| |
UK-NICE Guidance - Sofosbuvir-velpatasvir for treating chronic hepatitis C
|
| |
| |
Download the PDF here
Lancet March 2017
*Sophie Cooper, Nwamaka Umeweni, Helen Knight, Gary McVeigh
National Institute for Health and Care Excellence, Manchester M1 4BT, UK
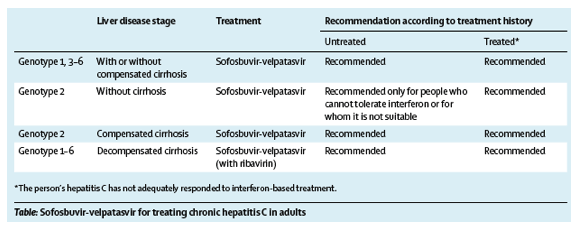
On Jan 25, 2017, the UK National Institute for Health and Care Excellence (NICE) published guidance1 recommending sofosbuvir-velpatasvir (Gilead Sciences Ltd, Uxbridge, UK) as an option for treating chronic hepatitis C in adults (table), only if the manufacturing company provides the drug with the price reduction in the simple discount agreement.
NICE appraised sofosbuvir-velpatasvir as a single technology appraisal.2 Gilead submitted clinical and cost-effectiveness evidence, which was critiqued by an independent evidence review group (Kleijnen Systematic Reviews, York, UK).3 An independent appraisal committee met twice to develop the guidance. Clinical, patient, and NHS commissioning experts attended the first meeting, and the company attended both meetings.
The clinical evidence for sofosbuvir-velpatasvir in this appraisal came from four randomised phase 3 clinical trials (ASTRAL-1, -2, -3, and -4) which included all six major genotypes of hepatitis C virus (HCV) in people with previously untreated or treated hepatitis C. ASTRAL-1, -2, and -3 included people with compensated cirrhosis and compared 12 weeks of sofosbuvir-velpatasvir with 12 weeks of placebo (ASTRAL-1) or sofosbuvir plus ribavirin (ASTRAL-2 and ASTRAL-3). ASTRAL-4 included people with decompensated cirrhosis and compared three different sofosbuvir-velpatasvir regimens. The primary outcome in all four trials was sustained virological response 12 weeks after the end of treatment (SVR12). The trials showed high rates of SVR12 irrespective of HCV genotype, cirrhosis stage, or treatment history; ranging from 89% (for people with previously treated genotype 3 HCV and compensated cirrhosis) to 100% (in several subgroups). The committee noted that the trial results showed that sofosbuvir-velpatasvir has a relatively favourable tolerability profile, especially when compared with peginterferon alfa plus ribavirin. The company explored the feasibility of doing a network meta-analysis to compare sofosbuvir-velpatasvir with all of the comparators in the NICE scope. The company was able to construct evidence networks for only two subgroups (untreated genotype 1 or 3 HCV), but these analyses were associated with several limitations.
To estimate cost-effectiveness, the company submitted a Markov model that compared sofosbuvir-velpatasvir with the relevant treatments for each subgroup according to genotype, cirrhosis stage, and treatment history. The structure of the model and its assumptions about the natural history of the disease were similar to models submitted for other NICE technology appraisals for chronic hepatitis C. Health states were based on stage of liver disease and transition probabilities between health states were taken from several studies. Gilead assumed that disease progression, from non-cirrhotic to compensated cirrhosis health state, is faster in genotype 3 HCV compared with other genotypes. The model did not allow for reinfection after SVR12 or include a risk of future virus transmission. Because the company could not perform a full network meta-analysis to inform the efficacy inputs in the model, it estimated SVR12 for each comparator based on individual arms of selected randomised controlled trials. Utility values for each health state were derived from published scientific literature. Health-state utilities during the on-treatment period were adjusted using treatment-specific utility increments or decrements. The company applied decrements for regimens containing peginterferon alfa or ribavirin to reflect the poor tolerability of these treatments, and applied utility increments for direct-acting antivirals to reflect the improved tolerability profile and benefits of rapidly suppressing HCV.
The committee would have preferred to see a model including both reinfection and transmission, but appreciated that this would have needed a different model structure. The committee understood that the company's assumption of faster disease progression in genotype 3 HCV was consistent with previous NICE technology appraisals in hepatitis C, but the committee disagreed with the company's approach to estimating HCV genotype-specific transition probabilities. The company used unadjusted results from Kanwal and colleagues' paper,4 but the committee agreed that the transition probabilities should be based on the prespecified analyses adjusting for patients' baseline characteristics. The committee was aware that transition probabilities for disease progression in people with cirrhosis were based on Cardoso and colleagues' paper5 and recalled its conclusion from previous technology appraisals for hepatitis C that these transition probabilities lay somewhere between the estimates from Cardoso and colleagues' paper5 and those from Fattovich and colleagues' paper.6 The committee heard from the clinical experts that data from Fattovich and colleagues' paper6 are generalisable to current practice and concluded that both sources should be used. The committee was prepared to accept the company's utility estimates from published literature, but emphasised that it prefers utility values collected from the clinical trials of the intervention under evaluation. The committee considered it acceptable to include treatment-specific utility increments and decrements in the model but was concerned that this could lead to double counting, because the company also included utility increments for SVR12 and utility decrements for adverse events.
The evidence review group presented the results of several exploratory analyses; these included the confidential simple discount agreement for sofosbuvir-velpatasvir and the confidential reduced contract prices for the comparators, when known and if important to the committee's decision-making. The committee was aware that incremental cost-effectiveness ratios (ICERs) incorporating its preferred assumptions about transition probabilities (ie, using data from Cardoso and colleagues5 and Fattovich and colleagues,6 and the adjusted data from Kanwal and colleagues4) were available in only two subgroups: people with untreated genotype 2 or genotype 3 HCV and no cirrhosis. The evidence review group focused on the comparisons with peginterferon alfa in these groups because they produced the highest ICERs for sofosbuvir-velpatasvir in the company's base case. The committee concluded that the most plausible ICERs for sofosbuvir-velpatasvir compared with peginterferon alfa plus ribavirin lay between 35 091 and 39 783 per quality-adjusted life-year (QALY) gained for people with untreated genotype 2 HCV without cirrhosis, and between 15 923 and 18 362 per QALY gained for people with untreated genotype 3 HCV without cirrhosis.
The committee considered the likely effect of including its preferred assumptions on the company's base-case ICERs for sofosbuvir-velpatasvir compared with relevant comparators in all other subgroups. It concluded that they would remain below 20 000 per QALY gained regardless of HCV genotype, treatment history, and cirrhosis stage. Exact ICERs for all comparisons cannot be reported because the contract prices for the comparators are confidential and cannot be disclosed.
Based on the most plausible ICERs agreed by the committee, it concluded that sofosbuvir-velpatasvir was cost effective for treating HCV genotypes 1 and 3-6 regardless of treatment history and cirrhosis stage, and for specific people with HCV genotype 2 (table). The committee could not recommend sofosbuvir-velpatasvir as a cost-effective use of NHS resources for untreated genotype 2 HCV in people without cirrhosis who can have interferon, because the ICER was above 30 000 per QALY gained compared with the only active treatment option for this subgroup (peginterferon alfa plus ribavirin). However, for people who cannot tolerate interferon or for whom it is not suitable, and for whom the only active treatment option is sofosbuvir plus ribavirin, the committee noted that the ICER for sofosbuvir-velpatasvir was below 20 000 per QALY gained after accounting for its preferred assumptions. Therefore, the committee concluded that sofosbuvir-velpatasvir could be recommended as a cost-effective use of NHS resources for untreated genotype 2 HCV for people without cirrhosis, only if they cannot tolerate interferon or it is not suitable for them. There were no requests to correct factual inaccuracies or any appeals against the recommendations.
|
|
| |
| |
|
|
|