 |
 |
 |
| |
Three-Day per Week Atripla Maintains Viral Suppression and Decreases Sub-clinical Toxicity: a Pilot Study
Three-Times-a-Week Atripla Maintains Viral Suppression in 24-Week Pilot Study
|
| |
| |
18th International Workshop on Comorbidities and Adverse Drug Reactions in HIV, September 12-13, 2016, New York
Mark Mascolini
Among people with at least 2 years of viral suppression on coformulated efavirenz/tenofovir/emtricitabine (Atripla), everyone who switched to 3-times-a-week Atripla and everyone who continued daily Atripla maintained a viral load below 37 copies for 24 weeks [1]. All 61 participants kept an undetectable viral load for 24 to 30 months after the original maintenance-therapy group switched to 3 Atripla tablets per week.
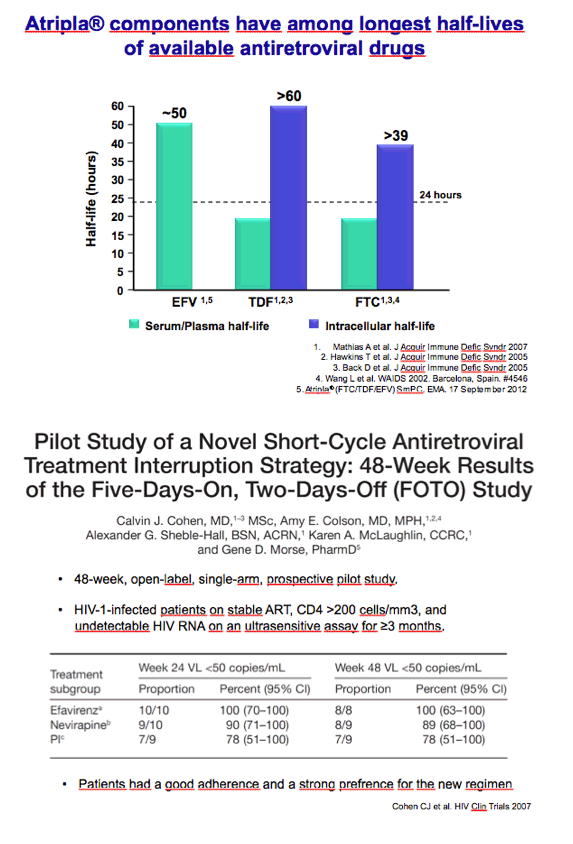
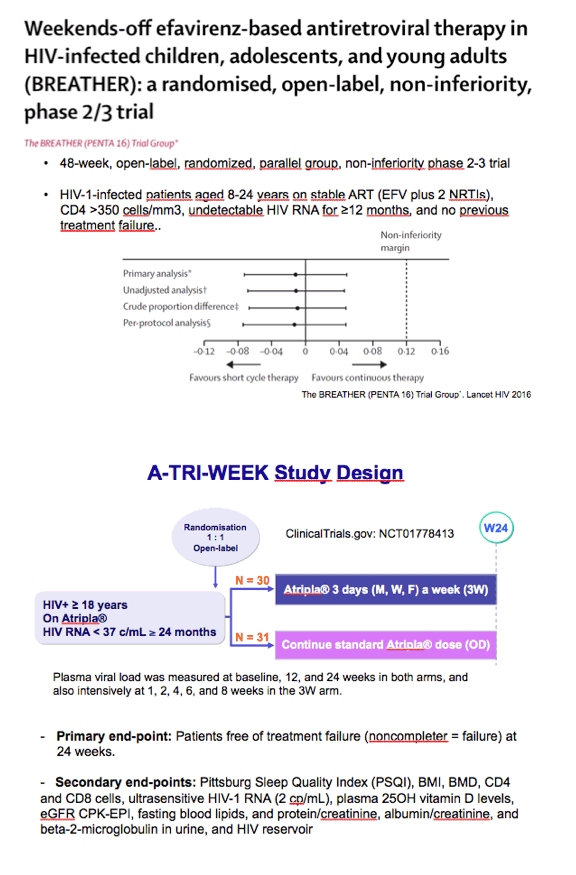
Two previous trials found encouraging results with 5-days-a-week regimens [2,3], and one trial recorded a 96% viral suppression rate through 48 weeks with 4-days-a-week therapy [4]. One of the 5-days-a-week trial tested efavirenz-based regimens in children, adolescents, and young adults [3]. Esteban Martinez and colleagues at the University of Barcelona Clinic pushed the envelope further, testing Atripla 3 times weekly in already-suppressed patients.
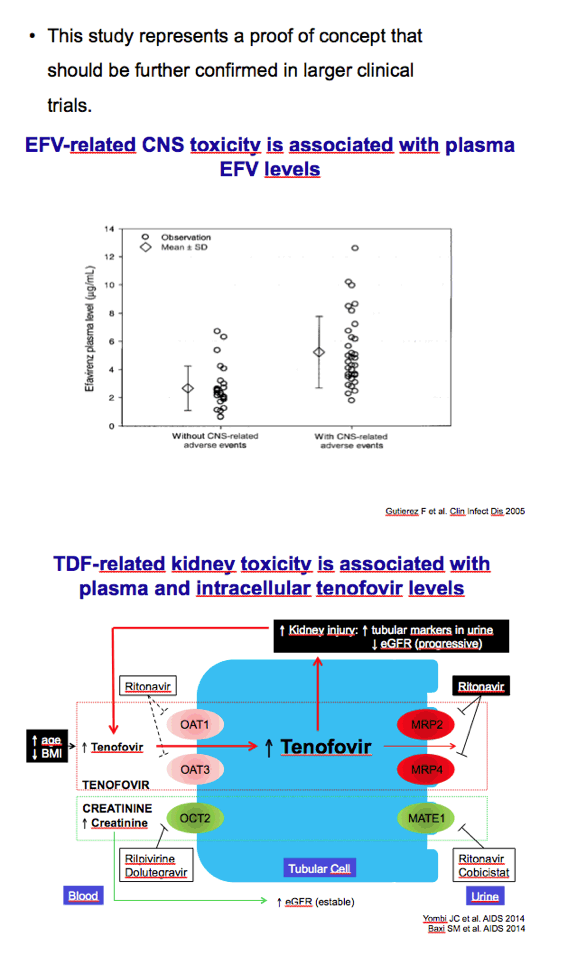
Efavirenz-based regimens have been nudged from the top spot among preferred first-line regimens by integrase inhibitors and ritonavir-boosted darunavir, but one-pill once-a-day Atripla remains a popular combination backed by voluminous data. The neurocognitive side effects of efavirenz and the renal and bone toxicity of tenofovir disoproxil fumarate (TDF) are well-appreciated limitations. Martinez noted that Atripla is a good candidate for less than daily dosing because efavirenz, TDF, and emtricitabine all have long half-lives.
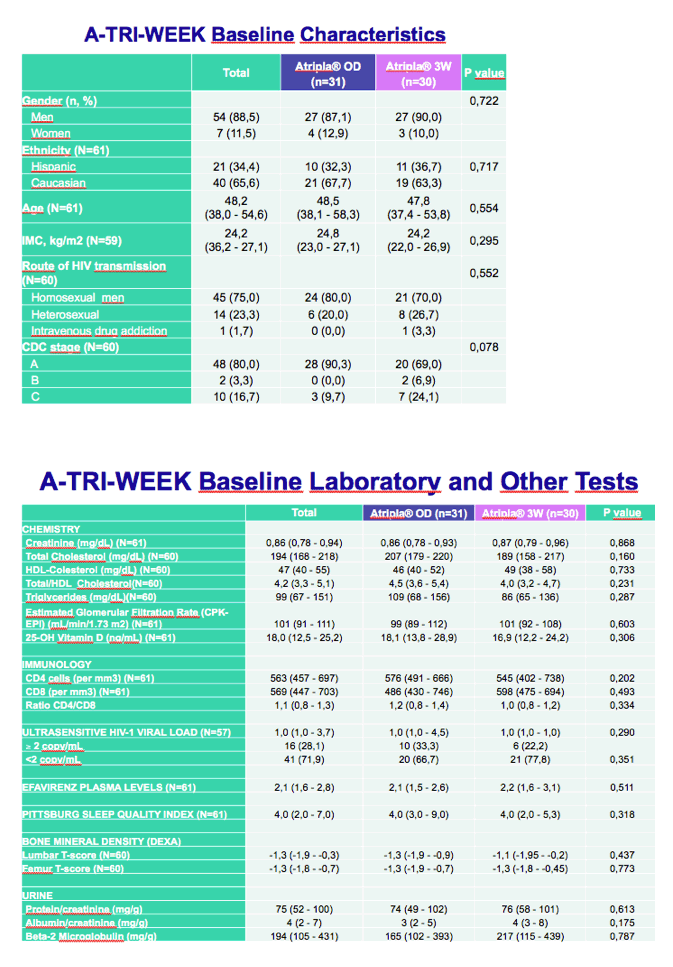
The pilot trial involved 61 adults who maintained a viral load below 37 copies for at least 2 years with Atripla. The Barcelona team randomized 31 participants to stay with once-daily dosing and 30 to start taking Atripla 3 days a week, on Monday, Wednesday, and Friday (the 3W arm). The investigators measured plasma viral loads in everyone at 12 and 24 weeks and intensively in the 3W arm at weeks 1, 2, 4, 6, and 8. The primary endpoint was the proportion of people who kept their viral load below 37 copies at week 24 in a noncompleter-equals-failure analysis.
Proportions of men in the once-daily and 3W arms were 87% and 90%, three quarters of all participants were men who have sex with men, and median age was similar in the two groups (48.2 years overall). Baseline CD4 counts and lab values did not differ significantly between study arms.
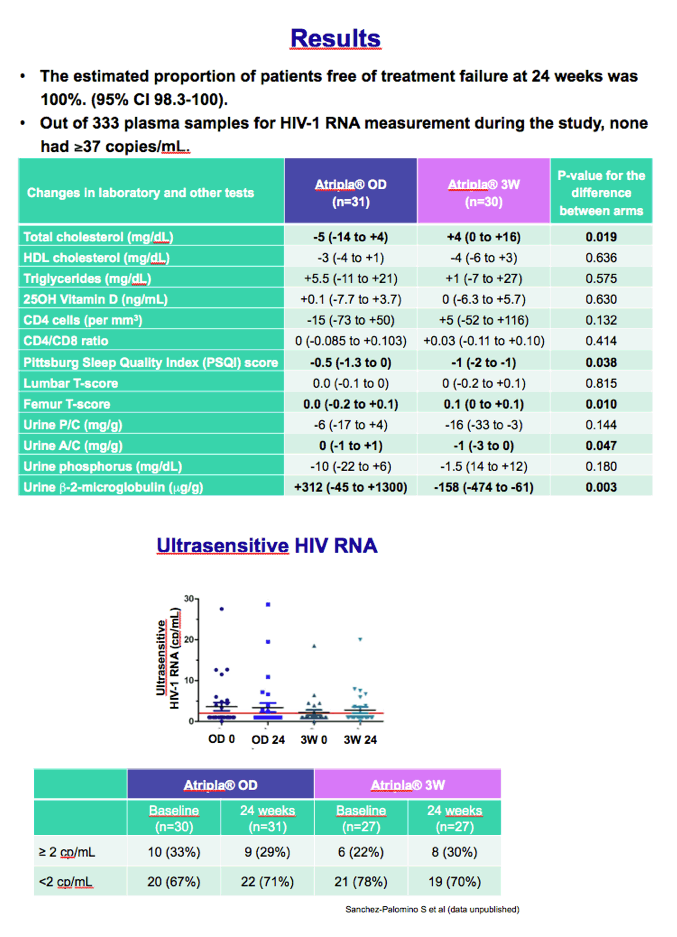
Twenty-four weeks after randomization, everyone in both groups had maintained a viral load below 37 copies (95% confidence interval 98.3 to 100). Among 333 plasma samples assessed during the study, none had HIV RNA above 37 copies. An ultrasensitive assay determined that about 70% of participants in each study arm had a viral load below 2 copies at week 24.
Four lab values at week 24 differed significantly between the once-daily arm and the 3W arm: total cholesterol (-5 versus +4 mg/dL, P = 0.019), Pittsburg sleep quality index (-0.5 versus -1, P = 0.038), femur bone density T score (0 versus 0.1, P = 0.047), and urine beta-2 microglobulin (+312 versus -158 ug/g, P = 0 .003).
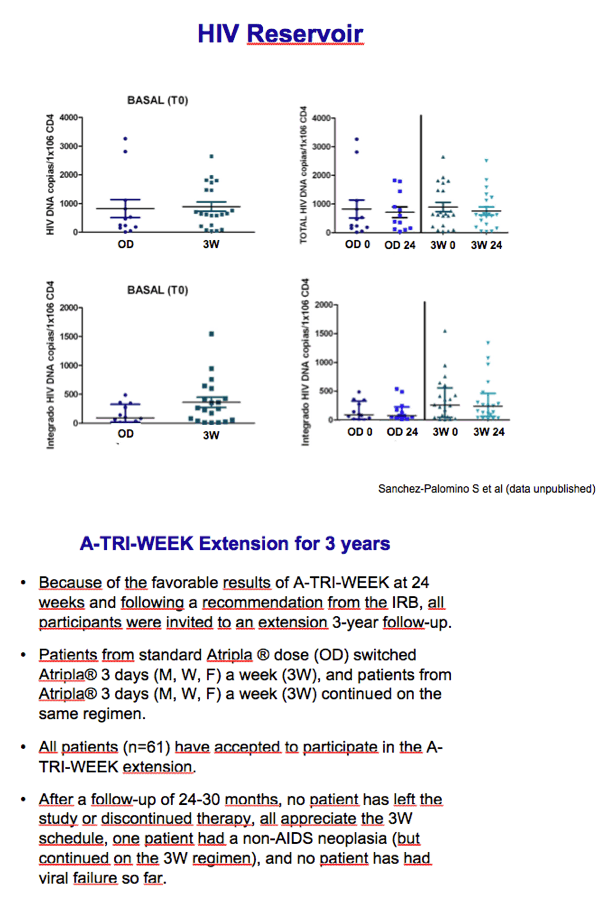
On the basis of these results, the researchers invited the once-daily group to start taking Atripla 3 times a week and asked everyone to continue follow-up for 3 years. All 61 study participants agreed to the trial extension. After 24 to 30 months of follow-up, everyone stayed on treatment and all kept their viral load below 37 copies. A non-AIDS cancer developed in one person, who continued the 3W regimen.
The investigators proposed that 3-times-a-week Atripla "is a feasible option that maintains efficacy and improves subclinical toxicity parameters." They called for larger trials to confirm their finding. The researchers reported no adherence measures. An unanswered question with all less-than-daily oral regimens is whether such dosing is harder to remember than daily dosing
References
1. Rojas J, Blanco JL, Sanchez S, et al. Three-day per week Atripla maintains viral suppression and decreases subclinical toxicity: a pilot study. 18th International Workshop on Comorbidities and Adverse Drug Reactions in HIV, September 12-13, 2016, New York. Abstract 022.
2. Cohen CJ, Colson AE, Sheble-Hall AG, McLaughlin KA, Morse GD, Pilot study of a novel short-cycle antiretroviral treatment interruption strategy: 48-week results of the Five-Days-On, Two-Days-Off (FOTO) study. HIV Clin Trials. 2007;8:19-23. http://www.tandfonline.com/doi/abs/10.1310/hct0801-19
3. Butler K, Inshaw J, Bernays S, et al. BREATHER (PENTA 16) short-cycle therapy (SCT) (5 days on/2 days off) in young people with chronic human immunodeficiency virus infection: an open, randomised, parallel-group phase II/III trial. Health Technol Assess. 2016;20:1-108. http://www.journalslibrary.nihr.ac.uk/hta/volume-20/issue-49#abstract
4. de Truchis P, Assoumou L, Landman R, et al. Efficacy of a maintenance four-days-a-week regimen, the ANRS162 4D trial. International AIDS Conference. July 18-22, 2016. Durban. Abstract THPEB 063. http://programme.aids2016.org/PAGMaterial/eposters/0_5947.pdf


|
| |
|
 |
 |
|
|