| |
Hep C patients to get NHS access to three new therapies....with or without cirrhosis
|
| |
| |
Daily News | November 26, 2015
Links to NICE Guidance on each of the 3 therapies below
http://www.pharmatimes.com/Article/15-11-26/Hep_C_patients_to_get_NHS_access_to_three_new_therapies.aspx
If you have genotype 1, 3 or 4 chronic hepatitis C that fits the criteria in tables 1, 2 or 3, and your healthcare team think that daclatasvir is the right treatment, you should be able to have the treatment on the NHS.
Patients with hepatitis C living in England and Wales could get 'routine' access to three new treatments - Gilead's Harvoni, AbbVie's Viekirax and Bristol-Myers Squibb's Daklinza - after cost-regulators approved their use on the National Health Service.
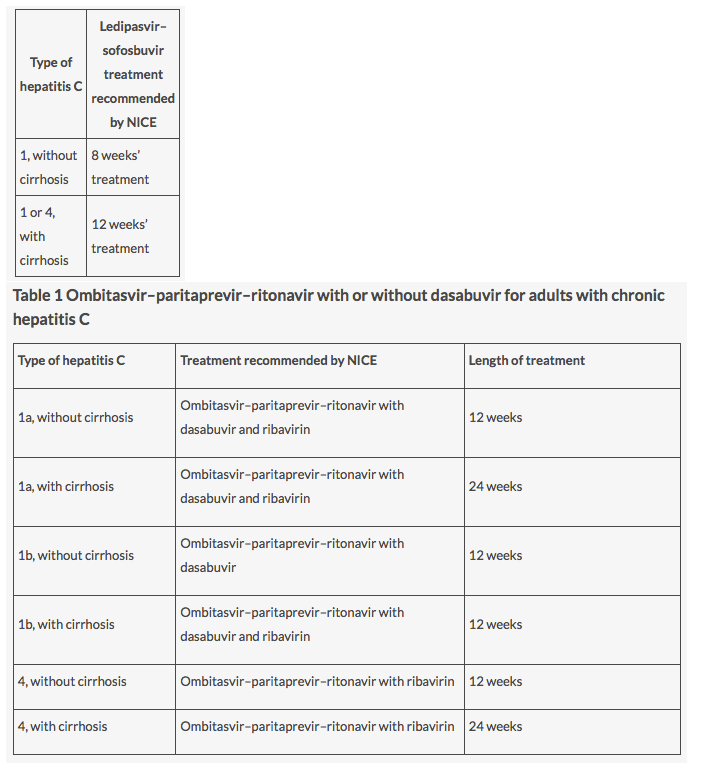
NICE is recommending Harvoni (ledipasvir-sofosbuvir) in various different scenarios for patients with hepatitis C genotypes 1 and 4. Compared with current treatment, Harvoni offers an oral, shortened, and interferon-free therapy and represents "a major development in the clinical management of chronic hepatitis C," the Institute noted......NICE technology appraisal guidance [TA363] Published date: November 2015: http://www.nice.org.uk/guidance/ta363....
Harvoni was approved in Europe in November on the back of Phase III data showing that is achieved cure rates - sustained virologic response - of 94%-99%, at a cost of around Ł39,000 for a 12-week course and Ł78,000 for a 24-week one (excluding VAT and ribavirin).
Viekirax (ombitasvir, paritaprevir, ritonavir) has also been approved for NHS use, with or without Exviera (dasabuvir), as a possible treatment for adults with genotypes 1 and 4. In a pooled analysis of three Phase III trials involving 1,083 subjects with HCV genotype 1, 97% achieved a sustained virologic response......NICE technology appraisal guidance [TA365] Published date: November 2015: http://www.nice.org.uk/guidance/ta365
What does this mean for me? If you have genotype 1 or 4 chronic hepatitis C, and your healthcare team think that ombitasvir-paritaprevir-ritonavir with or without dasabuvir is the right treatment, you should be able to have the treatment on the NHS. The treatment should be available on the NHS within 3 months of the guidance being issued.
Daklinza (daclatasvir) can be considered an option in patients with HCV genotypes 1,3 or 4. The drug is the first NS5A complex inhibitor to win clearance in the EU, and also offers patients the chance of a shorter treatment duration (12 or 24 weeks) compared to 48 weeks of treatment with interferon- and ribavirin-based regimens. Its cost is around Ł24,000 and Ł49,000 depending on the length of treatment......NICE technology appraisal guidance [TA364] Published date: November 2015: http://www.nice.org.uk/guidance/ta364
According to BMS, the drug, when used in combination with Gilead's Sovaldi (sofosbuvir), provides an all-oral, interferon-free regimen with cure rates of up to 100% in clinical trials, including patients with advanced liver disease, genotype 3, and those who have previously failed treatment with protease inhibitors.
NICE has recommended that the decision to treat and what to prescribe are made by multidisciplinary teams in the operational delivery networks put in place by NHS England, "to prioritise treatment for people with the highest unmet clinical need".
|
|
| |
| |
|
|
|