 |
 |
 |
| |
EDP-305, a novel and highly potent farnesoid X receptor agonist, improves liver steatosis, ballooning and non-alcoholic fatty liver disease (NAFLD) activity score (NAS) in a diet-induced murine model of non-alcoholic steatohepatitis
|
| |
| |
Reported by Jules Levin
EASL 2017 April 19-23 Amsterdam Netherlands
Li-Juan Jiang1, Mary Chau1, Yang Li1, François Briand2, Thierry Sulpice2, Yat Sun Or1
Enanta Pharmacueticals, Waterstown, Mass, Physiogenix, Laberge, France

Program Abstract
Background and aims
EDP-305 is a novel farnesoid X receptor (FXR) agonist with a single-digit nanomolar affinity for FXR in vitro. EDP-305 is highly selective for FXR and does not exhibit any cross-reactivity with other nuclear receptors, or the transmembrane G protein-coupled receptor TGR5. Herein, we compare and report the therapeutic efficacy of EDP-305 with the FXR agonist, obeticholic acid (OCA), in an experimental non-alcoholic steatohepatitis (NASH) mouse model.
Methods
NASH was induced in C57Bl/6 mice with a high fat/high cholesterol diet plus 10% fructose in drinking water (DIN). All treatments were administered between 6 wks (steatohepatitis with incipient ballooning) and 16 wks (advanced steatohepatitis with advanced ballooning) on DIN (n = 10/group). Treatments included: EDP-305 (10 or 30 mg/kg), OCA (30 mg/kg) or only DIN vehicle control (n = 10/group). Liver injury, progression of NASH, liver steatosis and ballooning were evaluated by serum chemistry and histology.
Results
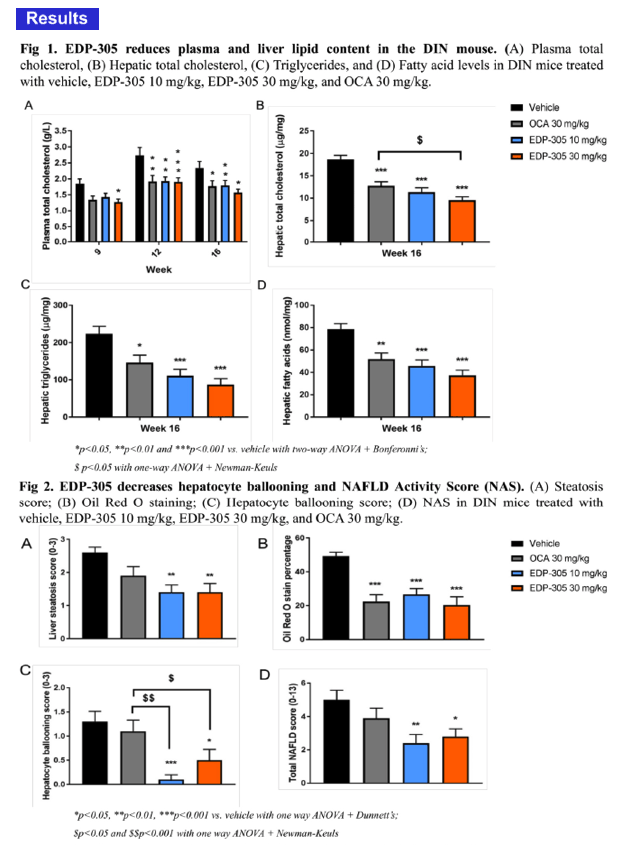
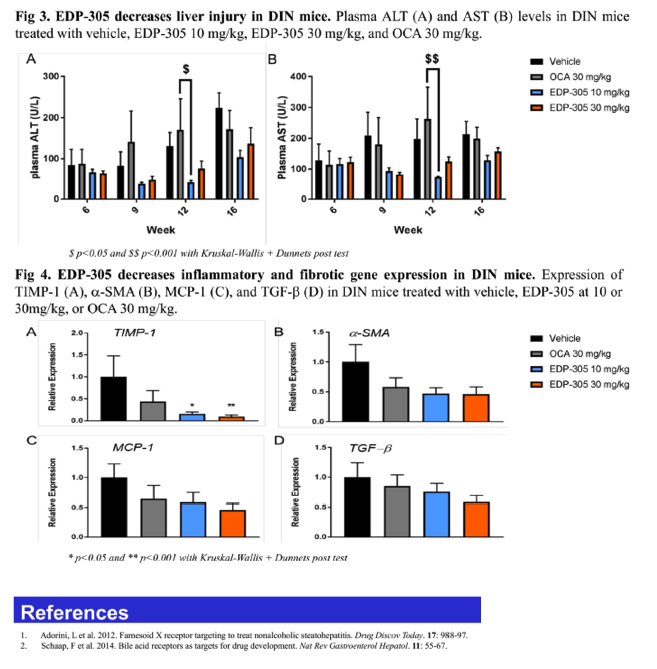
No apparent adverse effects of treatments were noted during the study. Mice receiving EDP-305 showed a distinct trend towards lower ALT/AST levels compared to vehicle control, whereas OCA had no effects. At both doses, EDP-305 significantly reduced liver lipids, such that there were significant reductions in hepatic cholesterol, triglycerides and fatty acids. OCA reduced liver lipids to a lesser extent than EDP-305. EDP-305 at 30 mg/kg significantly decreased liver cholesterol (48%↓: versus vehicle) compared to OCA at the same dose(31%↓: versus vehicle). Consistent with the observed reductions in liver lipids, liver steatosis was also significantly decreased in EDP-305-treated mice at both doses (p<0.01). Meanwhile, OCA did not exert a significant effect on liver steatosis. At both doses, EDP-305 significantly lowered hepatocyte ballooning scores, whereas treatment with OCA showed no improvement. Additionally, EDP-305 significantly reduced total NAS in both treatment groups, which is consistent with data showing that EDP-305 down-regulates expression of key genes involved in pathogenic inflammation (MCP-1, TGF-β) and fibrosis (TIMP-1, α-SMA).
Conclusions
In comparison to OCA, treatment with EDP-305 demonstrated significant therapeutic effect on NASH progression in the diet-induced NASH mouse model, such that it decreased liver steatosis, hepatocyte ballooning and total NAS score. These results warrant further investigation of EDP-305 as a potential therapy for NASH.

|
| |
|
 |
 |
|
|