 |
 |
 |
| |
Serious adverse events and direct-acting antivirals drugs (DAA): ANRS CO22 HEPATHER cohort
|
| |
| |
Alpha Diallo1, Imane Amri1, Severine Gibowski1, Celine Dorival2, Melanie Simony3, Helène Fontaine4, Stanislas Pol4, Fabrice Carrat2,5 et ANRS/AFEF HEPATHER study group.
Reported by Jules Levin
AASLD: The Liver Meeting 2017; Washington, DC, USA; October 20-24, 2017.
1. ANRS (France REcherche Nord&sud Sida-hiv Hepatites), Service de Vigilance des Recherches Cliniques, Paris, France. 2. UPMC Universite Paris 06, INSERM, Institut Pierre Louis d'epidemiologie et de Sante Publique (IPLESP UMRS 1136), Sorbonne Universite, Paris, France. 3. ANRS (France REcherche Nord&sud Sida-hiv Hepatites), Service de recherche clinique Hepatites, Paris, France. 4.Universite Paris Descartes ; APHP, Unite d'Hepatologie, Hopital Cochin ; INSERM U-818 et USM20, Institut Pasteur, Paris, France. 5. Unite de Sante Publique, Assistance Publique-Hopitaux de Paris, Hopital Saint Antoine, Paris, France.

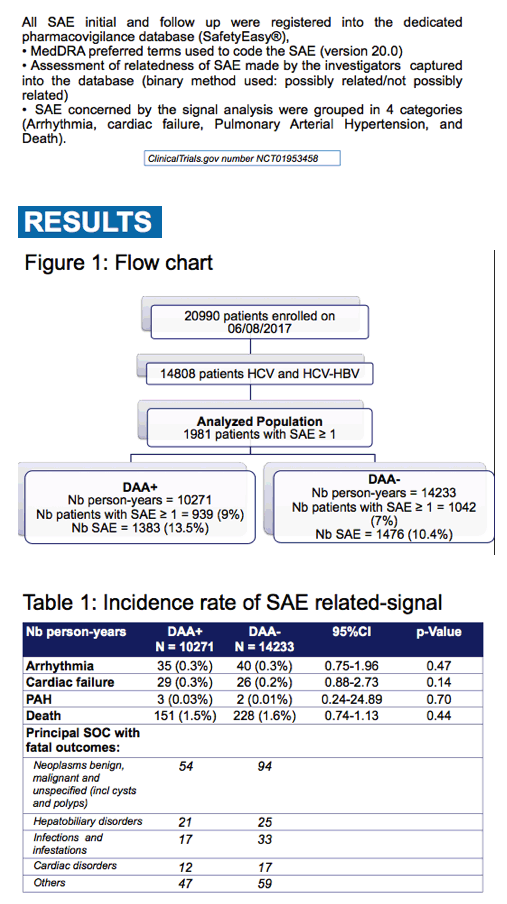


|
| |
|
 |
 |
|
|