| |
Johnson & Johnson at IAS 2017: Latest Innovations from its Janssen Pharmaceutical Companies Include First In-Human Data for Investigational Preventive HIV-1 Vaccine and Phase 3 Pivotal Trial Results for Darunavir-Based, Single-Tablet Regimen
|
| |
| |
First in-human, Phase 1/2a data from APPROACH preventive HIV-1 vaccine clinical trial to be featured in oral presentation and included in official IAS press program.
Interim results from the Phase 3 pivotal EMERALD trial investigating darunavir-based single-tablet regimen selected for oral presentation; data submitted to regulatory authorities for EMA approval, September 2017.
New Brunswick, NJ, 11 July 2017 - As part of its commitment to make HIV history, Johnson & Johnson today announced that the first in-human clinical trial results for the investigational human immunodeficiency virus (HIV-1) preventive vaccine regimen in development at its Janssen Pharmaceutical Companies will be presented at the 9th International AIDS Society (IAS) Conference on HIV Science on July 23-26 in Paris, France. The Phase 1/2a APPROACH study will report the clinical results on mosaic-based, heterologous prime-boost preventive vaccine regimens against HIV.
"The search for a vaccine to prevent HIV has been a 30-year quest. For more than a decade we have focused on bringing breakthrough science to reach the ultimate goal of ending this epidemic," said Paul Stoffels, MD, Executive Vice President, Chief Scientific Officer, Johnson & Johnson. "In parallel, we remain deeply focused on improving the lives of those living with HIV by continuing to develop effective and innovative therapies that reduce overall treatment burden."
Notable therapeutic innovations to be presented by Janssen include data from the Phase 3 pivotal EMERALD trial highlighting interim efficacy and safety results which support the ongoing regulatory submission preparation for a single-tablet regimen (STR) ofdarunavir/cobicistat/emtricitabine/tenofovir alafenamide (D/C/F/TAF). If approved, it would be the first darunavir-based STR indicated as a complete regimen for the treatment of human immunodeficiency virus type 1 (HIV‑1) infection in adults and adolescents (aged 12 years and older with body weight at least 40 kg), combining the proven efficacy and durability of darunavir with the improved renal and bone tolerability profile of F/TAF. It would also be the only treatment to deliver the adherence advantages of an STR coupled with the high genetic barrier to resistance that darunavir offers, providing added protection against the development of HIV drug resistance.
Further abstracts being presented will showcase data for both approved and investigational Janssen compounds for the treatment of HIV.
In addition to Janssen-sponsored abstracts, on Monday 24 July, 12:30-14:30 CEST (06:30-08:30 EDT) the International Partnership for Microbicides (IPM) will present Phase 3 pooled analysis data for the dapivirine vaginal ring as a late breaker poster. These data investigate the efficacy of the ring to reduce the risk of HIV infection in women in Sub-Saharan Africa and support the recent regulatory submission to the European Medicines Agency (EMA). In 2004, IPM were granted an exclusive, royalty-free license by Janssen Sciences Ireland UC to develop and commercialize dapivirine as a vaginal and rectal microbicide for HIV prevention in women.
An additional late-breaking oral presentation of unprecedented long-term data results from LATTE-2, the first study to analyze the efficacy and safety of a fully injectable two-drug (Janssen's rilpivirine with ViiV Healthcare's cabotegravir) long-acting antiretroviral therapy regimen in patients with HIV-1 infection, will be presented on Monday 24 July 2017 at 12:00 CEST (06:00 EDT). If approved, this first all-injectable regimen could offer a highly effective suppressive maintenance therapy for people living with HIV.
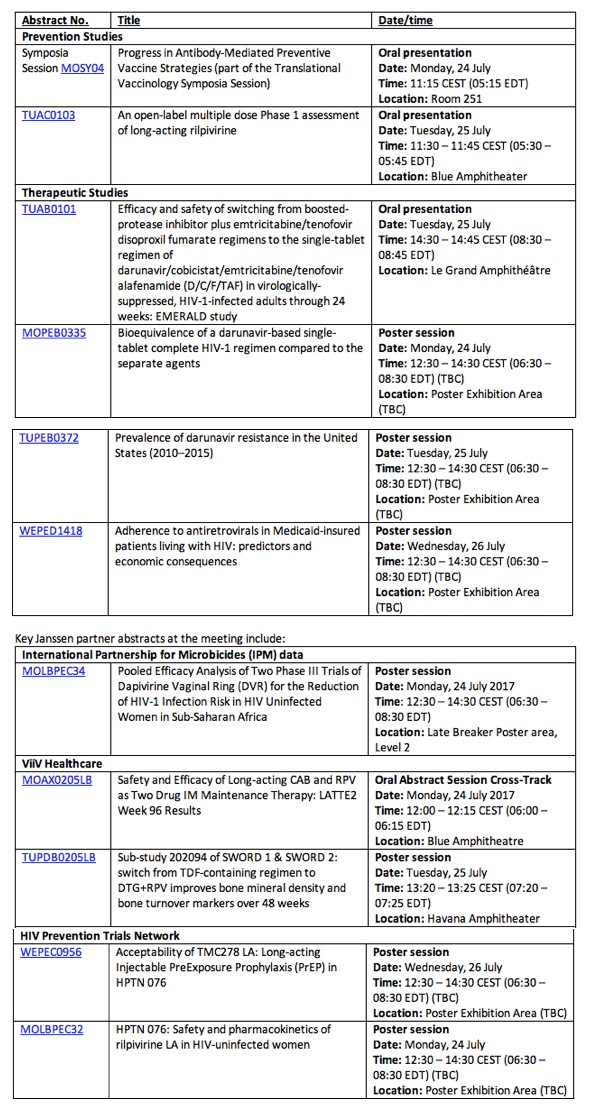
Key data presentations from our pipeline include:
HIV preventive vaccine - APPROACH data: Results from a Phase 1/2a study will provide the first look at in-human data for investigational "mosaic"-based, prime-boost regimens that are designed to elicit an immune response against a variety of HIV subtypes prevalent worldwide. The APPROACH study is evaluating the safety, tolerability and immunogenicity of heterologous vaccine regimens that contain two prime doses of a mosaic viral vectors based on Janssen's AdVac® technology (Ad26.Mos.HIV) and two boosts containing varying components depending on the regimen including Ad26.Mos.HIV, MVA-Mosaic, and the soluble protein Clade C gp140.
•These data will be presented at 11:15 CEST (05:15 EDT) on Monday 24 July. The presentation is entitled: "Progress in Antibody-Mediated Preventive Vaccine Strategies" and forms part of the Translational Vaccinology Symposia Session. The data will also be included in the official IAS press conference program.
Darunavir-based single-tablet regimen (D/C/F/TAF): Findings from the pivotal Phase 3 EMERALD trial will provide evidence in support of the first potential darunavir-based single-tablet regimen for the treatment of HIV-1. EMERALD investigated the efficacy and safety of switching from boosted-protease inhibitor plus emtricitabine/tenofovir disoproxil fumarate regimens to the single-tablet regimen of D/C/F/TAF in previously virologically-suppressed, HIV infected adults. [Abstract #TUAB0101]
•These data will be presented in an oral presentation at 14:30 CEST (08:30 EDT) on Tuesday 25 July.
Prevalence of darunavir resistance-associated mutations: Data presented assesses darunavir resistance, as measured by the prevalence of darunavir resistance-associated mutations (RAMs) in US clinical samples evaluated by PhenoSenseGT® commercial testing conducted by Monogram Biosciences. [Abstract #TUPEB0372].
• These data will be presented during a poster session at 12:30-14:30 CEST (06:30-08:30 EDT) on Tuesday 25 July.
Two-drug single-tablet regimen of dolutegravir (DTG) [ViiV Healthcare] and rilpivirine (RPV) [Janssen]: Data presented from a sub-study of SWORD 1 & SWORD 2 showing the impact on hip and spine bone mineral density and bone turnover markers following a switch from a TDF-containing regimen to DTG+RPV over 48 weeks. [Abstract #TUPDB0205LB].
• These data will be presented during a late breaker poster discussion session at 13:20 CEST (07:20 EDT) on Tuesday 25 July.
Further details on the breadth of science being presented by Johnson & Johnson companies and its partners will be released on thejnj.com/HIV microsite - coming soon.
Notes to editors
International Partnership for Microbicides (IPM)
IPM is an organization dedicated to providing women - who as a group bear a burden of HIV infection globally - with HIV-prevention strategies, both empowering them to take care of their own health, and to prevent HIV transmission to their children.
About Johnson & Johnson Caring for the world, one person at a time, inspires and unites the people of Johnson & Johnson. We embrace research and science - bringing innovative ideas, products and services to advance the health and well-being of people. Our approximately 127,100 employees at more than 250 Johnson & Johnson operating companies work with partners in health care to touch the lives of over a billion people every day, throughout the world.
About Janssen
At the Janssen Pharmaceutical Companies of Johnson & Johnson, we are working to create a world without disease. Transforming lives by finding new and better ways to prevent, intercept, treat and cure disease inspires us. We bring together the best minds and pursue the most promising science. We are Janssen. We collaborate with the world for the health of everyone in it. Learn more at www.janssen.com and follow us at @JanssenGlobal.
Cautions Concerning Forward-Looking Statements
This press release contains "forward-looking statements" as defined in the Private Securities Litigation Reform Act of 1995 regarding development of potential preventive and treatment regimens for HIV. The reader is cautioned not to rely on these forward-looking statements. These statements are based on current expectations of future events. If underlying assumptions prove inaccurate or known or unknown risks or uncertainties materialize, actual results could vary materially from the expectations and projections of the Janssen Pharmaceutical Companies and Johnson & Johnson. Risks and uncertainties include, but are not limited to: challenges and uncertainties inherent in product development, including uncertainty of clinical success and obtaining regulatory approvals; uncertainty of commercial success for new indications and therapeutic combinations; competition, including technological advances, new products and patents attained by competitors; challenges to patents; product efficacy or safety concerns resulting in product recalls or regulatory action; changes in behaviour and spending patterns of purchasers of health care products and services; changes to applicable laws and regulations, including global health care reforms; and trends toward health care cost containment. A further list and description of these risks, uncertainties and other factors can be found in Johnson & Johnson's Annual Report on Form 10-K for the year ended January 1, 2017, including under "Item 1A Risk Factors," its most recently filed Quarterly Report on Form 10-Q, including in the section captioned "Cautionary Note Regarding Forward-Looking Statements," and the company's subsequent filings with the Securities and Exchange Commission. Copies of these filings are available online at www.sec.gov, www.jnj.com or on request from Johnson & Johnson. None of the Janssen Pharmaceutical Companies or Johnson & Johnson undertakes to update any forward-looking statement as a result of new information or future events or developments.
Press Contacts:
Ronan Collins
Rcollin5@its.jnj.com
+47 488 42500
Katie Buckley
Kbuckle8@its.jnj.com
+ 44 7971956 179
Gavin Hart
+1 917 686 9221
Ghart9@its.jnj.com
INVESTOR RELATIONS:
Lesley Fishman
+1 732 524 3922
Joseph J. Wolk
+ 1 732 524 1142
|
|
| |
| |
|
|
|