 |
 |
 |
| |
A Randomized Trial of Bictegravir or Dolutegravir With Emtricitabine and Tenofovir Alafenamide (F/TAF) Followed by Open-label Switch to Bictegravir/F/TAF Fixed-Dose Combination - High Virologic Response Rates With Bictegravir or Dolutegravir Plus FTC/TAF
|
| |
| |
High Virologic Response Rates With Bictegravir or Dolutegravir Plus FTC/TAF
IDWeek2017/IDSA, October 4-8, 2017, San Diego
Mark Mascolini
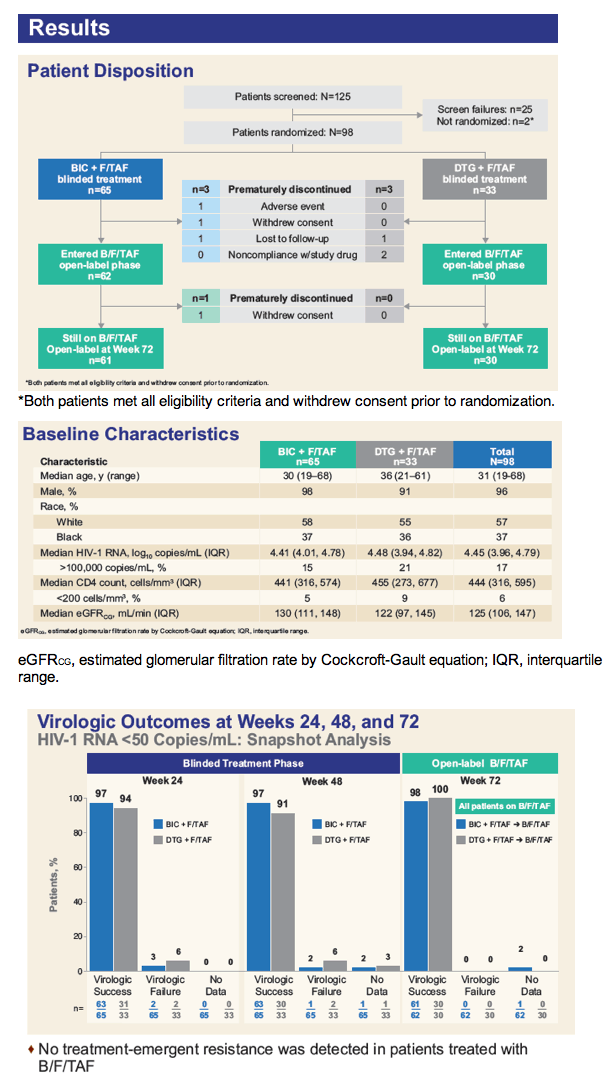
More than 90% of previously untreated adults randomized to either bictegravir or dolutegravir (plus emtricitabine/tenofovir alafenamide, FTC/TAF) had a viral load below 50 copies after 48 weeks of treatment in the blinded segment of a phase 2 trial [1]. Twelve weeks after everyone switched to single-tablet bictegravir/FTC/TAF at week 48, 91 of 92 participants maintained a sub-50-copy viral load.
Bictegravir is a once-daily integrase inhibitor being formulated as a single-tablet regimen with FTC/TAF. It has the longest plasma half-life among HIV integrase inhibitors, about 18 hours. This phase 2 trial aimed to compare once-daily bictegravir with dolutegravir, both given with separate FTC/TAF, through 48 weeks in antiretroviral-naive adults. Everyone still in the trial at week 48 could switch to the single-tablet formulation of bictegravir/FTC/TAF.
The study enrolled antiretroviral-naive adults and randomized them 2-to-1 to 75 mg of bictegravir (plus dolutegravir placebo) or 50 mg of dolutegravir (plus bictegravir placebo) plus 200/25 mg of FTC/TAF, all once daily. The 65 people randomized to bictegravir and the 33 randomized to dolutegravir had median ages of 30 and 36. Overall, 96% of participants were men, 57% black, and 37% white, with little difference between study arms. Median pretreatment viral load stood at 4.45 log10 (about 28,000 copies).
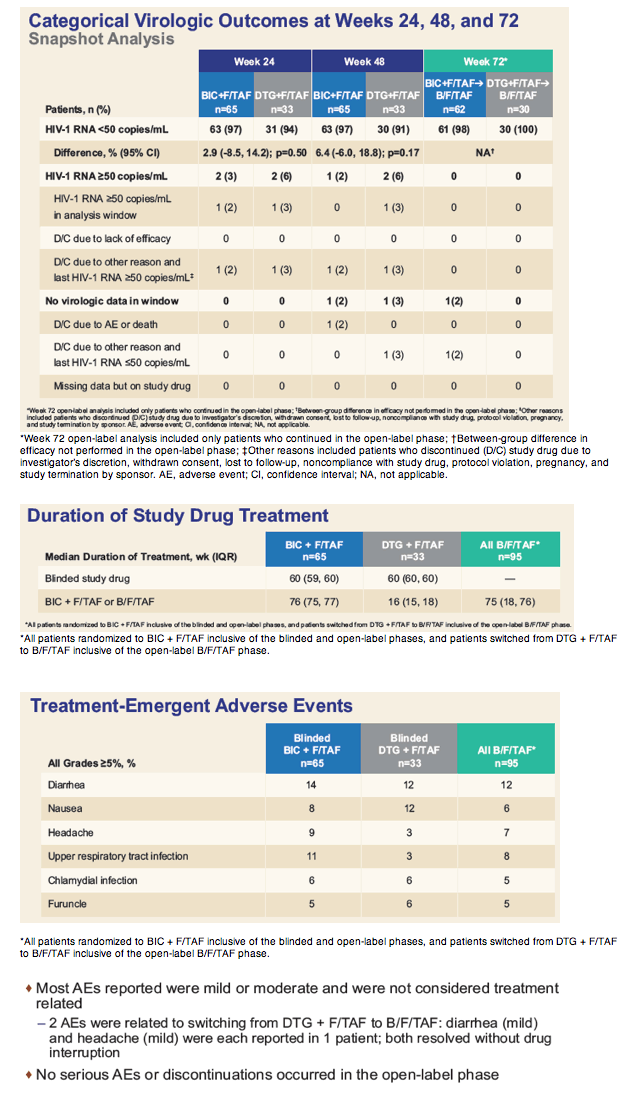
FDA snapshot analysis determined that 97% assigned to bictegravir and 94% randomized to dolutegravir had a viral load below 50 copies at week 24. Respective sub-50-copy rates at week 48 were 97% and 91%. Among 92 participants who completed week 48 and began single-tablet bictegravir/FTC/TAF, 91 (99%) maintained an undetectable viral load through week 72. One person withdrew before the week-72 analysis. No one stopped treatment because of lack of efficacy. Resistance to study drugs emerged in no one treated with bictegravir/FTC/TAF.
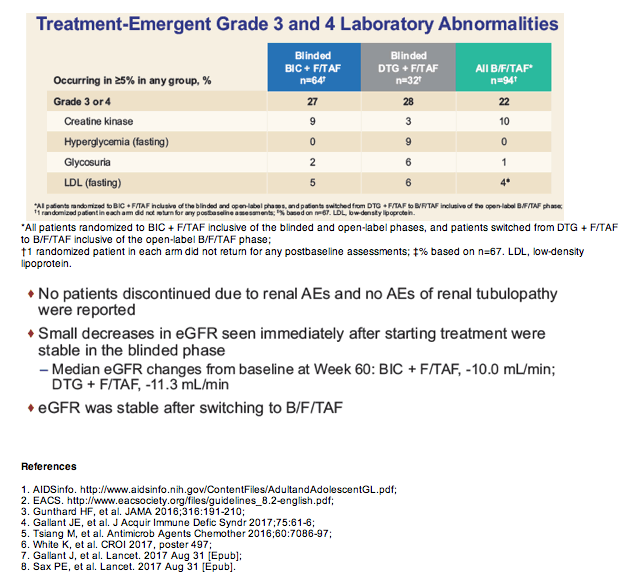
One person taking bictegravir stopped treatment after week 24 because of urticaria (hives). No one had a treatment-related serious adverse event. The most frequent adverse events were diarrhea, nausea, and headache. Median estimated glomerular filtration rate (eGFR, a kidney function marker) dropped 10 mL/min through week 60 in the bictegravir group and 11.3 mL/min in the initial-dolutegravir group. eGFR remained stable when people originally assigned to dolutegravir switched to bictegravir.
Three ongoing phase 3 trials are evaluating switching to bictegravir/FTC/TAF versus staying with a suppressive regimen including (1) a boosted protease inhibitor, (2) dolutegravir/abacavir/lamivudine, or (3) dolutegravir plus FTC/TAF.
Reference
1. Sax P, DeJesus E, Crofoot G, et al. A randomized trial of bictegravir or dolutegravir with emtricitabine and tenofovir alafenamide (F/TAF) followed by open label switch to bictegravir/F/TAF fixed dose combination. IDWeek2017/IDSA. October 4-8, 2017. San Diego. Abstract 1380.
------------------------------------
A Randomized Trial of Bictegravir or Dolutegravir With Emtricitabine and Tenofovir Alafenamide (F/TAF)
Followed by Open-label Switch to Bictegravir/F/TAF Fixed-Dose Combination
Paul E. Sax,1 Edwin DeJesus,2 Gordon Crofoot,3 Douglas Ward,4 Paul Benson,5 Xuelian Wei,6 Kirsten White,6 Sean E. Collins,6 Hal Martin,6 Andrew Cheng,6 Erin Quirk6
1Brigham and Women's Hospital and Harvard Medical School, Boston, MA; 2Orlando Immunology Center, FL; 3Gordon E. Crofoot MD, PA, Houston, TX; 4Dupont Circle Physicians Group, Washington, DC; 5Be Well Medical Center, Berkley, MI; 6Gilead Sciences, Inc., Foster City, CA


|
| |
|
 |
 |
|
|