| |
Glecaprevir/Pibrentasvir for HCV Genotype 3 Patients with Cirrhosis and/or Prior Treatment Experience: A Partially Randomized Phase III Clinical Trial
|
| |
| |
Download the PDF here
Hepatology Sept 19 2017- David Wyles, MD1, Fred Poordad, MD2, Stanley Wang, MD3, Laurent Alric, MD4, Franco Felizarta, MD5, Paul Y Kwo, MD6, Benedict Maliakkal, MD7, Kosh Agarwal, MD8, Tarek Hassanein, MD9, Frank Weilert, MD10, Samuel S Lee, MD11, Jens Kort, MD3, Sandra S Lovell, PhD3, Ran Liu, PhD3, Chih-Wei Lin, PhD3, Tami Pilot-Matias, PhD3, Preethi Krishnan, PhD3, Federico J Mensa, MD3
1Denver Health Medical Center, Denver, CO, USA; 2The Texas Liver Institute, University of Texas Health, San Antonio, TX, USA; 3AbbVie Inc., North Chicago, IL, USA; 4Department of Internal Medicine and Digestive Diseases, CHU Purpan, UMR 152, IRD Toulouse 3 University, France; 5Private Practice, Bakersfield, California, USA; 6Stanford University Division of Gastroenterology and Hepatology, Palo Alto, California, USA; 7University of Tennessee Health Science Center, Memphis, Tennessee, USA; 8Institute of Liver Studies, Kings College Hospital, London, United Kingdom; 9Southern California GI and Liver Centers and Southern California Research Center, Coronado, California, USA; 10Tawa Street Specialist Clinic, Hamilton, New Zealand; 11University of Calgary, Calgary, AB, Canada
Figure 1. SURVEYOR-II, Part 3 Study Design. Patients were enrolled into two arms to be treated with either 12 weeks (Arm A) or 16 weeks (Arm B) of G/P. Treatment-naiÅNve (TN) patients with cirrhosis received 12 weeks of treatment, while those with prior treatment experience (TE) and cirrhosis received 16 weeks. Patients with prior treatment experience but without cirrhosis were randomized 1:1 for treatment with either 12 or 16 weeks of G/P. Patients were followed for 24 weeks post-treatment to monitor safety and sustained virologic response.
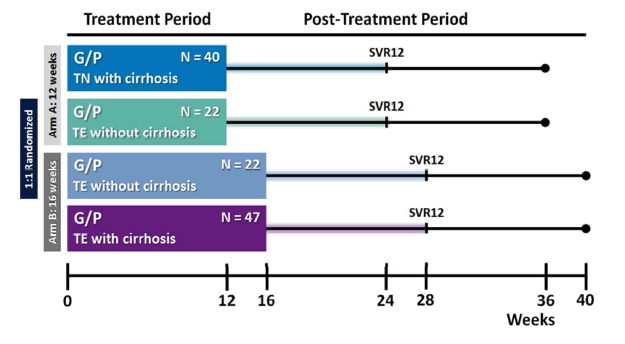
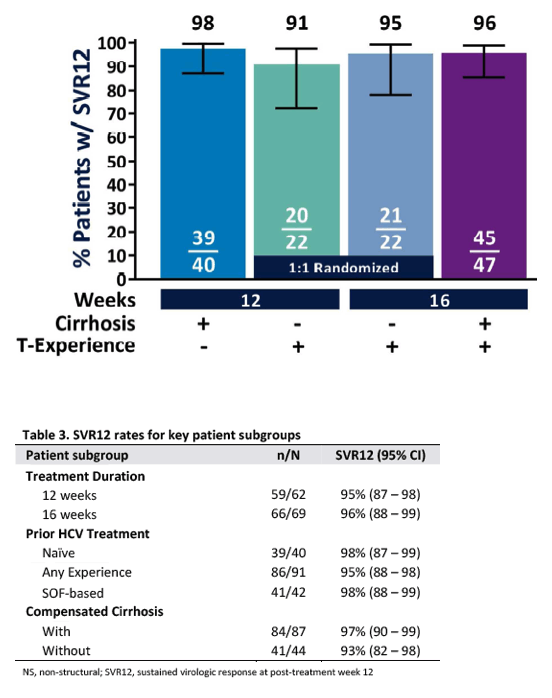
Figure 2. Sustained virologic response at post-treatment week 12
ABSTRACT
Background: This study assessed the efficacy and safety of ribavirin (RBV)-free coformulated glecaprevir/pibrentasvir (G/P) in patients with hepatitis C virus (HCV) genotype (GT) 3 infection with either prior treatment experience and/or compensated cirrhosis, a patient population with limited treatment options.
Methods: SURVEYOR-II, Part 3 was a partially-randomized, open-label, multicenter, phase 3 study. Treatment-experienced (prior interferon (IFN) or pegIFN ± ribavirin or SOF plus ribavirin ± pegIFN therapy) patients without cirrhosis were randomized 1:1 to receive 12 or 16 weeks of G/P (300 mg/120 mg) once daily. Treatment-naïve or treatment-experienced patients with compensated cirrhosis were treated with G/P for 12 or 16 weeks, respectively. The primary efficacy endpoint was the percentage of patients with sustained virologic response at post-treatment week 12 (SVR12). Safety was evaluated throughout the study.
Results: There were 131 patients enrolled and treated. Among treatment-experienced patients without cirrhosis, SVR12 was achieved by 91% (20/22; CI 72-97) and 95% (21/22; CI 78-99) of patients treated with G/P for 12 or 16 weeks, respectively. Among those with cirrhosis, SVR12 was achieved by 98% (39/40; CI 87-99) of treatment-naïve patients treated for 12 weeks, and 96% (45/47; CI 86-99) of patients with prior treatment experience treated for 16 weeks. No adverse events (AEs) led to discontinuation of study drug and no serious AEs were related to study drug.
Conclusions: Patients with HCV GT3 infection with prior treatment experience and/or compensated cirrhosis achieved high SVR12 rates following 12 or 16 weeks of treatment with G/P. The regimen was well tolerated.
Patient population. Patients were screened in the United States, Australia, Canada, France, New Zealand and the United Kingdom. Patients 18 years of age or older (no upper limit) were eligible if they had chronic HCV GT3 infection with HCV RNA >1000 IU/mL at screening. Patients were either treatment-naiÅNve or treatmentexperienced (previously treated with IFN or pegIFN Å} RBV or SOF + RBV Å} pegIFN regimens; those previously treated with NS5A or NS3/4A protease inhibitors were excluded). HCV genotype was assessed with the Versant® HCV Genotype Inno LiPA Assay, version 2.0 or higher, or Sanger sequencing of the NS5B region, if necessary. The absence of cirrhosis (e.g., METAVIR score ≤3, Ishak score ≤4) was determined by liver biopsy within 24 months prior to (or during) screening, transient elastography (eg, Fibroscan) score of <12.5 kPa within 6 months prior to (or during) screening, or a screening FibroTest score of ≤0.48 and an aspartate aminotransferase to platelet ratio index (APRI) <1. The presence of cirrhosis was delineated by prior histologic diagnosis of cirrhosis on liver biopsy (eg, METAVIR score of >3 [including 3/4], Ishak score of >4), prior transient elastography (eg, Fibroscan) score of ≥14.6 kPa, or a screening FibroTest score of ≥0.75 and an APRI >2. Indeterminate Fibroscan or FibroTest scores required qualifying liver biopsy or Fibroscan (to confirm indeterminate FibroTest scores only) to determine cirrhosis status. Patient exclusion criteria included coinfection with hepatitis B virus, human immunodeficiency virus, or non-GT3 HCV. Details on laboratory abnormality criteria (eg, alanine aminotransferase, platelets, and albumin), as well as all other inclusion and exclusion criteria, are shown in Supporting Table 1.
DISCUSSION
The patient population treated with G/P in Part 3 of SURVEYOR-II included some of the most difficult to treat patients with HCV infection, incorporating many current and historic negative predictors of response to HCV therapy; the treated population, including patients with HCV genotype 3, compensated cirrhosis, and prior treatment experience (including treatment with SOF-based regimens), was the largest such patient group to date.(10, 11, 14) Among patients with GT3 infection and compensated cirrhosis, treatment with 12 and 16 weeks of RBV-free G/P yielded high rates of SVR12 in treatment-niave and –experienced patients, respectively. Additionally, the regimen was well tolerated with no serious adverse events related to study drug and no adverse events leading to study drug discontinuation.
Treatment options for patients with HCV GT3 infection and compensated cirrhosis and/or prior treatment experience are currently limited and often require testing for baseline polymorphisms, and/or coadministration of RBV, or treatment duration of 24 weeks to maximize SVR12 rates. Due to higher rates of relapse observed with SOF/VEL treatment in GT3-infected patients with NS5A polymorphisms, particularly those with Y93H (20% [3/15] relapse rate(24)), pre-treatment testing for the presence of Y93H is recommended when using this regimen for patients with treatment experience and/or cirrhosis.(12, 13) Coadministration of RBV with SOF/VEL is recommended: if a patient has prior SOF-containing therapy experience, if Y93H is present upon testing,(12,13) or regardless of baseline polymorphism testing for patients with both treatment experience and cirrhosis.(12) Similarly, among GT3-infected patients with treatment experience and/or cirrhosis, RBV coadministration is recommended when using the combination of SOF plus DCV in patients with baseline Y93H or regardless of baseline polymorphism testing for patients with both prior treatment experience and compensated cirrhosis. Extended 24 week treatment duration of SOF/DCV is also recommended for all patients with cirrhosis.(10, 11) In vitro, the Y93H polymorphism results in a greater than 100-fold reduction in VEL susceptibility and more than 3700-fold reduction in DCV susceptibility.(24, 25) Similarly, susceptibility to VEL and DCV is reduced by 16-fold and 117-fold, respectively, in the presence of A30K.(25) In contrast, these single A30K or Y93H polymorphisms confer minimal changes to in vitro PIB susceptibility (less than 2.5-fold each).(18, 19)
In this study, treatment with G/P for 12 and 16 weeks achieved 95% or greater rates of SVR12 in treatment-naive and treatment experienced patients, respectively. While treatment-naive and treatment-experienced patients with cirrhosis were treated for 12 weeks and 16 weeks, respectively, treatment-experienced patients without cirrhosis were randomized to either 12 or 16 weeks of G/P treatment, achieving SVR12 rates of 91% (20/22) and 95% (21/22), respectively. The difference in SVR12 rate between 12 and 16 weeks of treatment was driven by a single additional relapse in the 12-week arm, making it difficult to determine the optimal treatment duration for this population based on these results alone. However, results from prior phase 2 studies in treatment-experienced GT3-infected patients without cirrhosis treated for 12 weeks demonstrated a similar tendency toward lower efficacy, with an SVR12 rate of 89% (24/27) with 2 relapses.(23) Notably, the 16-week regimen of G/P is now approved by the US FDA and EMA for the treatment of GT3-infected patients with prior treatment experience (with or without cirrhosis), supported by data from prior phase 2 studies and this phase 3 study, demonstrating an overall 96% (66/69) SVR12 rate in this subpopulation.(21, 22)
Amongst patients with baseline NS5A polymorphisms and compensated cirrhosis (n = 15; including 5 with Y93H at baseline), all patients achieved SVR12. Among treatment-experienced patients without cirrhosis, 3 patients had Y93H at baseline and 2/3 (67%) achieved SVR12; both patients with A30K at baseline had virologic failure.
With a limited sample size available and the low prevalence of these polymorphisms in this NS5A-inhibitor naive population, it is difficult to determine the true impact of such polymorphisms on treatment outcome. Prior studies have also reported low prevalence of both the Y93H (8.3-8.8%(11, 26)) and A30K (4.5-6%(26, 27)) polymorphisms in patients with HCV genotype 3. Moreover, as discussed, neither of these polymorphisms have a significant impact on in vitro susceptibility to pibrentasvir.(18) In support of these data, per the US FDA and EMA product use labels, GT3-infected patients with compensated cirrhosis and/or prior treatment experience do not require viral resistance testing prior to treatment with G/P.(21, 22)
Altogether, the efficacy results from this study demonstrate that G/P is an all-oral DAA regimen with high cure rates that does not require ribavirin coadministration in HCV GT3-infected patients, regardless of other traditional negative predictors of response such as prior treatment experience or compensated cirrhosis. Retreatment of the patients that had virologic failure to G/P therapy may require a regimen with more than two mechanisms of action, such as G/P plus SOF, which is a DAA combination currently being evaluated in an ongoing clinical study (registered clinical trial: NCT02939989).
Treatment with G/P was safe and well-tolerated, regardless of treatment duration or baseline patient characteristics. Previous-generation NS3/4A protease inhibitor-containing regimens have been associated with AEs, including severe rash, marked hyperbilirubinemia, neutropenia, anemia, and elevated ALT, particularly in patients with cirrhosis.(28)
However, all of these events were absent or rarely observed in SURVEYOR-II Part 3, even in patients with compensated cirrhosis. No patients discontinued or interrupted study drugs due to AEs. Moreover, there were no incidences of drug-induced liver injury or hepatic decompensation. Overall, the safety profile of patients with compensated cirrhosis was similar to those without cirrhosis. Serious AEs reported in this study were uncommon (n = 6), and none were deemed related to study drugs. The most commonly reported AEs were fatigue and headache, and they were reported at rates similar to placebo control groups in other clinical trials.(29, 30)
A potential limitation of this study was the relatively low patient numbers within individual subpopulations (e.g., treatment-experienced non-cirrhotics). However, these patient numbers are not inconsistent with other studies within similar subpopulations.(11, 14) Moreover, the largest cohort was patients with both treatment experience and cirrhosis (n = 47), with a sample size larger than evaluated in other studies; an SVR12 rate of 96% was observed in this more difficult-to-cure population after 16 weeks of G/P treatment. Finally, GT3 infected patients with prior exposure to NS5A inhibitor-containing regimens were not studied here; thus, the impact of treatment emergent NS5A resistance associated substitutions on the efficacy of G/P in this patient subpopulation is currently unknown.
In summary, SURVEYOR-II Part 3 enrolled and treated some of the most difficult-to-cure HCV patients: those with GT3 infection and prior treatment experience and/or cirrhosis. Overall, the fixed-dose combination of once daily RBV-free G/P was well-tolerated and demonstrated high SVR12 rates (≥95%) in treatment-naive patients with cirrhosis treated for 12 weeks, and treatment-experienced patients with or without cirrhosis treated for 16 weeks. Therefore, G/P provides an efficacious and well tolerated once-daily RBV-free treatment option for patients with HCV genotype 3 and prior treatment experience and/or cirrhosis.
| |
| |
| |
|
|
|