 |
 |
 |
| |
Anti-PCSK9 Monoclonal Antibodies: The Future Lipid-Lowering Agent of Choice?
|
| |
| |
http://www.acc.org/latest-in-cardiology/articles/2015/07/06/12/16/anti-pcsk9-monoclonal-antibodies
Jul 07, 2015
There is an established link between elevated circulating low-density lipoprotein cholesterol (LDL-C) levels and the atherosclerotic burden associated with cardiovascular disease (CVD).1,2 While statin-based LDL-C lowering has been a cornerstone of secondary prevention of CVD,3,4 many individuals fail to adequately lower their LDL-C levels, either due to insufficient response or intolerance to statin therapy. Statin-related adverse effects have been reported in approximately 5-10% of patients,5 and it is estimated that approximately 10-20% of patients on high-dose statins perceive muscle-related adverse effects.6 While ezetimibe also reduces cardiovascular events after an acute coronary syndrome,7 the effects of ezetimibe on LDL-C (~20% reduction)8 and clinical events (6.4% relative risk reduction) are modest in subjects who already have a baseline LDL-C of ~70 mg/dL.9 Thus, more potent novel lipid-lowering agents could play an important role as supplemental medications in patients who fail to reach their target LDL-C level despite optimal statin therapy +/- ezetimibe.
Normally, circulating LDL-C is removed once it binds the LDL receptor (LDLR) on hepatic cells.10 Proprotein convertase subtilisin/kexin type 9 (PCSK9) is a member of the subtilisin family of serine proteases that binds to the LDLR promoting LDLR degradation, thereby resulting in less LDL sequestration and increased levels of circulatory LDL-C.10 The targeted inhibition of PCSK9 would promote LDL catabolism and, thus, allow for a reduction of LDL-C to levels below those commonly achieved to date.
PCSK9 inhibition, in the form of anti-PCSK 9 antibodies, such as alirocumab (SAR236553/REGN727), evolocumab (AMG145), and bococizumab (PF04950615/RN316), represents a novel approach to LDL-C lowering. As antibodies, these drugs must be delivered parenterally, although small-molecule oral PCSK9 inhibitors are in early developmental stages.
There are a number of clinical trials with PCSK9 monoclonal antibodies demonstrating their excellent efficacy and short-term safety profile in a variety of patients, including those with elevated cardiovascular risk, patients with heterozygous or homozygous familial hyperlipidemia, and statin-intolerant patients. In phase 2 trials (Table 1 below), the LDL-C reductions versus placebo have ranged from 50-70% with the top doses of PCSK9 inhibitors.11 In addition, statistically significant reductions in total cholesterol (26-38%), apolipoprotein B (ApoB) (34-52%), triglycerides (12-26%), and lipoprotein(a) (15-31%) have been reported.12 There appears to be little (2-8%), if any, increase in HDL-C.12 Longer-term follow-up up to two years demonstrates persistence in these effects without attrition over time, notably in the Open-Label Study of Long-Term Evaluation Against LDL Cholesterol (OSLER)13 and the ODYSSEY LONG TERM trial.14
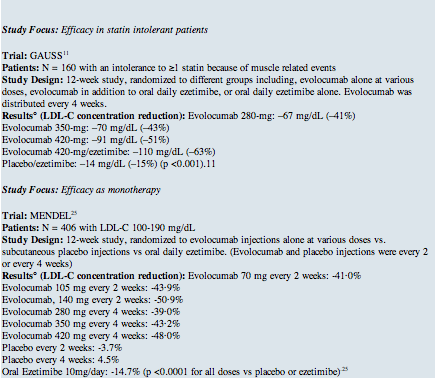
The aforementioned phase 2 trials have been reinforced by phase 3 trials (Table 2 below). The efficacy of evolocumab as a monotherapeutic agent was demonstrated in the Monoclonal Antibody Against PCSK9 to Reduce Elevated LDL-C in Subjects Currently Not Receiving Drug Therapy for Easing Lipid Levels-2 (MENDEL-2) trial, a phase 3, randomized, controlled clinical trial. Among 614 patients, monotherapy with evolocumab reduced LDL-C from baseline, on average, by 55% to 57% more than placebo and 38% to 40% more than ezetimibe (p <0.001 for all comparisons).15 Similar results of a phase 3, randomized, double-blinded trial with alirocoumab demonstrated a ≥50% LDL-C reduction in the majority of patients who received 24 weeks of alirocumab (75 mg every two weeks) versus ezetimibe therapy alone.16 Statin-intolerant patients in the Goal Achievement After Utilizing an Anti-PCSK9 Antibody in Statin Intolerant Subjects -2 (GAUSS-2) study, a phase 3, randomized, placebo-controlled clinical trial studying 370 patients, showed a reduction of LDL-C from baseline by 53% to 56% with evolocumab, corresponding to treatment differences versus ezetimibe of 37% to 39% (p <0.001).17
A longer phase 3 trial of evolocumab over the course of 52 weeks randomized 901 individuals with an LDL-C of at least 75 mg/dL, despite previous treatment with various agents. Each of the doses of evolocumab achieved a significant and sustained reduction in LDL-C compared to placebo, with an overall mean reduction of 57.0 ± 2.1% (p <0.001) at 52 weeks.18 Similarly, the OSLER trial, a 52-week, phase 3, open-label extension study that enrolled patients from four previous phase 2 parent studies of evolocumab, randomized patients to either evolocumab 420 mg every four weeks with standard of care (SOC) (evolocumab + SOC, n = 736) or SOC alone (n = 368).13 Patients who received evolocumab during the OSLER trial had a mean reduction in LDL-C of 52% at week 52 (P <0.0001), whether they had received placebo or evolocumab during the preceding 12-week parent study.13 On the other hand, patients randomized to SOC only (and discontinued evolocumab on entry into the OSLER trial) had a return of LDL-C levels to baseline levels prior to initial entry into the parent study,13 illustrating the importance of maintaining patients on a steady regimen in order to sustain optimal LDL-C levels.
There are four large phase 3 outcomes studies currently ongoing with three different PCSK9 inhibitors on top of background statin therapy. The Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk (FOURIER) trial is a randomized, double-blinded, placebo-controlled study that is following 27,500 patients treated with statins who have prior MI, ischemic stroke, or peripheral arterial disease for approximately three to four years to evaluate the impact of LDL-C reduction on major cardiovascular events with evolocumab versus placebo. The ODYSSEY OUTCOMES trial is a randomized, double-blinded, placebo-controlled study in 18,000 patients comparing the effect of monoclonal antibody alirocumab with placebo on the occurrence of cardiovascular events in patients who have experienced an acute coronary syndrome (ACS) event four to 52 weeks prior to randomization and are treated with evidence-based medical and dietary management of dyslipidemia.14
The Evaluation of Bococizumab (PF-04950615;RN316) in Reducing the Occurrence of Major Cardiovascular Events in High Risk Subjects (SPIRE-1) trial is an ongoing study in 17,000 patients evaluating the PCSK9 inhibitor, bococizumab compared to placebo, in reducing the occurrence of major cardiovascular events, including cardiovascular death, myocardial infarction, stroke, and unstable angina requiring urgent revascularization, in high-risk subjects on background lipid-lowering therapy with an LDL-C of 70-100 mg/dL or non-HDL-C of 100-130 mg/dL. The SPIRE-2 trial has a similar design but will enroll 9,000 high-risk patients with an LDL-C ≥100 mg/dL or non-HDL-C ≥130 mg/dL.
The efficacy and early safety of PCSK9 inhibitors via the use of monoclonal antibodies to achieve lower LDL-C concentrations appears promising to date. However, the future of these agents as a potentially widely used medication depends on the outcomes of the ongoing phase 3 trials currently underway and their ability to lower CVD risk. Nonetheless, the excitement surrounding anti-PCSK9 antibodies as a potential breakthrough agent is warranted as it may potentially change the paradigm surrounding the treatment and management of patients for years to come.

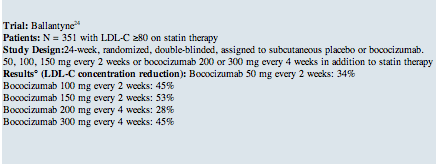









- See more at: http://www.acc.org/latest-in-cardiology/articles/2015/07/06/12/16/anti-pcsk9-monoclonal-antibodies#sthash.tLRDsxtg.dpuf
|
| |
|
 |
 |
|
|