0.01]. A lower CD4 T-cell count was highly, and significantly, predictive of onset of FRP at the subsequent visit. Finally, men with a plasma viral load level >50,000 copies per milliliter were more likely to manifest the FRP than men with a plasma viral load level �400 copies per milliliter (P < 0.01), whereas a plasma viral load level of 40-501,000 copies per milliliter was not significantly associated with a higher prevalence of the FRP (P = 0.81)."
-------------------------------
Age, Comorbidities, and AIDS Predict a Frailty Phenotype in Men Who Have Sex With Men (MACS)
Abstract
Background. Adults aging with HIV infection are at risk for age-related comorbidities and syndromes, such as frailty. The objective of this study was to evaluate the expression and predictors of the frailty phenotype (FP) among HIV-infected (HIV+) and HIV-uninfected (HIV-) men who have sex with men.
Methods. A prospective, observational cohort study was nested in the Multicenter AIDS Cohort Study from October 2007-September 2011. FP conversion was defined as the onset of FP over two consecutive study visits. Adjusted odds ratios and 95% confidence intervals ([,]) for FP conversion were estimated using logistic regression models with generalized estimating equations.
Results. Of 10,571 completed study visits from 1,946 men who have sex with men, 12% and 9% were FP+ among HIV+ and HIV- men, respectively (p = .002). The proportion of FP+ visits increased with age regardless of HIV status, but was significantly greater in HIV+ compared to HIV- men aged 50-64 years. Of the 10,276 consecutive visit pairs contributed by participants, 5% (537) were classified as FP conversion, and 45% of the men with FP conversion had only one FP+ study visit. FP conversion was significantly associated with a history of AIDS (adjusted odds ratios = 2.26 [1.50, 3.39], but not with HIV+ alone (adjusted odds ratios = 1.26 [0.98, 1.64]). Among men who had one or more FP+ visits, 34% of HIV+ and 38% of HIV- men had less than two comorbidities.
Conclusions. These findings suggest that expression of the FP can be measured in men who have sex with men with and without HIV infection and reflects multisystem dysfunction in this population; further investigations are needed to better understand clinical utility.
Introduction
The proportion of Americans living with HIV infection (HIV+) who are older than 50 years is increasing, due to both increased survival from effective antiretroviral treatment (1,2) and incident infections in persons aged 50 and older, estimated at 5,500/y or 10.8% of all new infections in the U.S. (3). In the 40 states with confidential name-based reporting, it was estimated that 31% of HIV+ persons were age 50 years or older in 2008 (4). This proportion is expected to exceed 50% by 2015 (5).
Frailty is an aging-related condition characterized by increased vulnerability to stressors, thought to be due to multisystem dysregulation (6-14). Frailty manifests clinically as a decrease in muscle mass, energy, weight, physical strength, and physical activity (7,8,15). The most widely used frailty phenotype (FP) was defined by Fried and colleagues (7), in a population of older (65-101 years), presumably HIV-uninfected (HIV-) adults, and requires the presence of three or more of five criteria: weakness, slowness, unintentional weight loss, low energy, and low physical activity. This FP has been validated as a clinical syndrome in elderly populations, and its presence predicts loss of independence, disability, falls, and death (6,7,16-20).
As more and more HIV+ individuals receiving antiretroviral therapy are developing non-AIDS-defining diseases that have traditionally been associated with aging (21-25), it is important to understand the effects of HIV and of antiretroviral therapy on the physiology and manifestations of aging. To begin to address these questions, the Multicenter AIDS Cohort Study (MACS) defined a frailty-related phenotype (FRP) that approximated the FP using existing data. The FRP was defined as expression of at least three of the following four components: physical shrinking, exhaustion, slowness, and low physical activity level. The FRP differed from the FRP by containing a self-reported measure of slowness, and no measure of weakness (26). Nonetheless, studies using this FRP in the MACS showed: (a) expression of the FRP was positively associated with duration of HIV infection in the absence of highly active antiretroviral therapy (HAART) (26); (b) lower CD4 T-cell counts were independently associated with expression of the FRP after accounting for use of HAART (27); and (c) expression of the FRP prior to HAART initiation independently predicted risk of AIDS or death after HAART initiation (28). However, these studies were limited because they used the FRP, rather than the validated FP, and because they could not fully adjust for the effect of other age-related comorbidities and adverse health conditions such as hepatitis C. Additionally, if frailty is an indicator of vulnerability to stressors in the HIV+ adults, and if HIV+ adults have a greater burden of frailty than HIV- adults, then HIV+ adults would be likely to have a greater burden of negative clinical outcomes as they age. Therefore, to examine the relationship of frailty and aging with HIV in more detail, in 2007 the MACS began assessing all five components of the FP at each study visit for all participants. Here, we report (a) the prevalence of the FP in the MACS, by age and HIV status; (b) predictors of conversion to the FP; and (c) the concordance of the FP with the presence of two or more comorbidities.
Methods
Study Population
The MACS is an ongoing, prospective, interval cohort study of the natural and treated histories of HIV infection. It enrolled 4,954 HIV+ and HIV- men who have sex with men (MSM) in 1984-1985, 668 in 1987-1991, and 1,350 in 2001-2003 in four U.S. cities (Baltimore, Chicago, Los Angeles, and Pittsburgh). Study visits are semiannual and include a standardized interview, physical examination, questionnaires (including the self-administered short form-36 quality-of-life questionnaire (29,30) and items of the Centers for Epidemiologic Studies Depression Scale) (31), and collection of blood for laboratory testing and storage in local and national repositories (32). Additional details of MACS methods have been published (33-35). Study design and questionnaires are available at http://www.statepi.jhsph.edu/macs/macs.html. Informed consent was obtained from all study participants. The institutional review boards at each study site approved this study.
MACS participants were included in this study if they completed at least one study visit between October 1, 2007 and September 30, 2011; analyses of conversion to frailty were restricted to men who completed two study visits during this time. HIV+ men who had never received antiretroviral therapy as of October 1, 2007 were excluded to restrict the study to treatment-experienced men.
Conversion to the FP
The FP defined by Fried and colleagues (7), requires the presence of three of more of the following five components: (a) weakness (present if grip strength measured using a dynamometer is less than 20th percentile of HIV- men); (b) slowness (present if time to walk 4 m is more than 80th percentile of HIV- men); (c) unintentional weight loss (present if participant answers "yes" to the question "Since your last visit, have you had unintentional weight loss of at least 10 pounds?"); (d) exhaustion (present if participant answers "yes" to the question "During the past 4 weeks, as a result of your physical health, have you had difficulty performing your work or other activities (for example, it took extra effort)?"); and (e) low physical activity (present if participant answers "yes, limited a lot" to the question "Does your health now limit you in vigorous activities, such as running, lifting heavy objects, participating in strenuous sports?"). These measures were standardized across study sites and were collected at each MACS study visit beginning October 1, 2007; our study period was from October 1, 2007 to September 30, 2011.
Men who did not meet the criteria for the FP at a given study visit (FP-) but did meet these criteria at their next visit (FP+) were classified as having FP conversion. Two consecutive visits were used as the unit to examine predictors of FP conversion among participants. Each two-visit pair was defined as two consecutive study visits (vi and vi+k), regardless of missed visits (eg, vi and vi+1, vi+1 and vi+2, vi+2 and vi+3 for an individual with no missing study visits, and vi and vi+2, vi+2 and vi+3 for an individual missing visit vi+1). FP converters were compared with individuals without the FP at both visits in the consecutive visit pair.
Predictors of Conversion to the FP
Date of birth, race, and education were self-reported at enrollment into the MACS. Cigarette smoking and injection drug use were all self-reported at every MACS study visit.
Comorbidities were measured at every study visit during the study period.
Hepatitis C infection was defined as detectable hepatitis C RNA in serum; all others were considered uninfected. Depressive symptoms were defined as a Centers for Epidemiologic Studies Depression Scale score greater than 16 (31). High blood pressure was defined as systolic pressure greater than 140 mmHg or diastolic pressure greater than 90 mmHg. Diabetes mellitus was defined as a fasting glucose greater than or equal to 126mg/dL or a self-report of previous clinical diagnosis with use of medication. Dyslipidemia was defined as either a fasting total cholesterol greater than or equal to 200mg/dL, low-density lipoprotein greater than or equal to 130mg/dL, high-density lipoprotein less than 40mg/dL, triglycerides more than or equal to 150mg/dL, or use of lipid-lowering medications with self-report of a previous clinical diagnosis. Kidney disease was defined as an estimated glomerular filtration rate less than 60mL/min/1.73 m2 body surface area using the Modification of Diet in Renal Disease equation (36) or a urine protein-to-creatinine ratio greater than or equal to 200mg protein/1g creatinine. Cancer was defined as diagnosis at, or within a year of, the study visit; 97.4% of cancer diagnoses were medically confirmed.
HIV serostatus was assessed at every visit for HIV- men and positivity determined by a positive ELISA confirmed by Western blot. T-lymphocyte subsets were measured at each MACS center using standardized flow cytometry (37). Plasma HIV RNA concentrations (viral load) were measured using either the standard reverse transcription polymerase chain reaction assay (limit of detection = 400 copies/mL; Roche Diagnostics, Nutley, NJ) or with the Roche ultrasensitive assay (limit of detection = 50 copies/mL; Roche Diagnostics). HAART was defined according to the U.S. Department of Health and Human Services Kaiser Panel guidelines (38) as three or more antiretroviral drugs including either: (a) a protease inhibitor; (b) a nonnucleoside reverse transcriptase inhibitor; (c) an entry or integrase inhibitor; or (d) three nucleoside reverse transcriptase inhibitors, including abacavir or tenofovir. A history of clinical AIDS was defined as the occurrence of a clinical AIDS diagnosis (per the Centers for Disease Control and Prevention's 1993 definition, but excluding a CD4 T-cell count >200 cells/mm3) (39) occuring at or before the time of frailty assessment.
Statistical Analysis
The significance of differences in proportions and medians was determined using the chi-square test and the Kruskal-Wallis test, respectively. For analyses of study visits (as opposed to participants), participants could contribute data from multiple study visits; statistical comparisons were made using generalized linear models with generalized estimating equations that included an independent working correlation matrix to take into account repeated measures from individuals (log link with binomial variance was specified for differences in categorical variables). The intraclass correlation coefficient was used to determine the proportion of the variability that was attributable to differences between study participants (as opposed to the variability within an individual in a repeated measures setting).
Predictors of conversion to the FP were measured at all study visits during the study period; predictors measured at the first visit of each consecutive visit pair were used in the models, with the exception of race, education, and study center, which were measured once at the time of enrollment into the MACS. Predictors with face validity and/or statistically significant univariate associations were included in multivariate models. Logistic regression models with generalized estimating equations (40) were used to estimate the odds of FP conversion for the selected predictors.
The concordance between FP+ status and having two or more comorbidities was determined by estimating the proportion of men with FP+ status at the first visit who had two or more comorbidities (7).
A p value less than .05 guided statistical interpretation, and all analyses were completed using SAS version 9.2 (SAS Institute, Cary, NC).
Results
The study population consisted of 1,946 men (898 HIV+ and using HAART and 1,048 HIV-) who completed at least one study visit between October 1, 2007 and September 30, 2011. These men contributed 10,571 visits (4,842 from HIV+ men and 5,729 from HIV- men). The median number of visits per participant was six (interquartile range: 4, 7) for both HIV+ and HIV- men (p = .76). Twenty-two percent of HIV+ men and 21% of HIV- men completed all eight possible study visits (p = .29).
Prevalence and Durability of the FP
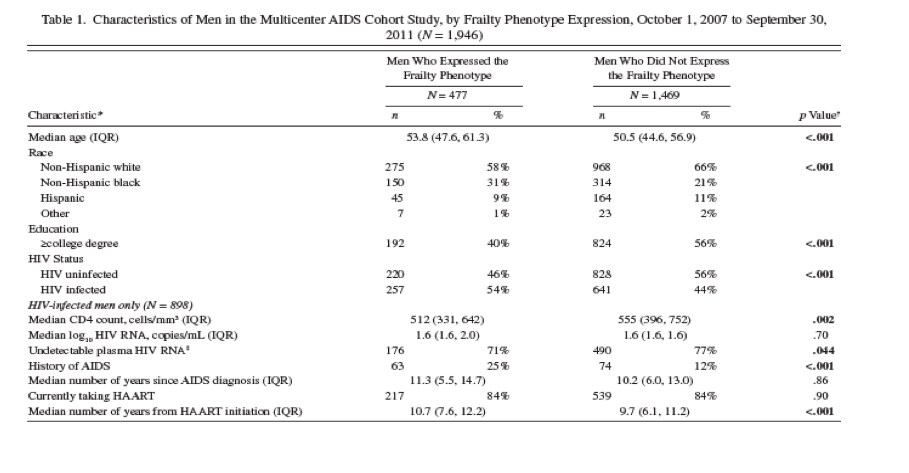
Of the 1,946 men studied, 1,469 (75%) were FP- at all visits contributed, and 477 (25%) were FP+ at one or more visits (Table 1). The latter were older and more likely to be HIV+ or non-Hispanic black, and of lower educational attainment (Table 1). The HIV+ men were more likely to have at least one FP+ visit than the HIV- men (29% [257/898] vs 21% [220/1,048], respectively; p < .001). Overall, 10% (1,116/10,571) of visits were FP+, and this proportion was also significantly higher in HIV+ than HIV- men (12% [601/4,842] vs 9% [515/5,729], respectively; p = .002).There were no significant differences by study site.
Among the 898 HIV+ men, those who were FP+ during at least one visit had a slightly lower median CD4 T-cell count, were slightly less likely to have a suppressed viral load, were twice as likely to have a history of AIDS, and had been receiving HAART for a year longer than those with no FP+ visits; all of these differences were statistically significant. In addition to having the same median number of completed study visits (median = 6, interquartile range: 4, 7), HIV+ and HIV- men also had similar proportions of FP+ study visits before September 2010 that were followed by one or more completed study visit before September 2011 (92% [384/417] and 90% [328/365], respectively; p = .29). The median number of completed study visits among men with zero FP+ study visits and at least one FP+ study visit was six (interquartile range: 4, 7). Thus, expression of the FP was not associated with differential follow-up by HIV status or FP status.
As expected, the proportion of FP+ visits increased with increasing age (Figure 1). This proportion was greater in HIV+ men for all age groups except for 45-49 years, and these differences were statistically significant for men aged 50-65 years. The intraclass correlation coefficients were 0.491 for HIV+ men and 0.579 for HIV- men, suggesting that a substantial proportion of the variability in expression of the FP was within men, similar to the level between men.
Among the 477 men who were ever FP+, 216 (45%) were FP+ at only one visit. Because the vast majority of these men completed six or more visits, as mentioned earlier, it can be inferred that FP+ expression was often transient (see Supplementary Appendix A). Of these 477 men, 293 (61%) had at least one subsequent FP- visit, 85 (18%) had only FP+ visits, and 99 (21%) were FP+ only at their last visit. Of the 10,276 consecutive visit pairs contributed by HIV+ and HIV- men, 5% (537) were classified as conversion and 4% (423) had an FP+ visit immediately followed by an FP- visit (ie, FP reversion); 5% (535) were FP+ at both visits, and 85% (8,772) were FP- at both visits.
To better understand the fluctuations in FP status and the higher prevalence of FP+ visits among HIV+ compared with HIV- men, predictors of conversion to the FP were examined.
Factors Related to Conversion to the FP (FP- to FP+)
This analysis was based on 9,309 (8,772 FP-, FP- and 537 FP-, FP+) consecutive visit pairs from 778 HIV+ and 935 HIV- men. Univariate and multivariate results are shown in Table 2. Overall, HIV infection was associated with a 46% increase in the odds of conversion in univariate models (odds ratio = 1.46 [1.18, 1.81]); HIV infection with and without AIDS was significantly associated with conversion (odds ratios relative to HIV- men = 2.56 [1.75, 3.75] and 1.31 [1.05, 1.64], respectively). In multivariate models, the association of HIV and AIDS status with conversion remained elevated but only HIV infection with AIDS was significant. Additionally, the odds of FP conversion increased with older age, non-Hispanic black race/ethnicity, cigarette smoking, hepatitis C infection, depressive symptoms, history of diabetes, and kidney disease, whereas the opposite was true for education to college or beyond (Table 2).
Multivariate models restricted to HIV+ men included 4,171 consecutive visit pairs (Table 2). Age, lower educational attainment, history of AIDS, depressive symptoms, diabetes, and kidney disease increased the odds of conversion; however, current and nadir CD4 T-cell counts did not. A test of interaction of age and HIV status (with and without AIDS) on FP+ study visits was not statistically significant (p = .91), suggesting that the relationship of age with conversion was not significantly different by HIV status.
Predictors of reversion from the FP (FP+ to FP- in consecutive study visits) were also examined and can be found in Supplementary Appendix B.
Relationship Between Comorbidities and the FP
To better understand the relationship between comorbidities and HIV and AIDS status, we examined the prevalence of hepatitis C and age-related comorbidities measured at the first visit during the study period (N = 1,496). HIV+ men were more likely to have hepatitis C infection, depression, diabetes, and kidney disease and less likely to have high blood pressure, than HIV- men (Table 3). Dyslipidemia was the most common comorbidity in both HIV+ and HIV- men. HIV+ men also had a higher proportion with two or more comorbidities. Among men who were FP+ at their first study visit, 66% (62/94) of HIV+ men and 62% (48/78) of HIV- men had two or more comorbidities; these proportions did not differ dramatically in other study visits.
Discussion
To our knowledge, this study is the first to evaluate the prevalence and expression of the FP defined and validated by Fried and colleagues (6,7), in HIV+ and similar HIV- MSM. Prevalence of the FP was higher in HIV+ compared with HIV- men, particularly in those aged 50 and older. We found substantial fluctuation in FP+ status in our study population. Examining factors associated with FP conversion allowed for consideration of this fluctuation. Concordance of FP+ status and comorbidities was high, but a sizable proportion of HIV+ and HIV- men were FP+ with fewer than two comorbidities, suggesting that FP+ and comorbidities are not synonymous.
The FP+ prevalence of 12% among visits from HIV+ men in this study is similar to cross-sectional estimates in some studies, but not all. Other reported prevalences include 9% in a cohort with well-controlled HIV infection (41), 11% among HIV+ women in New York City (42), 5% in HIV+ women in the Women's Interagency HIV Study (43), 12.3% among injection drug users in the AIDS Link to the Intravenous Experience Study (44), and 19% in HIV+ adults in South Africa (45). All of these studies assessed the same FP used in this study, but differences in these study populations by age, sex, HIV transmission risk, underlying comorbidities, and severity and treatment of HIV disease likely account for the different prevalences of the FP. In the Cardiovascular Health Study, the population of presumably HIV-, aged 65 and older, community-dwelling adults in which the FP was developed, the prevalence of the FP was 7% (7). It is intriguing that the prevalence of the FP in much younger HIV- men in this study was higher (9%). Possible reasons for this unexpectedly high prevalence could include different lifestyles and exposures, or misclassification due to lack of validation of the FP phenotype in younger MSM. Further studies will be needed to address whether the definition of the FP needs to be modified for this population.
The prevalence of the FP increased with age in both HIV+ and HIV- men, as expected, but was higher in HIV+ men aged 50-64 years compared with HIV- men of the same age. These data should not be interpreted as evidence of "accelerated" aging (46) for several reasons. First, this study did not define age at onset of the FP, only prevalence. Second, rates of comorbidities associated with expression of the FP were higher in HIV+ than HIV- men, as discussed later.
Third, there was substantial fluctuation in expression of the FP, and it may not be valid to equate expression of the FP at just one study visit with the presence of frailty (or aging in general) in this population. Additional studies are needed to identify potential modifications to the FP to capture incident frailty events in this younger population of HIV+ and HIV- adults.
Conversion from FP- to FP+ status was significantly associated with the presence of conditions known to be associated with expression of the FP in elderly HIV- people (eg, depressive symptoms, diabetes, kidney disease, hepatitis C infection, and cigarette smoking). The fact that many factors and organ systems were associated with conversion suggests that expression of the FP is indicative of multisystem dysfunction in our study population, as it is in the older HIV- population. HIV infection, independent of these comorbidities, was significant only in the presence of a history of AIDS, suggesting that men who had AIDS before the introduction of HAART may have driven the association of HIV infection with expression of the FP. Median time from AIDS diagnosis did not differ, however, among HIV+ men with and without at least one FP+ visit. We did not see an independent association between lower CD4 T-cell count and FP+ status, as reported in other studies (27,43); this may be due to (a) collinearity due to the inclusion of comorbidities in multivariate models, as this association was present in univariate models; and/or (b) the limited number of men with a very low CD4 T-cell count in this highly treated cohort. Due to this high level of treatment, the effect of HIV infection cannot be separated from that of HAART. Finally, having attained a college degree or beyond was significantly associated with a lower odds of frailty conversion, similar to cross-sectional studies of HIV+ adults in which frailty was associated with lower education attainment (44), unemployment (41), and homelessness (44). It is important to note that some of our null findings may be due to collinearity in the multivariate models, such as no difference in the odds of frailty conversion by race/ethnicity with the inclusion of education attained and no difference by nadir CD4 T-cell count with the inclusion of a history of clinical AIDS.
The previous cross-sectional studies of the FP in HIV+ populations could not assess fluctuation in FP status. One such study did reassess a small subset (7/100) of their study population 6 months after expressing the FP; one was still FP+, four were FP-, and two had died (47). In the present study, almost half of the men who expressed the FP did so at only one study visit over 4 years. The proportion of consecutive visit pairs in which reversion occurred was 4%. In a 4.5 year study of HIV- elderly people, the 18-month reversion rate ranged from 13%-24% in three 18-month periods (48). In the Women's Health and Aging study, the rate ranged from 35%-85% in three 18-month periods (49). Finally, in a longitudinal study of the FP in HIV-infected and HIV-uninfected injection drug users, fluctuation in the FP status was not assessed (44). It is difficult to compare these rates across studies because of the differences in outcome, study design, and sample size; most notably, our outcome was based on consecutive visit pairs, not individuals, and the studies among the elderly people had 18-month follow-up periods compared with our 6-month intervals. A recent qualitative study that aimed to create a framework for disability (an outcome linked to frailty in HIV-, older populations) in HIV+ adults described the disability experience as "episodic in nature" (50).
There was a greater prevalence of comorbidities (hepatitis C, depression, diabetes, and kidney disease) in HIV+ compared with HIV- men. Most men who were FP+ had two or more comorbidities at first visit in this study (66% of HIV+ and 62% of HIV- men). The proportions of men with the FP but less than two comorbidities (34% for HIV+ and 38% for HIV- men) were similar to that in the study population in which the FP was first described (30%) (7). These data suggest that comorbidities are potential etiologic agents for frailty, as hypothesized by Fried and colleagues (7), and also that the FP and comorbidities are not synonymous in either the present, younger HIV+ or the older HIV- population. An alternative interpretation is that the severity of comorbidities and unmeasured comorbidities not accounted for in this study could be contributing to the increased prevalence of FP expression. Further studies are needed to determine the relationship between the FP and comorbidities more precisely and whether comorbidities are etiologic agents of the FP in this younger, HIV+ population.
There are important limitations to this study. First, the FP has not been validated as a measure in HIV+ individuals, or in individuals aged 65 and younger. However, the FRP based on four of the five criteria used to define the FP was shown to predict AIDS-free survival in the MACS after initiation of HAART (28), and the FP predicted death in a cohort of HIV-infected and HIV-uninfected injection drug users (44), suggesting that the FP is likely to predict clinical outcomes in HIV+ people. Second, the MACS includes only men, so the present findings may not be generalizable to women. However, previous studies showed similar correlates of the FP in male-only (27) and female-only (43) populations, and expression of the FP predicted mortality in both HIV+ and HIV- people in a cohort of injection drug users that includes both men and women (44). Third, the variability in FP status and the brevity of the study period (4 years) complicate the interpretation of the FP in HIV+ adults and limit our ability to make causal inference regarding the relationship of HIV and FP. Despite these limitations, the MACS cohort is well suited to this question, due to the presence of an HIV- group that is similar to the HIV+ men (ie, the appropriate comparison group in which to study the effects of chronic HIV infection) and the large proportion of HIV+ men receiving HAART with suppression of HIV replication to undetectable levels, as would be needed for long-term effects of HIV infection to become a possible health problem.
This study has contributed to a better understanding of the FP in treated HIV+ MSM by demonstrating measurement of the FP in a younger and HIV-infected population, quantifying the prevalence of and the fluctuation in FP+ status in HIV-infected MSM, determining the predictors of conversion to the FP+ status, and showing the degree of concordance of FP+ status and comorbidities. Nevertheless, our understanding of frailty in HIV+ adults is in its infancy. Modifications to the definition of frailty in younger, HIV+ adults are likely to be needed, such as requiring two consecutive study visits in which the FP is present. Though the clinical importance of the FP in elderly, HIV-uninfected adults is well established (6), its importance in HIV+ populations has not been well studied; however, evidence of its importance continues to emerge (44). Outcomes associated with the FP and mechanisms of frailty in HIV+ adults must be identified and articulated so that interventions can be developed to prevent it or lessen its impact. Ongoing follow-up of MSM in the MACS will continue to contribute to answering these questions and to understanding of the aging process among those living with HIV infection.
|
|
| |
| |
|
|
|