| |
Generics HIV ART PK Risky?
|
| |
| |
Download the PDF here
There are a lot of concerns about the effectiveness of Generic HIV ARTs....they may putpatientsat risk for failure & drug resistance
HIV Medications are not like candy or some lightweight medication. ARTs are antivirals that require strict PK levels that crucially are enforced in branded drug manufacturing because the development of drug resistance leads to drug failure, drug resistance & ultimately to death possibly. Standards for generic FDA PK are very flexible, the FDA permits PK to be within 80% to 125% of branded pk requirements. Even with that some generics have been banned from the USA for corrupt & bad manufacturing.CONSIDER THIS, an ART with PK at 80-125% off branded pk & a person with poor levels anyway as you know there can be quite a inter person variability in PK/drug levels - this person takes another medication that can reduce the levels of the ART drug & this results in even lower ART drug levels - then consider non-adherence by that patient - he/she misses a dose - then consider polypharmacy in an older 70 year old HIV+ person who is on 5 drugs in addition to ARTs and there might be interactions between some of them & the ARTs. We have no actual well done data proving that generics are as good & effective in developing world compared to the USA.
CONSIDER THIS: 15% of HIV+ who are taking Reyatax are taking 400 once daily Reyataz without ritonavir booster. We all know ritonavir booster significantly increases blood ATV levels so adherence & missing code has a little flexibility. When a generic reyataz 200 mg pill comes out this will remove the buffer these patients on unboosted ATV have if their ATV levels are not great, we all know there is inter patient variability, some patients for some reason achieve high levels & some fall below the levels needed.
FDA
"Before July, 1992, 90% confidence intervals for AUC and
Cmax had to be within the range of 80 to 120% around
the RLD mean value
July, 1992 statistical procedures guidance recommends
confidence intervals of 80 - 125% for AUC and Cmax
after log transformation"
Both AUC and Cmax - 90% CI of T/R
ratios should fall within 80 - 125%
http://www.fda.gov/downloads/ AdvisoryCommittees/.../UCM209319.pdf
FDA PK ART Requirements are Too Lenient.....FDA bioequivalence range permits variance within these ranges - 0.80-1.25 . Some studies find PK outside even these ranges.Generics in other diseases have been found to fail patients. Generic makers have been fined for not even following FDA PK Guidelines.
FDA prohibits Ranbaxy's Toansa, India facility from producing and distributing drugs for the U.S. market
Another Ranbaxy facility added to existing consent decree.....http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm382736.htm
(1) Dr. Harry Lever is a cardiologist at the Cleveland Clinic, and, like all cardiologists, he prescribes diuretics—including furosemide—to help prevent heart failure. "Some of my patients," recalls Dr. Lever, "were taking the brand medication [Lasix]; then under cost pressure from insurers, I switched them to a generic and they reacted very badly [one patient added ten pounds of weight rapidly]. Only when I switched them back did they recover."[2]....http://www.aei.org/publication/cheap-indian-generic-drugs-not-such-good-value-after-all/
(2)In November, the FDA reported that the Gurgaon, India-based Ranbaxy Laboratories Limited had issued a proactive voluntary recall of its anticholesterol drug atorvastatin because of possible glass particles in the medicine. Though the FDA had not received any reports of Americans being injured at the time of the recall, the quality of Ranbaxy's drugs has been a hot topic in the pharmaceutical industry for some time. Ranbaxy is one of the largest and most respected Indian drug companies, and the FDA granted it the sole generic license for the manufacture of generic Lipitor (atorvastatin) beginning in November 2011. Lipitor is the most valuable medicine by sales in the world, therefore it was a coup for Ranbaxy to be awarded the license.In 2005, whistleblowers from Ranbaxy alerted the FDA that members of Ranbaxy's staff were deliberately cutting corners in producing HIV medication to be bought with US taxpayer funds. The FDA and US Department of Justice identified two questionable Ranbaxy plants and 30 suspect medications, yet only restricted their US-bound sales in 2010.....On May 9, 2012, the Indian Government's Parliamentary Standing Committee on Health and Family Welfare (PSCHFW) presented a 118-page evaluation of the Central Drugs Standard Control Organization (CDSCO), the government's federal drug regulator. PSCHFW eviscerated the CDSCO for corruption, and concluded that many apparently independent expert health opinions about the safety of drug products "were actually written by the invisible hands of drug manufacturers."[7]
(3) The bioequivalence range of 0.80-1.25 for AUC0-t, AUC0-¥, and Cmax wich is accepted by major health authorities including the European CPMP (Committee for Proprietary Medicinal Products), and the US FDA (United State Food and Drug Administration), was used in this study (Committee for Proprietary Medicinal Products. CPMP/EWP/QWP/1401/98. Note for guidance: investigation of bioavailability and bioequivalence. Food and Drugs Administration (FDA) Division of Biopharmacutics. Bioavailability Protocol Guideline for ANDA and NDA Submission. 1977. Food and Drug Administration (FDA), The Division of Bioequivalence. 1992. Statistical procedures for bioequivalence studies using a standard two-treatment crossover design. FDA Guidance).
Results: The present study showed that no significant differences were observed in the means of AUC0-12, AUC0-¥, C max, and T max of both formulations of stavudine, lamivudine zidovudine, and indinavir. It wasconcluded that the studied pharmacokinetic parameters are withinthe established range. The ranges for Cmax, AUC0-12, and AUC0-¥ were 80-125 %, whereasthe range for Cmax of zidovudine was 70-143 %. ....http://www.bvs.sld.cu/revistas/far/vol40_2_06/far02206.htm
Stavudine, lamivudine and nevirapine pharmacokinetic parameterinter-individual variability ranged from 29% to 99%(Table 2).....Nevirapine exposure inour study was lower than that reported in a recent study fromMalawi.10
Median nevirapine Ctrough was 3615.0 ng/mL (range, 1236-6138) while on the generic formulation and 4479.5 ng/mL(range, 1070-8375) while on Viramunew. Eleven of the 16subjects for the generic and 10 of 16 for the branded hadnevi rapineconcentrations above the suggested minimum effectiveconcentration (MEC) of 3000 ng/mL.8 Only one subject hadCtrough below the MEC while on both formulations.
Although the mean nevirapine pharmacokinetics were similarfor the two formulations, there was considerable inter- and intrasubjectvariability. Steady-state Ctrough concentrations were abovethe alleged MEC of 3000 ng/mL8 for 11 of 16 subjects for thegeneric and 10 of 16 for the branded
Both the branded and generic formulations of stavudine,lamivudine and nevirapine were well tolerated and no differenceswere reported.
The geometric mean ratio (90% CI) for stavudine, lamivudine and nevi rapinewas 0.92 (0.78-1.08), 1.11 (0.95-1.30) and 0.84 (0.64-1.11), respectively, for Cmax, and 0.83 (0.70-0.97), 1.06 (0.94-1.20) and 0.88 (0.71-1.10), respectively, for AUC. Stavudine plasma concentrations weresignificantly lower for the generic formulation. Pharmacokinetic parameter inter-individual variability ranged from 29% to 99%. There were no differences in tolerability for the two formulations.
Conclusions: Pharmacokinetic profiles of generic and branded drugs were similar. Differences particularlywith regard to stavudine were demonstrated. Surveillance of the quality of generic antiretroviral drugs in the target populations is needed. Capacity building for pharmacokinetic research in resource limitedsettings is a priority.
Mean [+standard error (SE)] concentration-time profile forthe three drugs is shown in Figure 1. The mean (+SD) stavudine,lamivudine and nevirapine steady-state pharmacokinetic parametersmeasured during the administration of branded and genericformulations are reported in Table 2. Mean (SD)nevirapineexposure (AUC0-12) while on branded and generic drugs was75 192.7 ng.h/mL (29 294.9) and 64 338.3 ng.h/mL (19 944.6),respectively. GMR of the test (generic) to reference (brand) ofCmax and AUC values and 90% CI are given for each drug in Table 2. While no significant differences in lamivudine andnevirapine Cmax and AUC were observed [GMR (90% CI): 1.11 (0.95-1.30) and 0.84 (0.64-1.11) for Cmax, and 1.06 (0.94-1.20)and 0.88 (0.71-1.10) for AUC], stavudine plasma concentrations were significantly lower for the generic formulation for Cmax 0.92(0.78-1.08) and AUC 0.83 (0.70-0.97).
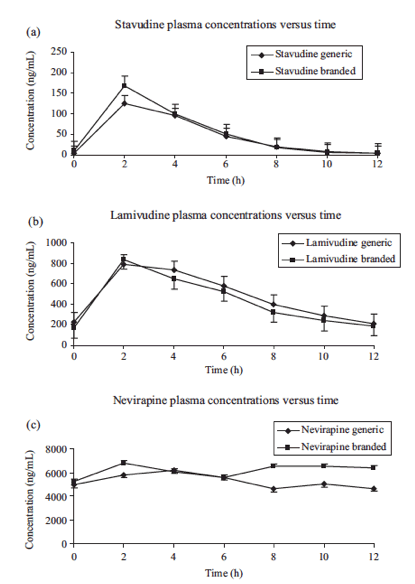
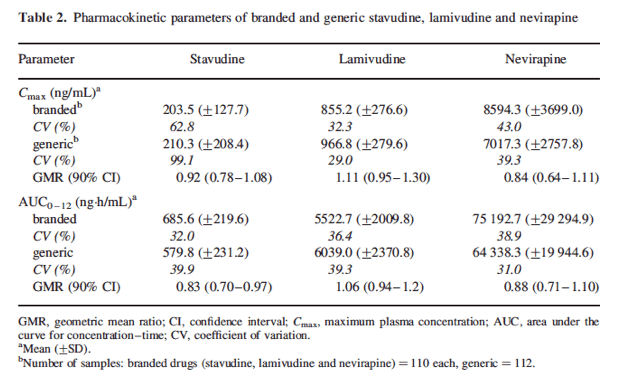
With the exception of lamivudine, steady-statepharmacokinetic parameters of Triomune 40 did not fall withinthe limits of the US Food and Drug Administration standardsfor bioequivalency testing.6 However, pharmacokinetic profileswere similar, and there was no difference in tolerability between
the two regimens. Evidence for bioequivalence of Triomune40 to branded products was based on a single-dose study inhealthy volunteers.9 Since adequate drug concentrations must bemaintained for long-term suppression of HIV, it is arguable thatsteady-state pharmacokinetic studies are of greater clinicalsignificance than single-dose studies.
|
|
| |
| |
|
|
|