| |
Sofosbuvir-velpatasvir-voxilaprevir with or without ribavirin in direct-acting
antiviral-experienced patients with genotype 1 hepatitis C virus - Phase 2
|
| |
| |
Download the PDF here
Hepatology June 2017 - Eric Lawitz,1 Fred Poordad,1 Jennifer Wells,1 Robert H. Hyland,2 Yin Yang,2 Hadas Dvory-Sobol,2 Luisa M. Stamm,2
Diana M. Brainard,2 John G. McHutchison,2 Carmen Landaverde,1 and Julio Gutierrez1
Abstract
The optimal retreatment strategy for hepatitis C virus (HCV) genotype 1-infected patients who fail direct-acting antiviral (DAA)-based regimens remains unknown. In this phase 2, open-label study conducted at a single center in the United States, patients with HCV genotype 1 infection who previously failed to achieve sustained virological response (SVR) on a DAA-based regimen were randomized to receive treatment with a fixed-dose combination tablet of sofosbuvir-velpatasvir-voxilaprevir with or without ribavirin (RBV) for 12 weeks. Patients were stratified by their cirrhosis and past nonstructural protein (NS) 5A inhibitor exposure. The primary efficacy endpoint was the proportion of patients with SVR at 12 weeks after treatment (SVR12). SVR12 was achieved by 24 of 24 patients (100%; 95% confidence interval [CI], 86-100) receiving sofosbuvir-velpatasvir-voxilaprevir alone and 24 of 25 (96%; 95% CI, 80-100) receiving the same treatment with RBV. None of the patients discontinued sofosbuvir-velpatasvir-voxilaprevir therapy because of an adverse event (AE). The most commonly reported AEs with sofosbuvir-velpatasvir-voxilaprevir alone were diarrhea and bronchitis; and with sofosbuvir-velpatasvir-voxilaprevir plus RBV were fatigue, anemia, gastroenteritis, and nausea. Conclusion: A fixed-dose combination of sofosbuvir-velpatasvir-voxilaprevir was well tolerated and effective at achieving virological response in patients with HCV genotype 1 infection and past DAA treatment experience.
Of the six major hepatitis C virus (HCV) genotypes, infection with genotype 1 is the most prevalent, accounting for nearly half of all HCV infections worldwide.[1] A paradigm shift in the treatment of patients with chronic HCV genotype 1 infection occurred in 2011 with the introduction of direct-acting antivirals (DAAs), which interfere with HCV replication by targeting viral proteins such as the nonstructural protein (NS) 3/4A serine protease, NS5A, and the polymerase, NS5B.[2-4] For the few patients with genotype 1 infection who fail interferon (IFN)-free DAA-based therapies, retreatment strategies remain unclear.
A fixed-dose combination of the nucleotide analog inhibitor of the NS5B polymerase, sofosbuvir, and the NS5A inhibitor, velpatasvir, is approved for treatment of HCV genotype 1-6 infection.[5] Voxilaprevir is a potent inhibitor of the NS3/4A protease that maintains its activity against common genotype 1 NS3 variants.[6] In a previous phase 2 study, all DAA-experienced patients with genotype 1 infection achieved a sustained virological response (SVR) upon the administration of a fixed-dose combination of sofosbuvir-velpatasvir plus voxilaprevir once-daily for 12 weeks.[7] The addition of ribavirin (RBV) has been shown to improve rates of SVR in response to treatment with a number of regimens, including sofosbuvir-velpatasvir for patients with decompensated cirrhosis.[8] The potential benefit to DAA-experienced patients of the addition of RBV to a sofosbuvir-velpatasvir-voxilaprevir regimen has not been explored.
In this study, we evaluated the efficacy and safety of a fixed-dose combination of sofosbuvir-velpatasvir-voxilaprevir with and without RBV administered once-daily for 12 weeks in patients with genotype 1 HCV infection who were previously treated with a DAA-based regimen.
PATIENTS
This study included patients with chronic HCV genotype 1 infection who were aged ≥18 years and had a body mass index (BMI) ≥18 kg/m2. Eligible patients were required to have HCV RNA ≥104 IU/mL at screening and must have received previous HCV treatment for a duration of ≥6 weeks with a regimen containing at least one DAA (NS3/4A protease inhibitor, NS5A inhibitor, or NS5B inhibitor) with or without RBV and/or pegylated IFN. The most recent treatment must have been completed ≥8 weeks before screening. Patients were excluded from the study if they met any of the following criteria at screening: had clinically significant electrocardiogram abnormalities; alanine aminotransferase (ALT) >10 × the upper limit of normal (ULN); aspartate aminotransferase levels >10 × ULN; direct bilirubin >1.5 ULN; platelets <75,000/μL for patients without cirrhosis or <50,000/μL for patients with cirrhosis; hemoglobin A1c >8.5%; estimated creatinine clearance <60 mL/min; hemoglobin <11 g/dL for female patients and <12 g/dL for male patients; albumin <3 g/dL; or international normalized ratio (INR) >1.5 × ULN unless the patient had hemophilia or was receiving anticoagulants affecting INR. Patients with hepatitis B or human immunodeficiency virus infection, or those with current or past history of clinical hepatic decompensation, solid organ transplantation, significant pulmonary or cardiac disease, or porphyria, were also excluded from the study.
All patients provided written informed consent before the initiation of any study-related procedures. This study was approved by an institutional review board and was conducted in accord with the principles of the Declaration of Helsinki and the International Conference on Harmonization guidelines.
ASSESSMENTS
Plasma samples were collected at screening, predose, weeks 1, 2, 4, 8, and 12 of treatment, and at posttreatment weeks 4, 12, and 24. HCV-RNA levels were assessed using the COBAS AmpliPrep/COBAS TaqMan HCV Quantitative Test (version 2.0; lower limit of quantification [LLOQ], <15 IU/mL; Roche, Indianapolis, IN, USA). HCV genotype and subtype were identified using the Siemens VERSANT HCV genotype INNO-LiPA 2.0 assay (Siemens, Camberley, Surrey, UK). Interleukin (IL) 28B genotype was determined by PCR amplification of the rs12979860 locus. Deep sequencing of the HCV NS3, NS5A, and NS5B coding regions was performed on samples obtained from all patients at baseline and at virological failure; resistance-associated substitutions (RASs) are reported using a 15% cutoff.[9]
Safety assessments included recording of adverse events (AEs), clinical laboratory tests, physical examinations, and vital sign measurements.
OUTCOMES
The primary objectives of the study were to evaluate the proportion of patients achieving SVR 12 weeks after the end of treatment (SVR12) and assess the safety and tolerability of sofosbuvir-velpatasvir-voxilaprevir. Secondary endpoints included determining the proportion of patients with virological failure and assessing the emergence of viral resistance to sofosbuvir, velpatasvir, and voxilaprevir during and after the end of treatment.
STATISTICAL ANALYSIS
No formal statistical hypothesis testing was performed in this study. All patients who received ≥1 dose of study drug were included in the primary efficacy and safety analysis sets. The 95% confidence intervals (CIs) for the SVR12 were calculated using the Clopper-Pearson method. Statistical analyses were summarized using SAS software (SAS Institute Inc., Cary, NC).
Results
PATIENT CHARACTERISTICS
A total of 61 patients with HCV genotype 1 were screened for this study, of whom 49 were randomized to receive treatment (Fig. 1). Among these patients, 24 and 25 patients received sofosbuvir-velpatasvir-voxilaprevir without and with RBV, respectively. A majority of the patients were male (65%), white (80%), and under 65 years of age (90%; Table 1). Slightly more than half of patients presented with cirrhosis at baseline (51%). Most patients had been treated for HCV infection only once before this study (88%), and 41% had previously received an NS5A inhibitor with or without another DAA. Five patients overall had a history of previous IFN-based therapy.
RESPONSE DURING TREATMENT
By week 4 of treatment, HCV-RNA levels were < LLOQ for all 24 patients receiving sofosbuvir-velpatasvir-voxilaprevir therapy and for 23 of 25 patients (92%) receiving sofosbuvir-velpatasvir-voxilaprevir plus ribavirin (Table 2). All patients in both treatment groups achieved HCV-RNA < LLOQ by week 8 of treatment, which was sustained through the end of treatment.
POSTTREATMENT RESPONSE AND VIRAL RESISTANCE TESTING
Overall, 24 of 24 patients (100%) receiving sofosbuvir-velpatasvir-voxilaprevir alone and 24 of 25 (96%) receiving sofosbuvir-velpatasvir-voxilaprevir with RBV achieved SVR12 (Table 2). A 61-year-old black male with HCV subtype 1a infection, cirrhosis, and IL28B CC genotype with undetectable HCV RNA from treatment weeks 2 through 12 relapsed at follow-up week 4. This patient, who had previously received treatment with ledipasvir-sofosbuvir for 24 weeks, presented with the NS5A RAS L31M at baseline; no RASs were detected in NS3 and NS5B. At relapse, M28T, Q30R, and L31M RASs were observed in NS5A, and V36M, Q41R, and D168G in NS3.
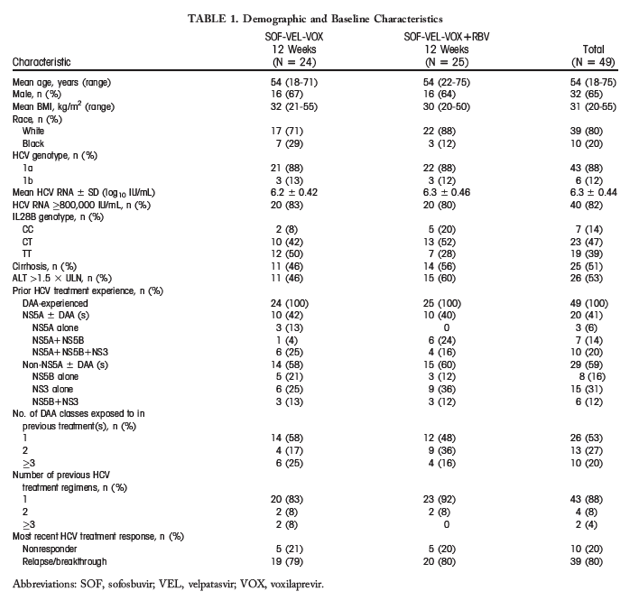
Virological response was achieved by 13 of 13 (100%) patients without baseline RASs and by 34 of 35 (97%) with baseline RASs (Fig. 2). Subgroup analyses indicated that treatment was effective regardless of the presence or absence of cirrhosis, NS5A inhibitor exposure, and the number of DAA classes used in past therapy.
Figure 2.
Virological response by baseline RASs. *Deep sequencing with 15% assay cutoff; baseline sequence was not available for 1 patient.
SAFETY
There were no discontinuations from sofosbuvir-velpatasvir-voxilaprevir therapy attributed to AEs (Table 3), whereas 3 patients (6%) discontinued RBV as a result of the AEs of anemia, fatigue, and rash, respectively. RBV dose was modified or interrupted for 3 patients (12%) because of the AE of anemia. AEs were reported in 11 patients (46%) receiving sofosbuvir-velpatasvir-voxilaprevir alone and in 15 (60%) receiving sofosbuvir-velpatasvir-voxilaprevir plus RBV, most of which were mild or moderate in severity. The most common AEs were diarrhea (13%) and bronchitis (8%) in patients receiving sofosbuvir-velpatasvir-voxilaprevir alone, and fatigue (36%), anemia (16%), gastroenteritis (8%), and nausea (8%) in those receiving sofosbuvir-velpatasvir-voxilaprevir plus RBV. Overall, 1 serious AE—pneumonia—was reported in a patient receiving sofosbuvir-velpatasvir-voxilaprevir alone and was considered unrelated to treatment. Grade 3 laboratory abnormalities were detected in 1 patient receiving sofosbuvir-velpatasvir-voxilaprevir and in 6 receiving sofosbuvir-velpatasvir-voxilaprevir plus RBV; no grade 4 abnormalities were reported. Platelet abnormalities were reported for 1 patient in the sofosbuvir-velpatasvir-voxilaprevir group. In the sofosbuvir-velpatasvir-voxilaprevir plus RBV group, abnormalities in hemoglobin were observed in 4 patients (16%), lymphocytes in 1 (4%), platelets in 1 (4%), ALT in 1 (4%), and serum glucose in 1 (4%).
Discussion
Although IFN-free DAA-based regimens are highly successful for the vast majority of patients with genotype 1 HCV infection, treatment is nevertheless unsuccessful in some patients. Retreatment options for these patients remain unclear. Current guidelines from the American Association for the Study of Liver Diseases and the Infectious Diseases Society of America recommend delaying treatment for these patients until the emergence of new data, provided they do not have cirrhosis or do not require immediate retreatment.[10]
A few studies involving a small number of patients have explored retreatment options in genotype 1-infected patients who failed to achieve a virological response with IFN-free DAA-based therapies. Retreatment approaches in these studies include prolonging treatment duration or supplementing therapy with RBV or other DAAs. For example, among patients who failed 8 or 12 weeks of previous ledipasvir-sofosbuvir-containing therapy, administration of a fixed-dose combination of ledipasvir-sofosbuvir for 24 weeks resulted in an SVR12 rate of only 71%.[11] The presence of baseline NS5A RASs increased the likelihood of treatment failure in these patients. In the C-SWIFT retreatment study, 25 patients who failed short-term (4-8 weeks) treatment with a fixed-dose combination of the NS5A inhibitor, elbasvir, and the NS3/4A protease inhibitor, grazoprevir, plus sofosbuvir were retreated with the same regimen supplemented with RBV for 12 weeks, and all patients completing treatment achieved SVR12.[12] In the QUARTZ-1 study, 21 of 22 (95%) patients who were unsuccessfully treated with previous DAA-based therapies achieved virological response upon retreatment with ombitasvir-paritaprevir-ritonavir and dasabuvir plus sofosbuvir for 12 or 24 weeks depending on genotype 1 subtype and cirrhosis status.[13] In another study, administration of sofosbuvir-velpatasvir plus voxilaprevir for 12 weeks resulted in a virological response in all 61 patients who had previously failed a DAA-based regimen, regardless of their cirrhosis status.[7]
In this open-label study, a fixed-dose regimen of sofosbuvir-velpatasvir-voxilaprevir with or without RBV administered for 12 weeks was highly effective in patients with HCV genotype 1 infection who were previously unsuccessfully treated with a DAA-based therapy. Only 1 patient in the sofosbuvir-velpatasvir-voxilaprevir plus RBV treatment arm of the study relapsed after the end of treatment. This patient had genotype 1a infection, cirrhosis, and the baseline NS5A RASs L31M, which confers a 16-fold shift in half maximal effective concentration to velpatasvir. At virological failure, the NS5A RASs M28T, Q30R, and L31M were detected. The double mutant, Q30R+L31M, is predicted to confer > 100-fold resistance to velpatasvir.[14] Additionally, no NS3 RASs were detected at baseline, but V36M, Q41R, and D168G emerged at relapse. The triple mutant, V36M+Q41R+D168G, confers 11.5-fold resistance to voxilaprevir. Overall, treatment efficacy was maintained regardless of the presence of baseline RASs, status of cirrhosis, or previous NS5A inhibitor therapy, or the number of DAA classes used in previous therapy regardless of the presence of RBV. It appears that the combination of three highly potent DAAs can obviate the need for RBV to be included in a regimen to maximize efficacy. Study treatment was also well tolerated with the observed safety profile consistent with earlier findings. This study is limited by its relatively small sample size, lack of enrollment of Asian patients, and open-label design. In four subsequent phase 3 registrational trials, patients of all genotypes and treatment histories were enrolled in blinded and open-label studies, in which treatment was similarly well tolerated and efficacious.[15, 16]
In conclusion, 12 weeks of treatment with a fixed-dose combination of sofosbuvir-velpatasvir-voxilaprevir was effective and well tolerated among patients with genotype 1 infection who had previously failed a DAA-based regimen.
|
|
| |
| |
|
|
|