| |
IL-17 Treatment, Anti-Inflammatory in Diabetes/Oral Microbiome - Diabetes Enhances IL-17 Expression and Alters the Oral Microbiome to Increase Its Pathogenicity
|
| |
| |
Download the PDF here
"treatment with IL-17 antibody decreases the pathogenicity of the oral microbiota in diabetic mice.....inhibits inflammation through local injection of IL-17"
Diabetes Enhances IL-17 Expression and Alters the Oral Microbiome to Increase Its Pathogenicity
Highlights
⋅ Diabetes increases periodontal bone resorption and tooth loss in mice
⋅ Diabetic mice have increased periodontal inflammation and IL-17 levels
⋅ Diabetic oral microbiota induces periodontitis in wild-type germ-free recipients
⋅ Blocking IL-17 reduces the pathogenic effect of diabetic oral microbiota
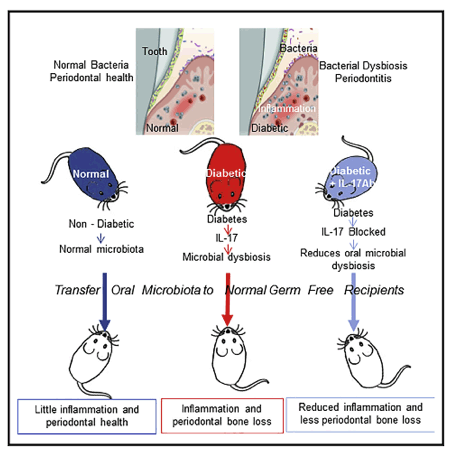
Summary
Diabetes is a risk factor for periodontitis, an inflammatory bone disorder and the greatest cause of tooth loss in adults. Diabetes has a significant impact on the gut microbiota; however, studies in the oral cavity have been inconclusive. By 16S rRNA sequencing, we show here that diabetes causes a shift in oral bacterial composition and, by transfer to germ-free mice, that the oral microbiota of diabetic mice is more pathogenic. Furthermore, treatment with IL-17 antibody decreases the pathogenicity of the oral microbiota in diabetic mice; when transferred to recipient germ-free mice, oral microbiota from IL-17-treated donors induced reduced neutrophil recruitment, reduced IL-6 and RANKL, and less bone resorption. Thus, diabetes-enhanced IL-17 alters the oral microbiota and renders it more pathogenic. Our findings provide a mechanistic basis to better understand how diabetes can increase the risk and severity of tooth loss.
Local injection of IL-17 altered the microbial composition and decreased the capacity of bacteria transferred from diabetic animals to stimulate periodontal inflammation, osteoclast formation, and periodontal bone loss in normal germ-free mice.
The state of chronic inflammation induced by diabetes could disrupt the homeostasis of the bacterial communities in favor of dysbiotic communities capable of inducing periodontal tissue destruction. Though several diabetes-associated taxa were unaffected by local injection of IL-17, the abundance of Enterococcus was altered between groups and may be a taxon associated with inflammation in the context of diabetes. These results indicate that high levels of IL-17 contribute to diabetes-induced microbial changes that, in turn, increase pathogenicity.
In summary, there is a complex interaction between diabetes, inflammation, the oral microbiome, and periodontal disease. We were able to dissect each component separately by inhibiting inflammation through local injection of IL-17 antibody and by examining oral microbial pathogenicity by transfer to normal germ-free recipients. These results suggest that the microbiome and the host response interact with each other and each contributes to diabetes-enhanced periodontal disease. This is based on previous findings that the same bacterial stimulus induces greater inflammation in diabetic animals compared to matched normoglycemic controls (Liu et al., 2006, Naguib et al., 2004, Wu et al., 2015) and findings here that transfer of microbes from diabetic mice stimulates greater inflammation and bone loss than transfer from normoglycemic donor mice. Moreover, inhibition of IL-17 establishes that the pathogenicity of the diabetic microbiome is affected by the level of inflammation. Thus, we suggest that diabetes enhances periodontal inflammation and periodontal inflammation causes a pathogenic change in the microbiome to significantly increase the susceptibility and severity of periodontal disease.
Introduction
Health is dependent on the homeostasis of both inner and external microenvironments. Diabetes mellitus is characterized by hyperglycemia, inflammation, and high oxidative stress, which leads to systemic complications (Wu et al., 2015). Type 2 diabetes is caused by insulin resistance frequently associated with obesity and a failure to produce enough insulin. The microbiome, an important component of the external microenvironment, plays a key role in homeostasis and affects several pathologic processes, including diabetes (Ussar et al., 2016), hematopoiesis (Khosravi et al., 2014), skeletal health (Hernandez et al., 2016), obesity (Sweeney and Morton, 2013), and carcinogenesis (Li et al., 2017). Thus, dysbiosis of the microbiome has a significant effect on health and disease (Sekirov et al., 2010).
The oral cavity is the initial point of entry to the digestive and respiratory tract. Over 700 bacterial species may be found in the oral cavity of humans (Paster et al., 2006). Oral microbial dysbiosis is linked to oral inflammation, is thought to trigger periodontal disease (Curtis et al., 2011, Hajishengallis et al., 2011), and may contribute to systemic conditions through bacteremia (Han and Wang, 2013). The oral microbiome may impact bacteria that colonize the gut microbiome (Nakajima et al., 2015). There is evidence that some species are more pathogenic than others; the dynamic balance of various bacteria that make up the oral microbiome has been proposed to determine periodontal disease activity (Colombo et al., 2012, Griffen et al., 2012, Hong et al., 2015).
Diabetes is a risk factor for periodontitis and increases disease severity (Emrich et al., 1991, Loe, 1993). Host factors are modified by diabetes and may play a causative role in bacteria-induced tissue damage (Andriankaja et al., 2012, Lalla and Papapanou, 2011, Pacios et al., 2012). Although some microbial studies show changes in the abundance of oral microorganisms in diabetes mellitus, there is no direct evidence that diabetes increases the pathogenicity of the oral microbiome (Ohlrich et al., 2010). A consensus report from the European Federation of Periodontology and the American Academy of Periodontology found that there is no compelling evidence that diabetes has a significant impact on the oral microbiota (Chapple et al., 2013). To investigate this important issue, we examined a mouse model and the impact of diabetes on the oral microbial composition and its pathogenicity.
|
|
| |
| |
|
|
|