| |
Fatty Liver in HIV+ / Changes in Liver Steatosis After Switching From Efavirenz to Raltegravir Among Human Immunodeficiency Virus-Infected Patients With Nonalcoholic Fatty Liver Disease
|
| |
| |
Download the PDF here
-------------
from Jules: the main concern is that fatty liver [steatosis] can lead to advanced liver disease [without having HCV, HBV or excessive alcohol consumption]....BMI, metabolic syndrome & history use of certain ARTs can increase fatty liver and risk for developing liver disease, Fibroscan is used to evaluate liver disease (LSM) & fatty liver (CAP score). HIV+ have higher rates of fatty liver than HIV-negatives. It's estimated that fatty liver is present in from 35-65% of HIV-infected and appears to be higher compared to HIV-uninfected. Earlier research tied older NRTIs to fatty liverdisease apparently as discussed below due to mitochondrial toxicity, but HIV itself also caan cause mitochondrial toxicity. What is contribution of other ART classes. Well the study reported here discusses how efavirenz can cause mitochondrial toxicity and is associated with fatty liver and they found the integrase raltegravir improved fatty liver in HIV+ who switch to raltegravir from efavirenz. What about all other ARTs, a study linked to below from IAS found duration of cART associated with fatty liver. Clearly HIV+ appear at greater risk for fatty liver and therefore at greater risk for liver disease and fibrosis associated with fatty liver vs HIV-negatives. This is another aging/HIV related issue as most of the older HIV+ aging have experience with old and new nukes and many of the other ART drug classes. The METAFIB study at IAS found 25% of HIV monoinfected with metabolic syndrome had fairly advanced liver disease (F2/3 fibrosis) and 8% or more had cirrhosis. The 2nd study at IAS in HIV monoinfected found 9% or more had advanced liver disease. So HIV+ even without HCV or HBV can have fatty liver and liver disease, and HIV, CART duration, abnormal metabolics & lipids/diabetes can drive this.
NATAP Section dedicated to Fatty Liver http://www.natap.org/liver.htm
Fatty Liver, NAFLD, NASH Definitions - (06/29/17)
Current and Future Therapeutic Approaches to NAFLD/NASH - (08/05/17)
IAS: Fatty Liver in HIV+ at IAS - (08/05/17)
Fatty Liver / NASH at EASL - (07/03/17)
IAS: Metabolic syndrome and obesity are the cornerstones of liver fibrosis in HIV-monoinfected patients: results of the METAFIB study - (07/25/17)
IAS: Predictor factors associated with liver fibrosis and steatosis by transient elastography in HIV mono-infected patients under long-term combined antiretroviral therapy - (07/26/17)
CROI: FATTY LIVER DISEASE: A GROWING CONCERN - - (04/12/17) a plenary talk by noted fatty liver expert Rohit Loomba from UCSD
CROI: Changes in Liver Fibrosis and Steatosis in HIV Mono-Infected patients over 24 months - 50% have fatty liver at average age of 46 - (03/28/17)
CROI: LIVER STEATOSIS AND FIBROSIS IN AT-RISK EUROPEAN HIV-MONOINFECTED PATIENTS - 64% with steatosis among those who had elevated LFTs and/or metabolic syndrome and/or lipodystrophy - (03/28/17)
IAS: Predictor factors associated with liver fibrosis and steatosis by transient elastography in HIV mono-infected patients under long-term combined antiretroviral therapy - (07/26/17)
Its estimated 30-40% of HIV+ have fatty liver (NAFLD) and studies suggest these rates may be higher than in HIV-negatives. BUT here is a study finding 67% fatty liver in HCV/HIV coinfected .....67% Steatosis in HCV/HIV Coinfected: : Liver inflammation, HCV genotype 3, and BMI are associated with steatosis, a common finding in HCV-HIV-coinfected patients http://www.natap.org/2007/HIV/062007_02.htm
------------------
Changes in Liver Steatosis After Switching From Efavirenz to Raltegravir Among Human Immunodeficiency Virus-Infected Patients With Nonalcoholic Fatty Liver Disease
CID July 31 2017 -Juan Macias,1 Maria Mancebo,1 Dolores Merino,2 Francisco Tellez,3 M. Luisa Montes-Ramirez,4 Federico Pulido,5 Antonio Rivero-Juarez,6Miguel Raffo,7 Montserrat Perez-Perez,7 Nicolas Merchante,1 Manuel Cotarelo,8 and Juan A. Pineda1; for the Spanish AIDS Research Network-HEP09Study Group
"In conclusion, replacement of EFV by RAL among individuals with significant hepatic steatosis led to reductions in the grade of hepatic steatosis, even reversal of fatty liver in some cases, after 48 weeks. This effect may be related to the discontinuation of EFV and its potential mitochondrial toxicity, plus the metabolic-friendly properties of RAL. These results need confirmation in a larger study, also including drugs commonly used currently (eg, dolutegravir, elvitegravir, and protease inhibitors). Finally, studies using noninvasive diagnostic tools that enable the accurate identification of steatohepatitis are also desirable.....The difference in outcomes in this trial was due to both increases in CAP levels in the EFV group and decreases in CAP values in the RAL arm. In vitro data indicate that EFV can induce hepatic steatosis [13].....Concerning RAL, conversely to EFV, it does not alter the mitochondrial function of hepatic cells in vitro [14]......In addition, RAL was associated with a trend to lower frequency of fatty liver measured by CAP in a cross-sectional study, similar to other metabolic-friendly drugs such as maraviroc or nevirapine [3]....The reduction in CAP values and hence the lower rate of significant hepatic steatosis in patients switched from EFV to RAL may be explained by different mechanisms......EFV could induce hepatic steatosis through mitochondrial toxicity....the more friendly metabolic profile of RAL might have contributed to reversal of fatty liver.....This in vitro effect could account for the increased risk of lipodystrophy associated with EFV, and it may also be involved in hepatic steatosis. In this regard, EFV enhances fatty accumulation in hepatocyte cell lines in vitro [13, 14]. In the herein-reported trial, patients continuing on EFV showed increasing CAP measurements. In this way, lack of mitochondrial damage induced by RAL could have allowed the reductions in CAP values after stopping the mitochondrial toxicity of EFV.....In a recent survey evaluating the prevalence of significant hepatic steatosis using CAP among HIV-infected patients, metabolic factors were the strongest predictors of fatty liver [3]. Hepatic steatosis was less likely among those exposed to RAL, maraviroc, or nevirapine [3]. However, BMI was the only independent predictor of significant hepatic steatosis in the multivariate analysis
The CAP cutoff value of 238 dB/m exhibits high positive and negative predictive values for significant hepatic steatosis [23], and this cutoff has been validated in a prospective study [27]....Given that progression of fibrosis and the emergence of cirrhosis seem tightly linked with steatohepatitis, further studies evaluating the influence of antiretroviral drugs on steatohepatitis as end-point are needed."
INTRODUCTION - Nonalcoholic fatty liver disease (NAFLD) is frequently observed in human immunodeficiency virus (HIV)-infected patients [1-4]. NAFLD itself can promote fibrosis progression, evolving to steatohepatitis, on which cirrhosis may develop and hepatocellular carcinoma emerge [5-7]. Hepatic steatosis is linked with metabolic disorders and exposure to certain antiretroviral drugs [1-4, 8-11]. The main potential mechanism involved in antiretroviral drug-related hepatic steatosis seems to be mitochondrial toxicity. Thus, the use of nucleoside analogues associated with more severe mitochondrial toxicity, such as dideoxynucleoside analogues, has been linked with hepatic steatosis [1, 8-11]. Efavirenz (EFV) has been related with clinical manifestations of mitochondrial toxicity, such as lipoatrophy [12]. Likewise, EFV enhances fatty accumulation in hepatocyte cell lines in vitro [13]. Moreover, the cumulative use of EFV seemed to be associated with an increased risk of hepatic steatosis progression in HIV/hepatitis C virus (HCV)-coinfected patients [8].
Given the potential of NAFLD for causing advanced liver disease, the effects of antiretroviral drug regimens less likely to induce increases in hepatic steatosis need to be investigated. Drug combinations based on raltegravir (RAL) may fulfill that requirement. Thus far, RAL does not induce mitochondrial toxicity [14] and has demonstrated a safe metabolic profile [14-20]. Cross-sectional and longitudinal observational data indicate that metabolic factors are the strongest predictors of hepatic steatosis in HIV infection [3, 4, 21]. In addition, use of RAL was associated with a trend to lower frequency of hepatic steatosis [3] and lesser likelihood of progression of hepatic steatosis [21], but these associations did not stand up after controlling for metabolic factors. Therefore, the role of RAL preventing the progression of hepatic steatosis, which could be driven by its metabolic safety profile, needs to be clarified in controlled, prospective studies. Because of these issues, in this trial we aimed at comparing the impact of switching from EFV to RAL in patients receiving EFV plus 2 nucleoside analogues vs continuing with the same therapy on hepatic steatosis as measured by controlled attenuation parameter (CAP) among HIV-infected patients with NAFLD.
Changes in Controlled Attenuation Parameter Values
At baseline, the median CAP values were 273 (Q1-Q3, 246-303) dB/m for the RAL group and 263 (Q1-Q3, 245-294) dB/m for the EFV group (P = .607). At week 48, the median CAP for patients switching to RAL was 250 (Q1-Q3, 221-277) dB/m and 286 (Q1-Q3, 269-314) dB/m for those continuing on EFV (P = .035; Figure 2A). The median difference in CAP measurements between baseline and week 48 was -20 (Q1-Q3, -67 to 15) dB/m among individuals in the RAL arm and 30 (Q1-Q3, -17 to 49) dB/m for patients in the EFV group (P = .011; Figure 2B). CAP values <238 dB/m, indicative of absence of significant hepatic steatosis, were observed in 9 (47%) patients in the RAL group and 3 (15%) individuals in the EFV group at week 48 (P = .029).
After excluding patients with ABC/3TC backbone, the median CAP after 48 weeks of follow-up for patients starting RAL was 237 (Q1-Q3, 221-274) dB/m and 286 (Q1-Q3, 267-315) dB/m for those continuing on EFV (P = .025).
Median liver stiffness at week 48 was 6.3 (Q1-Q3, 5.6-12) kPa for the RAL group and 5.9 (Q1-Q3, 4.5-9.3) kPa for the EFV group (P = .550). The median of the change in liver stiffness between baseline and week 48 was -0.3 (Q1-Q3, -1.9 to 1.7) kPa for patients in the RAL arm and 0.05 (Q1-Q3, -0.78 to 2.1) kPa for individuals in the EFV arm (P = .351).
In an analysis restricted to the 26 patients with detectable HCV RNA, the baseline median CAP values were 277 (Q1-Q3, 242-305) dB/m for the RAL group and 262 (Q1-Q3, 245-285) dB/m for the EFV group (P = .432). At week 48, the median CAP was 266 (Q1-Q3, 221-301) dB/m for patients switching to RAL and 285 (Q1-Q3, 270-316) dB/m for those continuing on EFV (P = .118). The median difference in CAP measurements between baseline and week 48 was -7 (Q1-Q3, -67 to 26) dB/m among individuals in the RAL arm and 30 (Q1-Q3, -3.5 to 49) dB/m for patients in the EFV group (P = .019). CAP values <238 dB/m, indicative of absence of significant hepatic steatosis, were observed in 5 (36%) patients in the RAL group and 1 (8.3%) individuals in the EFV group at week 48 (P = .099).
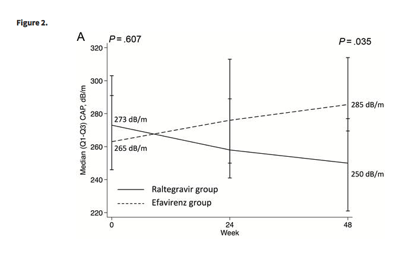
Metabolic Parameters and Controlled Attenuation Parameter Changes
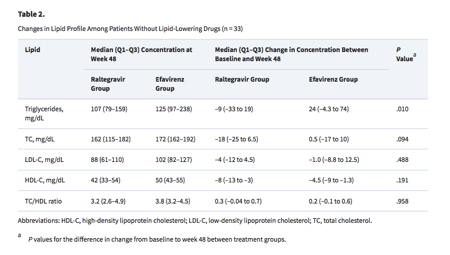
At week 48, the median BMI for individuals switched to RAL compared with those continuing on EFV was 27.6 (Q1-Q3, 24.3-31) kg/m2 vs 25.5 (Q1-Q3, 23.8-29.9) kg/m2 (P = .084). Weight increase from baseline to week 48 was a median of 1.2 (Q1-Q3, -2.0 to 6.2) kg in patients on RAL and 0.5 (Q1-Q3, -1.5 to 2.5) kg in those on EFV (P = .284). The median waist-to-hip ratio was 0.97 (Q1-Q3, 0.91-1.0) for patients in the RAL group and 0.95 (Q1-Q3, 0.9-1) for those in the EFV group at week 48 (P = .682). Among individuals without lipid-lowering drugs, total cholesterol and triglycerides showed greater decreases from baseline among patients switched to RAL (Table 2).
At week 48, the median CAP of patients with and without increases in BMI ≥0.5 kg/m2 was 282 (Q1-Q3, 267-316) dB/m and 237 (Q1-Q3, 213-292) dB/m, respectively (P = .013). Individuals with BMI increase experienced a median CAP raise of 27 (Q1-Q3, -8.5 to 44) dB/m compared with -33 (Q1-Q3, -71 to 21) dB/m for those without BMI increase (P = .014). Among patients not exposed to lipid-lowering drugs, those with any decrease in triglyceride levels between baseline and week 48 showed a median change in CAP of -79 (Q1-Q3, -112 to 2.8) dB/m, whereas the median difference in CAP values between baseline and week 48 was 26 (Q1-Q3, -18 to 33) dB/m for those with increases in triglyceride levels (P = .005).
DISCUSSION
After 48 weeks, HIV-infected individuals with NAFLD switched from an EFV- to a RAL-based combination showed a decrease in the degree of hepatic steatosis, as measured by CAP, compared with those continuing with an EFV-based regimen. In addition, the proportion of patients showing regression in significant hepatic steatosis after 48 weeks was greater in those who switched from EFV to RAL.
The difference in outcomes in this trial was due to both increases in CAP levels in the EFV group and decreases in CAP values in the RAL arm. In vitro data indicate that EFV can induce hepatic steatosis [13]. In a prior study conducted in HIV/HCV-coinfected patients, progression of hepatic steatosis was related to cumulative exposure to EFV [8], so that the longer the time on EFV, the higher the frequency of patients with fatty liver progression, a behavior similar to the effect of dideoxynucleosides on hepatic steatosis [8]. However, in the above-mentioned study, the effect of EFV on hepatic steatosis progression could have been confounded by dideoxynucleoside concomitant use. Here we found that EFV plus the nucleoside analogues currently used, TDF/FTC or ABC/3TC, led to elevations in values of CAP with time. Concerning RAL, conversely to EFV, it does not alter the mitochondrial function of hepatic cells in vitro [14]. In addition, RAL was associated with a trend to lower frequency of fatty liver measured by CAP in a cross-sectional study, similar to other metabolic-friendly drugs such as maraviroc or nevirapine [3]. In a longitudinal study, patients using RAL during a 12-month period of observation showed a significant decrease in CAP measurements [21]. However, these observational data were subject to selection bias in patients with RAL, due to previous metabolic toxicities or concomitant combination of RAL with ritonavir-boosted protease inhibitors. In this controlled clinical trial, where this bias is eliminated, we found that RAL can prevent the progression of hepatic steatosis and even decrease the frequency of fatty liver determined by CAP.
The reduction in CAP values and hence the lower rate of significant hepatic steatosis in patients switched from EFV to RAL may be explained by different mechanisms. First, the more friendly metabolic profile of RAL might have contributed to reversal of fatty liver. In this regard, patients starting TDF/FTC plus RAL show fewer increases in the blood levels of low-density lipoprotein cholesterol, total cholesterol-to-HDL ratio, and triglycerides than individuals starting TDF/FTC plus EFV [16, 17]. Similarly, switching from a boosted-ritonavir protease inhibitor-based regimen to RAL-containing combinations is associated with improvements in the lipid profile [18, 19, 24]. Substitution of EFV by RAL had also a significant and favorable impact on lipid levels in a prior trial [20]. Similar changes in the lipid profile were found in the present study, with a greater effect on triglycerides. In addition, triglyceride level decreases between baseline and week 48 were associated with reductions in CAP values in the herein-reported trial. Second, the discontinuation of EFV itself may also have contributed to the decrease in CAP values. EFV could induce hepatic steatosis through mitochondrial toxicity [25]. In cell cultures of human adipocytes, EFV impairs the morphological adipogenic differentiation and adipogenic gene expression in a dose-dependent fashion [26]. This in vitro effect could account for the increased risk of lipodystrophy associated with EFV, and it may also be involved in hepatic steatosis. In this regard, EFV enhances fatty accumulation in hepatocyte cell lines in vitro [13, 14]. In the herein-reported trial, patients continuing on EFV showed increasing CAP measurements. In this way, lack of mitochondrial damage induced by RAL could have allowed the reductions in CAP values after stopping the mitochondrial toxicity of EFV.
In a recent survey evaluating the prevalence of significant hepatic steatosis using CAP among HIV-infected patients, metabolic factors were the strongest predictors of fatty liver [3]. Hepatic steatosis was less likely among those exposed to RAL, maraviroc, or nevirapine [3]. However, BMI was the only independent predictor of significant hepatic steatosis in the multivariate analysis. In a Canadian cross-sectional study, BMI was also the strongest predictor of significant hepatic steatosis as measured by CAP [4]. Similar findings were observed in a longitudinal study on short-term changes in hepatic steatosis measured by CAP [21]. Thus, increases in BMI were the only independent factor related with rise in CAP values. In the present trial, BMI and CAP measurements paralleled; that is, increases in BMI during the follow-up were related with higher CAP values at week 48. Patients who switched to RAL showed greater BMI, close to statistical significance, than those continuing with EFV at week 48. In spite of this and the correlation between BMI and CAP, individuals randomized to replace EFV by RAL presented lower CAP values at week 48. This finding suggests that the effects of EFV substitution by RAL on fatty liver could be independent of BMI, and related with both the interruption of a drug with mitochondrial toxicity and the favorable metabolic profile of RAL. It is not known whether the effect on hepatic steatosis of switching EFV by RAL might also be observed with other HIV integrase inhibitors. If improvements in fatty liver after discontinuing EFV were related with stopping EFV toxicity, then switching to another HIV integrase inhibitor could lead to reductions in hepatic steatosis similar to those obtained in the present trial, provided that switching to other HIV integrase inhibitors was followed by similar effects of the lipid profile as those seen after switching to RAL.
This clinical trial was initially designed to evaluate changes in CAP values among patients with active HCV infection. The availability of highly efficacious all-oral treatment against HCV meant that most HIV/HCV-coinfected patients were eligible for anti-HCV therapy and, thus, not candidates for the trial. As a consequence, a major amendment was introduced to allow the inclusion of patients without active HCV coinfection. In spite of this, individuals with detectable plasma HCV RNA represented more than two-thirds of the study population. Because factors associated with hepatic steatosis among HIV-infected patients with and without HCV coinfection were similar [3, 21], we expected comparable fatty liver changes after switching EFV by RAL in HIV-infected patients with and without active HCV infection. In the present study, an analysis of CAP changes restricted to patients with detectable plasma HCV RNA yielded similar results to the overall population analysis. Thus, it is highly likely that the results reported herein can be generalized to HIV-infected patients without HCV coinfection.
This study may have some limitations. First, the trial was interrupted before reaching the planned sample size. However, in an intermediate analysis the difference in CAP values at week 48 between groups was larger than the initial estimation. This allowed us to detect a statistically significant difference in CAP values between groups at week 48 with a smaller sample size. Nevertheless, these trial results should be regarded as exploratory and preliminary. Second, we used CAP instead liver biopsy, which can be considered the gold standard for the diagnosis of NAFLD. However, the use of CAP to grade hepatic steatosis has been validated in different populations using liver biopsy as reference [23, 27-31]. The CAP cutoff value of 238 dB/m exhibits high positive and negative predictive values for significant hepatic steatosis [23], and this cutoff has been validated in a prospective study [27]. In addition, studies evaluating switching antiretroviral drugs with repeated determinations of hepatic steatosis among HIV-infected patients might not be feasible using an invasive technique such as liver biopsy. Third, CAP allowed us to evaluate changes in fatty liver, but steatohepatitis cannot be ascertained using CAP. Given that progression of fibrosis and the emergence of cirrhosis seem tightly linked with steatohepatitis, further studies evaluating the influence of antiretroviral drugs on steatohepatitis as end-point are needed.
In conclusion, replacement of EFV by RAL among individuals with significant hepatic steatosis led to reductions in the grade of hepatic steatosis, even reversal of fatty liver in some cases, after 48 weeks. This effect may be related to the discontinuation of EFV and its potential mitochondrial toxicity, plus the metabolic-friendly properties of RAL. These results need confirmation in a larger study, also including drugs commonly used currently (eg, dolutegravir, elvitegravir, and protease inhibitors). Finally, studies using noninvasive diagnostic tools that enable the accurate identification of steatohepatitis are also desirable.
|
|
| |
| |
|
|
|