 |
 |
 |
| |
Switching from boosted protease inhibitors (PI/r) to dolutegravir (DTG) in virologically suppressed HIV-infected patients with high cardiovascular risk:
48-week effects on subclinical cardiovascular disease (CVD)
|
| |
| |
Switching From PI to DTG (Nonsignificantly) Improves Cardiovascular Markers
16th European AIDS Conference, October 25-27, 2017. Milan
Mark Mascolini
Older adults with a high Framingham cardiovascular risk score had slower progression of carotid artery intima media thickness (cIMT) and better arterial stiffness measures if switching to dolutegravir (DTG) rather than staying with a boosted protease inhibitor (PI) in the multicenter NEAT022 trial [1]. But between-arm differences did not approach statistical significance.
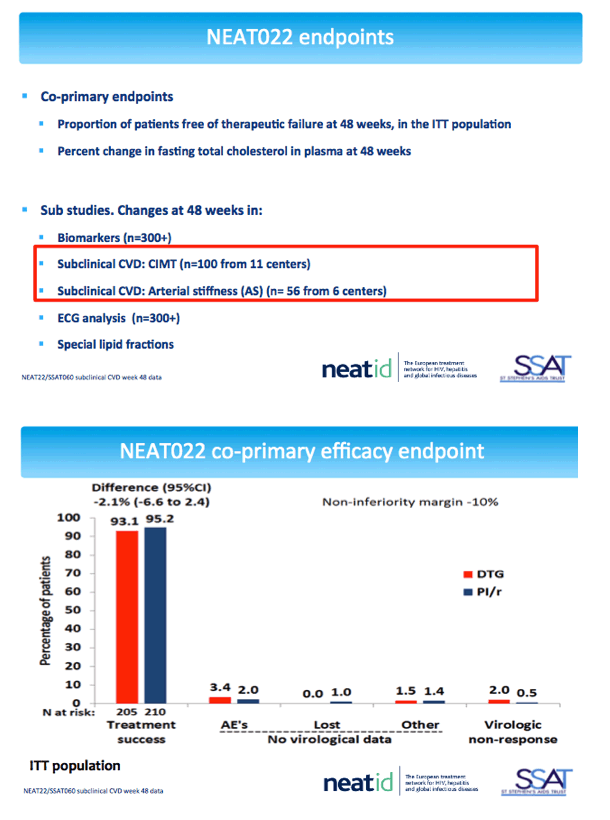
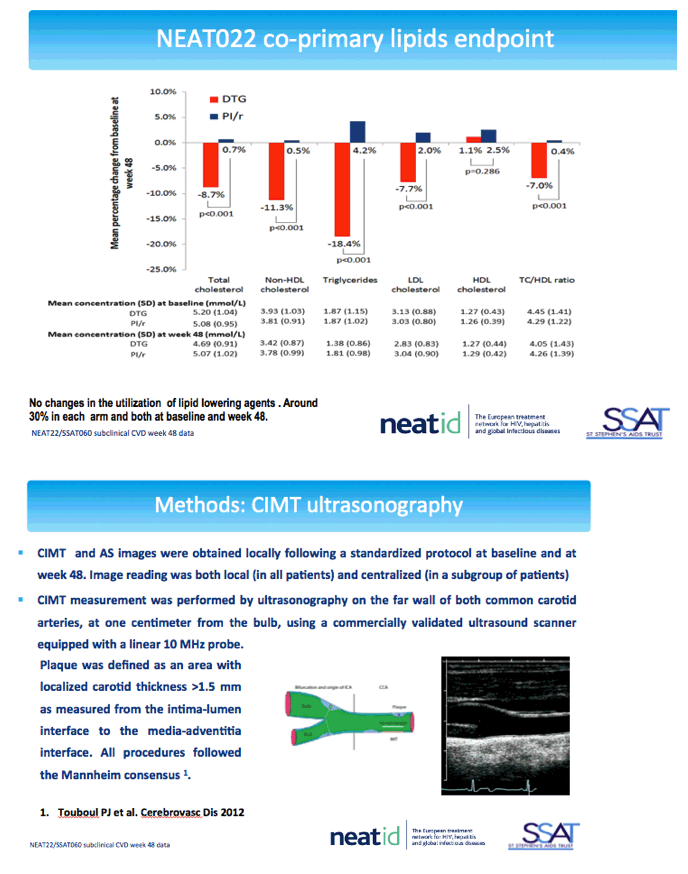
NEAT022 is a multicenter open-label noninferiority trial that randomized 415 participants to continue a boosted PI or switch to the integrase inhibitor DTG. Participants were older than 50, had a Framingham cardiovascular risk score above 10%, and kept a viral load below 50 copies for at least 6 months on their PI regimen. After 48 weeks participants in the DTG arm maintained viral control and significantly improved lipid levels [2].
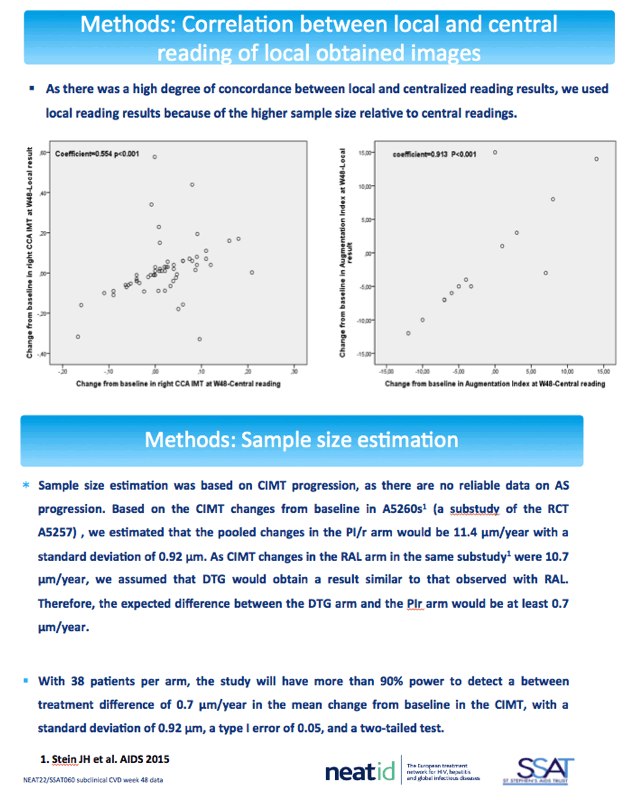
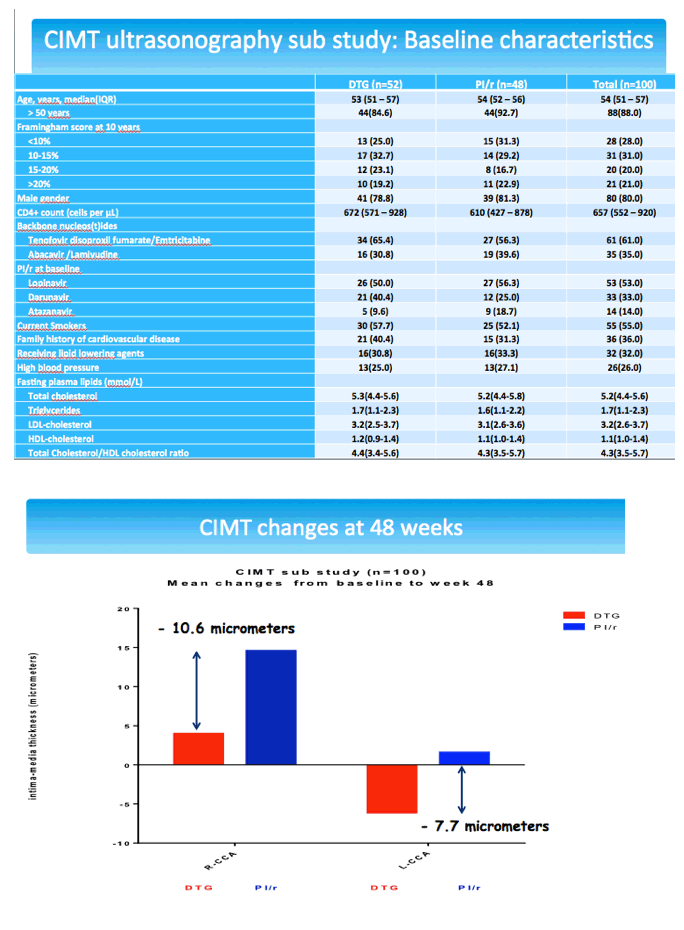
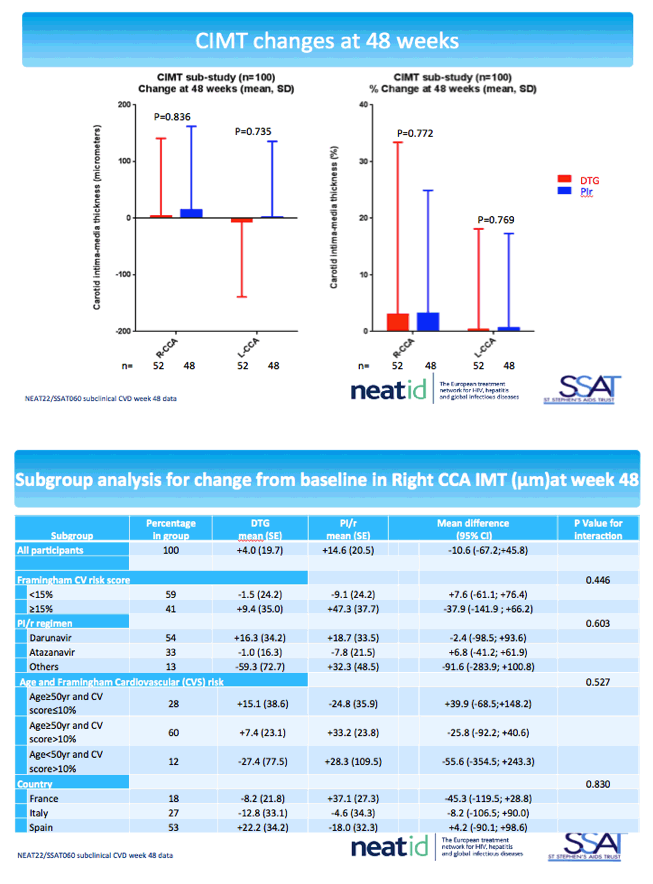
The cardiovascular substudy involved 100 people from 11 centers who had cIMT measured by ultrasonography at baseline and week 48 and 56 people from 6 centers who had baseline and week-48 measures of arterial stiffness by pulse wave velocity and augmentation index. The cIMT analysis involved 52 people randomized to DTG and 48 randomized to continue their PI. Median ages of the DTG and PI groups were 53 and 54, and about 80% in both groups were men. Proportions of current smokers with DTG and a continued PI were 58% and 52%, and proportions with a family history of cardiovascular disease 40% and 31%.
Through 48 weeks cIMT in the right common carotid artery increased less with DTG than a PI (average -10.6 micrometers difference) while left common carotid artery thickness dropped with DTG and rose slightly with a continued PI (average -7.7 micrometers difference). These differences lacked statistical significance. Subanalyses by Framingham risk score (above or below 15%), age and Framingham risk score, PI taken, and country (France, Italy, or Spain) also failed to discern statistically significant differences between study arms.
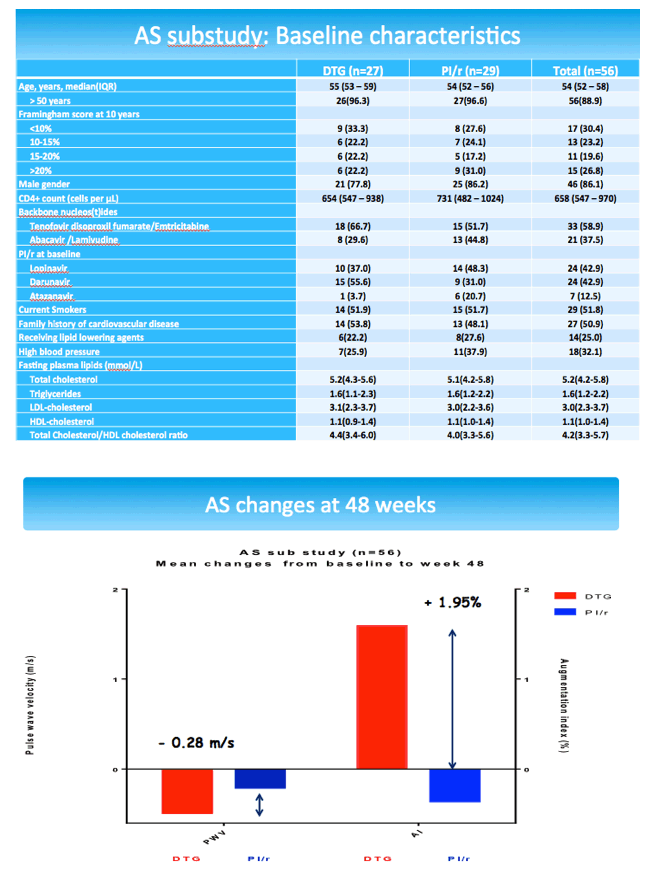
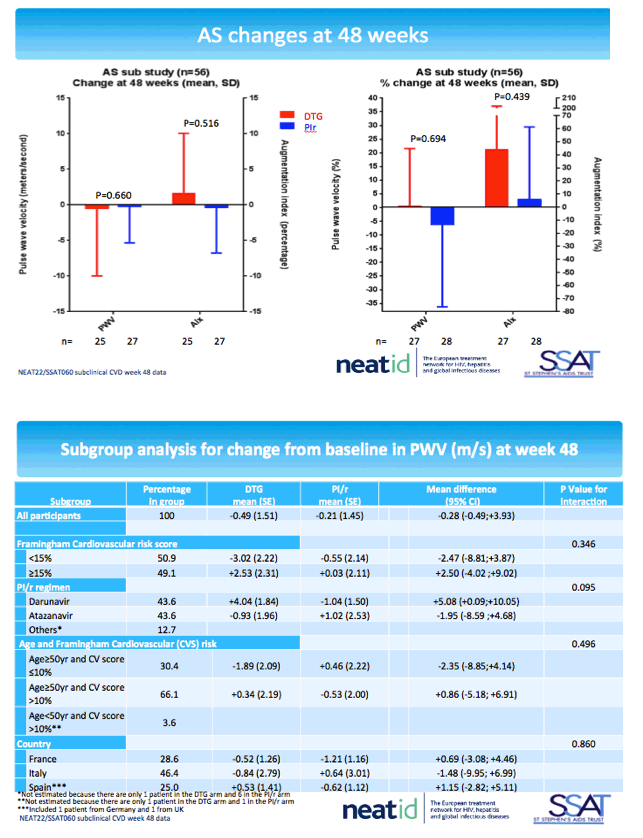
The arterial stiffness substudy had 27 participants in the DTG arm and 29 in the PI arm. Respective median ages were 55 and 54, proportions of men 78% and 86%, current smokers 52% and 52%, and family cardiovascular disease history 54% and 48%. Through 48 weeks pulse wave velocity dropped (improved) slightly more with DTG than a continued PI (average -0.28 m/s difference), while augmentation index rose with DTG and fell with a continued PI (average +1.95% difference). These differences did not reach statistical significance in the primary analyses or in the subanalyses described in the preceding paragraph.
The NEAT022 team concluded that switching from a boosted PI to DTG results in consistently favorable though nonsignificant changes in signals of subclinical cardiovascular disease. The researchers noted that variability in study measurements (images were collected at local sites) was much greater than in ACTG A5260s [3], the trial on which NEAT022 based sample-size estimates. They suggested that longer follow-up with more participants may detect significant subclinical cardiovascular improvements with a PI-to-DTG switch.
References
1. Martinez E, Assoumou L, Camafort M, et al. Switching from boosted protease inhibitors (PI/r) to dolutegravir (DTG) in virologically suppressed HIV-infected patients with high cardiovascular risk: 48-week effects on subclinical cardiovascular disease. 16th European AIDS Conference. October 25-27, 2017. Milan. Abstract2/6.
2. Gatell JM, Assoumou L, Moyle G, et al. Switching from a boosted protease inhibitor (PI/r) based regimen to a dolutegravir regimen in virologically suppressed patients with high cardiovascular risk or age >50 years is non-inferior and decreases lipids. 9th IAS Conference on HIV Science (IAS 2017), July 23-26, 2017, Paris. Abstract TUAB0102. http://www.natap.org/2017/IAS/IAS_82.htm
3. ClinicalTrials.gov. Impact of antiretroviral therapy on metabolic, skeletal, and cardiovascular parameters. ClinicalTrials.gov identifier NCT00851799. https://clinicaltrials.gov/ct2/show/NCT00851799
--------------------------
Switching from boosted protease inhibitors (PI/r) to dolutegravir (DTG) in virologically suppressed HIV-infected patients
with high cardiovascular risk:
48-week effects on subclinical cardiovascular disease (CVD)




|
| |
|
 |
 |
|
|