| |
Aging - New drugs
|
| |
| |
Download the PDF here
Download the PDF here
Download the PDF here
Can aging be 'drugged'?
"aging can be manipulated......commonly used drugs have been found to delay aging and increase lifespan in animal models, raising the idea that we might already have an elixir for longevity in hand"
"Chang et al.5 first tested a panel of small-molecule compounds known to target pathways that were predicted to be important for the maintenance of senescence in vitro......This revealed that the small-molecule compound ABT263, which is a specific inhibitor of the apoptosis genes BCL2 and BCL2L1 (encoding BCL-2 and BCL-xL, respectively), selectively induced apoptosis of senescent cells in culture in a cell type- and species-independent (mouse as well as human)."
----------------------
Clearance of senescent cells by ABT263 rejuvenates aged hematopoietic stem cells in mice
Nature Medicine
Published online 14 December 2015
Senescent cells (SCs) accumulate with age and after genotoxic stress, such as total-body irradiation (TBI)1, 2, 3, 4, 5, 6. Clearance of SCs in a progeroid mouse model using a transgenic approach delays several age-associated disorders7, suggesting that SCs play a causative role in certain age-related pathologies. Thus, a 'senolytic' pharmacological agent that can selectively kill SCs holds promise for rejuvenating tissue stem cells and extending health span. To test this idea, we screened a collection of compounds and identified ABT263 (a specific inhibitor of the anti-apoptotic proteins BCL-2 and BCL-xL) as a potent senolytic drug. We show that ABT263 selectively kills SCs in culture in a cell type- and species-independent manner by inducing apoptosis. Oral administration of ABT263 to either sublethally irradiated or normally aged mice effectively depleted SCs, including senescent bone marrow hematopoietic stem cells (HSCs) and senescent muscle stem cells (MuSCs). Notably, this depletion mitigated TBI-induced premature aging of the hematopoietic system and rejuvenated the aged HSCs and MuSCs in normally aged mice. Our results demonstrate that selective clearance of SCs by a pharmacological agent is beneficial in part through its rejuvenation of aged tissue stem cells. Thus, senolytic drugs may represent a new class of radiation mitigators and anti-aging agents.
-----------------
Can aging be 'drugged'?
Nature Medicine Published online
08 December 2015
"the modulation of genetic pathways to greatly increase longevity in model organisms has brought the realization that aging can be manipulated. Furthermore, simple, commonly used drugs have been found to delay aging and increase lifespan in animal models, raising the idea that we might already have an elixir for longevity in hand, albeit with unknown mechanisms. However, the formulation or identification of a pharmaceutical designed specifically to combat aging with fewer side effects and better potency than today's compounds is alluring and tempting. With the hurdles of clinical development for an anti-aging compound coupled with personalized gerontology and predictive, relevant biomarkers, the bar for a 'geraceutical' could be too high for a single entity to clear, but the future healthcare costs of our increasing aging population are too great to not try."
Abstract
The engines that drive the complex process of aging are being identified by model-organism research, thereby providing potential targets and rationale for drug studies. Several studies of small molecules have already been completed in animal models with the hope of finding an elixir for aging, with a few compounds showing early promise. What lessons can we learn from drugs currently being tested, and which pitfalls can we avoid in our search for a therapeutic for aging? Finally, we must also ask whether an elixir for aging would be applicable to everyone, or whether we age differently, thus potentially shortening lifespan in some individuals.
As a positive outcome of medical progress and the modern lifestyle, life is generally now prolonged in developed countries. The number of centenarians keeps rising worldwide, and since the mid-1970s a new category of centenarians has appeared: the supercentenarians, who are individuals over the age of 110 (www.grg.org). However, a downside of this increased longevity is the rise in the incidence of age-associated diseases, such as cardiovascular and metabolic disorders, cancer and neurodegenerative disorders1. By 2025, it is anticipated that more than 20% of Europeans will be 65 or older (Public health report, European Commission; http://ec.europa.eu/health/ageing/policy/index_en.htm). Similarly, in the US, it is predicted that Americans aged 65 or older will number nearly 89 million in 2050, more than doubling the number of older adults in the US in 2010 (Centers for Disease Control and Prevention (CDC); http://www.cdc.gov/aging/index.html). The aging population will have profound effects on public health, social services, and welfare systems.
Encouragingly, governments' awareness of this soon-to-be reality is rising, as reported recently (http://www.whitehouseconferenceonaging.gov/), and pilot initiatives to improve healthy aging and social care in Europe are being established (http://ec.europa.eu/research/innovation-union/index_en.cfm/section=active-healthy-ageing). Although the implementation of health and social measures is necessary to improve the quality of life of older adults, the immediate challenge for medical gerontology is to identify pharmacological strategies to promote healthy aging 'healthspan', with the idealistic goal of postponing the onset of geriatric syndromes, without necessarily extending lifespan itself.
Aging is considered to be the biggest risk factor for developing fatal chronic diseases, including cardiovascular, cerebrovascular and neurodegenerative diseases; metabolic syndromes; and most prevalent forms of cancer2.
Conceptually, aging is viewed as a general decline in cell and tissue function that is associated with an increase in low-grade inflammation and a deficiency in the adaptive immune response, thus increasing the susceptibility to disease, and ultimately leading to tissue failure and death3. The development of one chronic disease of aging is generally not an isolated event; there is a high incidence of chronic diseases and co-morbidities in the elderly. Therefore, it is crucial to define the molecular events required to boost the body's natural defenses, such as by identifying coping mechanisms that target the threats posed to an organism's cells and tissues.
We present here a broad overview of the progress achieved in animal research to identify drugs that extend lifespan and healthy aging, as well as the current understanding of the mode of action of these drugs. We also argue that the lessons learned in animal models provide an important cornerstone of the process of targeting human aging, but we also raise concerns about therapies that will work for all, as the heterogeneity of human aging remains a largely unexplored area of research.
Existing pharmacology
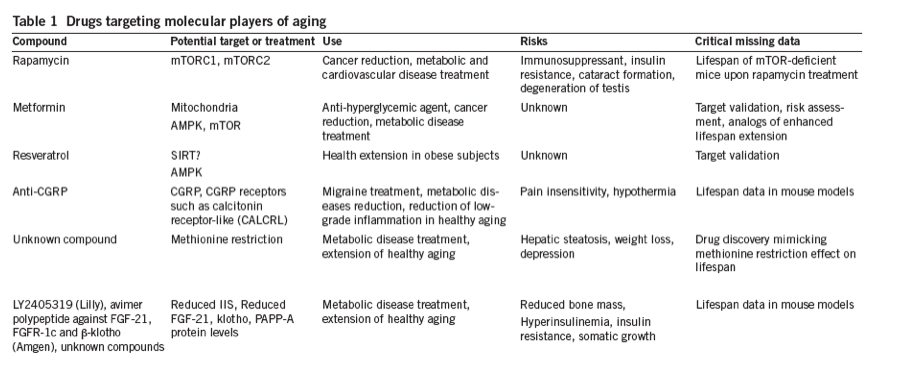
Multiple drugs have elicited major interest in aging research through their ability to activate signaling networks involved in aging (Table 1). An initiative from the US National Institute of Aging (Intervention Testing Program, ITP) tests compounds that may hold the promise of altering lifespan in mice4. We discuss some of the current therapies under investigation below.
Rapamycin. Rapamycin was identified by the ITP program to be a successful candidate for preventing age-related disease, as it is able to delay cancer formation in aged mice and extend their lifespan5, 6. This US Food and Drug Administration (FDA)-approved drug, which was discovered in a soil bacterium on Easter Island, has long been used as an immune modulator for organ transplantation and kidney cancer. Male and female mice fed rapamycin in middle or late in life show an increase in median and maximum lifespan by 10-18% as a result of inhibition of the mammalian target of rapamycin (mTOR) complex5, 6. Rapamycin was also shown to delay the rate of aging in female mice, by slowing metabolic decline, cancer formation and cardiovascular dysfunction7, 8, 9. An ongoing study is currently being undertaken in short-lived marmoset monkeys10, and it is hoped that this study will enlighten whether rapamycin can also be beneficial in non-human primates.
The method by which rapamycin regulates the rate of aging is complex, as short-term treatment results in immunodeficiency and favors glucose intolerance and insulin resistance11, 12. However, prolonged treatment results in improved metabolic profiles, increased oxygen consumption and ketogenesis, markedly enhanced insulin sensitivity, and improved immune response in mice13, 14. Beneficial effects on immunosenescence have also been observed in the elderly when treated for six weeks with a rapamycin analog (RAD001) (ref. 15). The metabolic side effects associated with rapamycin treatment are probably due to its action on mTOR complex 2 (mTORC2) in the liver11, 13. Importantly, rapamycin's anti-aging effects are associated with mTORC1 inhibition, and they are also observed upon the genetic inhibition of this complex and its downstream effectors16, 17, 18. In metazoans, TORC1 controls growth-related processes that influence the rate of aging, such as ribosome biogenesis, protein synthesis, transcription, nutrient uptake and autophagy in response to nutrients, growth factors and cellular energy status19. Thus, the design of a more-selective mTORC1 inhibitor may hold promise as a powerful human rejuvenating factor with reduced side effects compared to those of rapamycin. Because high doses of rapamycin suppress immune function and negatively affect metabolic health, it is unknown whether rapamycin's beneficial effects can be translated to humans, especially with prolonged exposure.
Metformin. Metformin, a biguanide isolated from the French lilac, is a clinically approved drug commonly prescribed as an anti-hyperglycemic agent in the treatment of type 2 diabetes20, 21, and it has also shown potential as an anti-aging remedy. Long-term treatment with low-dose metformin starting in middle age promotes healthy aging and longevity in male mice by 4-6%, although a higher dose shortens longevity22. Metformin improves global metabolic fitness similarly to dietary restriction in aged mice, resulting in improved endurance, insulin sensitivity, reduced oxidative damage and chronic inflammation22. Metformin decreases mitochondrial respiration through mitochondrial complex I inhibition23, a phenomenon that limits the ability of cancer cells to cope with energetic stress, thus providing a rationale for the powerful cancer reduction properties of this anti-diabetic drug24.
Reduction of ATP synthesis affects AMP-activated protein kinase (AMPK) activity; AMPK is an enzyme that is indirectly stimulated by metformin exposure25. Under low-energy conditions, AMPK acts as a master metabolic switch to inhibit anabolic processes and activate catabolic pathways that produce energy, such as cellular glucose uptake, fatty acid oxidation and respiration26, 27. AMPK activation is beneficial for longevity in worms28, 29, 30, but mammalian studies have not shown direct evidence of AMPK's role in aging. AMPK activation is positively correlated with improved insulin sensitivity in mice31 and the AMPK agonist 5-aminoimidazole-4-carboxamide riboside (AICAR) improves cognitive and motor function in young and aged mice32. Although metformin induces AMPK activation, its action on longevity and cancer might also depend on mTORC1 inhibition in an AMPK-independent manner22, 33. Despite the controversy regarding the molecular targets that underlie metformin's action, there is general agreement that low-dose metformin improves metabolic health, reduces tumorigenesis and positively affects longevity. Metformin's action on human aging will be investigated by the Targeting/Taming Aging With Metformin (TAME) study in an effort to encourage the FDA to view the aging process as an indication that can be targeted with better medications34. However, as outlined below, it must be recognized that longevity treatments that are mostly regarded as benign in isolated settings can have negative effects on health and lifespan when they are exposed to healthy, heterogeneous populations.
Resveratrol. Another molecule that has attracted much attention in anti-aging research is resveratrol, a polyphenol that is found in some fruit and in red wine. The positive link between resveratrol and lifespan has been established in organisms from yeast to mammals35, 36, 37. Resveratrol treatment prolongs the lifespan of mice fed a high-fat diet or fed every other day36, but not that of mice on a regular chow diet6. Interestingly, resveratrol treatment in non-human primates showed some beneficial effects on metabolic health, mostly conferring protection in models of stress- and age-associated diseases, including chronic overfeeding, insulin resistance, type 2 diabetes, and cardiovascular dysfunction38, 39, 40, 41, 42. In humans, resveratrol had a protective effect against human coronary artery endothelial cell damage43, and treatment for a month improved metabolic rates and muscle function in obese individuals44. Resveratrol was proposed to act on the oxidized nicotinamide adenine dinucleotide (NAD+)-dependent deacetylase SIRT1, but the connections between SIRT1 and longevity45 and between resveratrol and the activation of SIRT1 (refs. 46,47,48) have recently been challenged. For example, the beneficial effects of resveratrol have also been attributed to the activation of AMPK49, but the bona fide target(s) of resveratrol has yet to be defined.
Aspirin, NDGA and CGRP. Another promising route to treating age-associated damage is by dampening inflammatory signaling. Accumulation of low-grade inflammation is a hallmark of aging50, and increased levels of multiple inflammatory cytokines including tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), and acute-phase proteins such as C-reactive protein (CRP), may underlie the activation of pathological senescence51, 52. Lifelong administration of the anti-inflammatory agents aspirin (acetylsalicylic acid) or nordihydroguaiaretic acid (NDGA; also called masoprocol) results in an 8-12% median lifespan extension without increasing maximum lifespan in male mice, with no effect on female mouse lifespan53. Aspirin and NDGA also have anti-oxidant properties that could account for their beneficial effects on longevity. Aspirin, which is the most widely used drug in the world, has a variety of positive effects on immune, metabolic and cardiovascular health in mouse models54, 55. However, the potential of aspirin as a healthy aging agent is limited by its antithrombotic and anti-platelet aggregation effects, which, coupled with gastrointestinal irritation, can lead to a high risk of bleeding. Other salicylates with decreased bleeding risk improve glycemia and enhance glucose-stimulated insulin secretion in normal and diabetic patients56, 57, but without counterbalancing body weight gain in diabetes58. Aspirin's hypoglycemic properties are linked with inflammatory responses through the inhibition of nuclear factor of kappa light-chain enhancer of activated B cells (NF-κB)55, 59, a canonical pathway that was recently linked to lifespan extension in mice60. The precise targets of aspirin and NDGA in lifespan regulation are unknown.
Alternatively, targeting circulating factors that have a role in immune and cardiovascular decline with age may offer a promising route to altering aging and health. Notably, the vasodilator calcitonin gene-related peptide (CGRP), levels of which rise in circulation with age in rodent models61, 62, is naturally absent in the naked mole rat, a rodent of exceptional longevity63, 64. Our data highlight a role for CGRP as a critical neuroendocrine regulator of longevity in mice and a biomarker for healthspan62. Genetic inhibition of pain-sensitive transient receptor potential cation channel, subfamily V, member 1 (TRPV1) channels, which regulate the secretion of CGRP, preserves metabolic health in mice and extends the longevity of male and female mice, thereby reducing cancer formation and enhancing cognitive performance at old age. Pharmacological inhibition using a peptide antagonist against CGRP receptors improves metabolic health in aged mice, thus offering a potential therapeutic means to restore health in late-life62. Monoclonal antibodies specific to CGRP or its receptors are in phase 2 clinical trials against migraine65, and they may hold the potential to combat age-associated diseases, assuming that prolonged reduction of CGRP has few side effects in the elderly.
Insulin/IGF-1 signaling. Genetic mouse models of decreased insulin/insulin-like growth factor (IGF) signaling (IIS) extends lifespan in mice, and this increased lifespan is observed in mutants of the somatotropic axis that have a reduction of growth hormone (GH) and/or a reduction of IGF-1 compared to wild-type mice. Modulation of downstream IGF-1 signaling components extends both healthspan and lifespan in mice66, 67, 68, 69, 70, 71, 72, 73. Similarly, spontaneous mutations that result in GH deficiency result in dwarf mice of exceptional longevity that show decreased IGF-1 and fasted insulin levels74, 75, 76, 77, 78.
How could this pathway be manipulated therapeutically to enhance lifespan and healthspan? One promising solution is to use the natural activators and inhibitors of IIS to design drug therapies. As of now, three secreted proteins have been successfully found to extend lifespan through potential modulatory activity on IGF-1 (refs. 79,80).
Overexpression of the klotho protein (or a-klotho), a transmembrane protein that may undergo cleavage to release a bioactive hormonal signal that inhibits IGF-1, extends mouse longevity by 20-31% in males and 19% in females75. Despite their remarkable longevity, these mice display insulin resistance and hyperinsulinemia, presumably due to klotho inhibition of common IGF-1 and insulin signaling components. What is lacking at the moment is a survey of the physiological parameters associated with the increased longevity of these mice. Mice that lack klotho show accelerated aging (or progeria), infertility, arteriosclerosis, skin atrophy, osteoporosis and neurodegeneration-like phenotypes81, 82, suggesting that klotho overexpression might improve many parameters of healthy aging despite negatively affecting metabolic health. However, it is unclear whether klotho's only role is to inhibit insulin signaling, as it has been proposed to interact with Wnt signaling and multiple membrane-bound receptors.
Another inhibitor of IGF-1 signaling is fibroblast growth factor 21 (FGF-21), a liver hormone that is secreted during fasting, which modulates hepatic fatty acid oxidation and ketogenesis, thereby increasing insulin sensitivity while blocking somatic growth83. FGF-21 signaling requires the co-receptor ß-klotho, suggesting that ß-klotho and FGF-21 might work together to modulate IGF-1 signaling and longevity84. Remarkably, FGF-21 overexpression increases mouse longevity by 36% in both sexes and results in improved metabolic health compared to controls79. The adverse effects of FGF-21 overexpression include reduction in bone mass, female infertility and smaller size, but it is plausible that these side effects are due to overexpression of the Fgf21 transgene from birth, and that they might be avoided if this overexpression is induced exclusively during adulthood.
IGF-1 bioavailability is controlled by the inhibitory IGF binding proteins (IGFBPs), a class of binding partners that sequester IGF-1 (ref. 85). Pregnancy-associated plasma protein-A (PAPP-A) induces the proteolysis of IGFBP-4, and thus enhances IGF-1 action. Inhibition of PAPP-A expression or its proteolytic activity provides a third approach to decreasing IGF-1 availability, without interfering with the downstream IGF-1 signaling machinery. Strikingly, Pappa-knockout mice live 20 to 40% longer than wild-type animals, and they maintain a healthy immune system86, 87 and resistance to the development of atherosclerosis88, but they show no difference in the occurrence of neoplastic disease when compared to wild-type animals87. Similarly to the FGF-21 overexpressing mice, long-lived Pappa-knockout mice have decreased bone mineral density89.
Drugs capable of increasing the bioavailability and/or activity of klotho or FGF-21, or of blocking the proteolytic activity of PAPP-A, represent attractive routes toward the development of therapies that will positively impact select parameters of aging, if the adverse effects on insulin resistance can be bypassed. For example, LY2405319, an FGF-21 variant with enhanced stability, was shown to improve metabolic health in diabetic rhesus monkeys90, and it significantly ameliorated dyslipidemia in obese and diabetic patients, although its glucose-lowering effects were quite modest91. Bispecific avidity multimer (avimer) polypeptides that bind with high affinity and specificity to one of the receptor and co-receptor pairs used by FGF-21, FGFR-1c and ß-klotho, respectively, have been shown to mimic FGF-21 activity and improve body weight and metabolism in obese cynomolgus monkeys92. The consequences of long-term use of either LY2405319 or the avimer-based polypeptides are unknown.
Methionine restriction. Finally, it is also plausible that quite simple dietary changes can improve healthy aging and lifespan. One achievable modification for humans is methionine restriction in the diet, which confers functional benefits on longevity in yeast93, worms94, flies95 and rodents96. Several reports indicate that this diet is sufficient to delay multiple aspects of aging in rodents97, 98. Methionine restriction offers an attractive alternative to pharmaceutical interventions, at it may be achieved mostly through the practice of a vegan diet99. At the molecular level, the restriction of methionine is hypothesized to activate the transsulfuration pathway (TSP), which converts methionine into cysteine. Activation of the TSP pathway is required for dietary restriction-mediated lifespan extension in flies100. The TSP is the cell's primary source of the production of hydrogen sulfide, a gaseous signaling molecule implicated in nervous, vascular and intestinal systems, which underlies many of the protective effects on healthspan acquired during dietary restriction101, 102, 103. However, because methionine is an essential amino acid, health hazards associated with its removal in the diet need to be assessed. Methionine restriction may also cause hepatic steatosis104, severe body weight loss, anemia97 and epigenetic modifications that lead to depression105.
Overcoming the heterogeneity of aging
One reason it is unlikely that we will identify a 'magic bullet' to treat all aspects of aging in all individuals arises from the great inter-individual variability in the pathologies associated with aging across the human population. For example, not all 85-year-olds have Alzheimer's disease, a disease of non-dividing neuronal cells, nor do they have cancer, a disease of dividing cells. Therefore, a clinical trial to combat either of these two indications would need to take into account this heterogeneity. Until we can classify and categorize the early and pathological events of aging in each organ system of the human body, we cannot assume that the human population ages stereotypically.
Strikingly, in mice, recombinant inbred strains show marked variation in their longevity response to dietary restriction, a treatment that is widely recognized as the gold standard to extend lifespan in all species106, 107. Dietary restriction dramatically shortens the lifespan of multiple strains of mice while increasing that of others. Thus, phenotypic variation can greatly affect longevity and the response to drug treatment, and it is essential to understand how variation affects human aging populations and accepted longevity-extending treatments. Therefore, caution must be applied even to trials of drugs such as metformin (as used in the TAME proposed studies), for which the safety profile for prolonged doses in healthy individuals is largely unknown, despite its excellent history in the treatment of individuals with diabetes. Whether widespread exposure to metformin, a drug known to be a mitochondrial uncoupler, across a diverse healthy population would reveal a profile similar to that seen for diet restriction in mice106 is of great concern.
The absence of accessible molecular biomarkers of pathological aging has blocked the categorization of human aging as an indication in the clinic. Nevertheless, recent discoveries have provided promising candidate biomarkers that could be used to test interventions. One important observation is that human longevity and healthy aging show modest heritability therefore offering an opportunity to track genetic markers associated with aging108. Meta-analysis of human genome-wide association studies (GWASs) of aged individuals suffering from chronic disease has revealed single-nucleotide polymorphisms (SNPs) in genes linked to either inflammation or cellular senescence109. In addition, SNPs associated with absence of disease were found in genes involved in neural development and function, as well as in autophagy108. Genetic variations in the IIS-related gene FOXO3A are associated with healthy aging and lifespan extension in long-lived and healthy humans110. By recapitulating human polymorphisms in animal models, such as by using genome-editing approaches, researchers may be able to directly study these variants in inbred mouse strains of homogeneous background in order to advance our understanding of normative aging. Additionally, valuable information can be gathered from genetic and medical studies of centenarians. Centenarians seem to have protection against inflammation-based pathologies, including cancer and insulin resistance, and their genomes reveal the existence of genetic variants predisposing them to the production of large amounts of anti-inflammatory cytokines or smaller amounts of pro-inflammatory ones3.
In addition to polymorphism and natural variation, epigenetic drift could also be a valid predictor of human aging111. DNA hypomethylation is observed in human aging, and it is linked to increased cardiovascular disease and morbidity112. However, whether DNA methylation lowering over time can actually predict healthspan rather than chronological aging is still quite unclear. If specific epigenetic marks are found in humans, they could be validated in mice using established models of aging and thorough characterization of healthspan. Active demethylation of cytosine residues, the distribution of which is non-random throughout the genome, was recently described, and this demethylation may reflect the impact of lifestyle and environmental factors on healthspan112.
Many more molecular biomarkers remain to be identified, and could be of metabolomic and proteomic origin. Changes in energy and lipid metabolism as well as in oxidative stress are associated with aging in human plasma samples113. Again, whether these changes are passive or active biomarkers of aging remains to be determined. Another area that deserves much attention is the role of the microbiome in response to diet and aging. Studies in older adults demonstrate that the gut microbiota correlates with diet and lifestyle, and that it is associated with a basal level of inflammation which could directly enhance the susceptibility to disease114. It will be crucial to determine whether direct alteration of the microbiome can affect aging, as has been shown for metabolic dysfunction and obesity115.
The challenge of identifying biomarkers of aging
As discussed above, drug development is further hindered by the inability to define the parameters of aging itself. Early monitoring of these surrogates will be essential for therapeutic success. On the basis of the current literature, many physiological parameters that are indicators of frailty in humans might serve as readouts for aging. These include (but are not limited to) metabolic health, cardiac function, blood pressure, bone health, body weight, muscle strength, cognitive performance, cerebral blood flow, brain atrophy, circulating hormone levels, cataract formation, immune response and tumorigenesis116, 117, 118, 119. Standardizing these clinically relevant stresses in aging experimental animal models of human age-related diseases will allow a comprehensive overview of the ability of a drug to promote healthy aging. Ongoing research at the Buck Institute for Aging Research is systematically tracking many of these readouts in clinically based mouse studies of aging and of select anti-aging therapies (http://www.buckinstitute.org/BAIT-project-underway-at-the-buck-2014). The challenge for clinical trials will be to include as many of these parameters as possible, despite that many of these parameters require a vast diversity of ages, ethnicities and sexes to achieve statistical relevance. Importantly, evaluation of the phenotypic variations within these patients will help to provide a connection between observed genetic variation and physiological and molecular responses to drugs or treatments, which can already show high variability in inbred rodent models106.
Even if biomarkers of aging are validated, determining which biomarkers to measure in trials of each drug is a remaining challenge. For example, rapamycin treatment extends lifespan in mice by decreasing tumorigenesis and improving metabolic and cardiovascular health, and it could therefore be considered to have a positive effect on all biomarkers of age7, 8, 9. However, rapamycin exerts adverse effects on cataract severity in both sexes and testicular degeneration in males9. Furthermore, a trial designed to measure a single readout of aging is only reflective of the changes in this biomarker, which may not be related to the actual process of aging, thus reinforcing the necessity of surveying a wider array of clinical hallmarks of aging. Another critical variable in terms of measuring aging markers is the actual age at which measurements should be taken to observe functional decline. Whereas some parameters of normal aging, such as cognitive decline, only develop in later life, other parameters have an early onset: muscle atrophy and wasting are often the first signs of aging, starting to increase significantly around age 30 (ref. 120). Other parameters may also be quite variable, such as the percentage of body fat, which typically doubles around age 75, thereby increasing the risk of glucose intolerance and diabetes mellitus117. In addition, the onset of particular biomarkers may differ significantly between the sexes, as menopause favors a higher incidence of cardiovascular disease, loss of bone mass and cognitive impairment in the years after estrogen loss121.
Conclusions and future outlook
It is difficult to envision that a single medical strategy would be miraculous enough to treat all age-associated pathologies simultaneously and restore healthy aging in aged individuals. Furthermore, aging is not a clinical condition recognized by regulatory bodies or insurance providers for therapeutic reimbursement. However, there is hope for discovering therapeutics that could efficiently target several chronic diseases of aging through better scientific and clinical understanding of the diseases of aging. The ultimate goal of geroscience is to devise strategies to boost natural defenses and prolong healthspan through better management of the threats posed to an individual's cells and tissues.
To achieve this goal, important regulatory approaches must be implemented to improve diagnostic and screening tools, with the goal of targeting the primary source of homeostatic loss or tissue dysfunction at an early stage of disease before the onset of comorbidities. Conventional medical care is focused more on symptom relief with pain medications and surgical procedures, and it is not developed enough when it comes to delaying the aging process, which may potentially be more effective with respect to improving healthspan in the long term.
In conclusion, aging is remarkably complex, and many of its definitions and rules remain undefined. Genetic perturbations and diet restriction have increased longevity across species in well-defined experimental settings and isolated genetic backgrounds, but exceptions found within heterogeneous mouse populations find that these treatments can also result in shortened lifespan, raising concern that clinical geroscience is premature. Nonetheless, the modulation of genetic pathways to greatly increase longevity in model organisms has brought the realization that aging can be manipulated. Furthermore, simple, commonly used drugs have been found to delay aging and increase lifespan in animal models, raising the idea that we might already have an elixir for longevity in hand, albeit with unknown mechanisms. However, the formulation or identification of a pharmaceutical designed specifically to combat aging with fewer side effects and better potency than today's compounds is alluring and tempting. With the hurdles of clinical development for an anti-aging compound coupled with personalized gerontology and predictive, relevant biomarkers, the bar for a 'geraceutical' could be too high for a single entity to clear, but the future healthcare costs of our increasing aging population are too great to not try.
|
|
| |
| |
|
|
|