 |
 |
 |
| |
Should Guidelines Shorten Virologic Failure Cutoff for Integrase Inhibitors?
|
| |
| |
IDWeek2017/IDSA, October 4-8, 2017, San Diego
"Current guidelines define virologic failure as HIV VL >200 after 24 weeks on ART, but this research suggests that patients on INSTI-based ART regimens should be suspected as having virologic treatment failure if they have not achieved viral suppression as early as 12 weeks after ART initiation. This has important implications for clinical practice, particularly in situations
where patients would particularly benefit from shorter time to viral suppression, e.g. pregnancy."
Mark Mascolini
Because people beginning antiretroviral therapy (ART) in 2013-2016 had a significantly shorter time to viral suppression when starting an integrase inhibitor, Yale University researchers suggested virologic failure with this class might be reset from a viral load above 200 copies after 24 weeks to a 200-plus load after 12 weeks [1]. The authors also proposed that integrase inhibitors may be the best choice for people who would benefit from quick viral suppression--like pregnant women.
Clinical trials comparing integrase inhibitors with protease inhibitors (PIs) or nonnucleosides routinely found faster viral suppression in people randomized to the integrase class. To determine whether the same pattern applies in clinical practice, researchers retrospectively reviewed charts of antiretroviral-naive people starting ART in 2013-2016 at the Yale-New Haven Health Facilities. All eligible participants had documented pretreatment and on-treatment viral loads. The Yale team compared rates of achieving viral suppression, defined as a viral load below a detection limit of 20 copies.
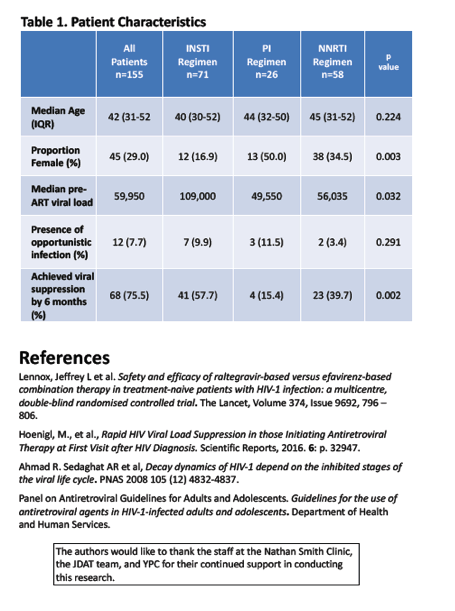
This preliminary analysis involved 155 people, 71% of them men. The PI group included a higher proportion of women (50%) than the nonnucleoside group (34.5%) or the integrase inhibitor group (17%) (P = 0.003). Age averaged 41.3 years overall, and pretreatment viral load 293,974 copies. Only 12 people (7.7%) had an opportunistic infection when they started ART, with no significant differences between the three treatment groups. While 46% started an integrase inhibitor, 37% started a nonnucleoside, and 17% started a PI.
Overall 81 people (52%) attained viral suppression in a median of 105 days (interquartile range 49 to 159). Six months after treatment began, 93% starting an integrase inhibitor had undetectable viremia, compared to 70% starting a nonnucleoside and 31% starting a PI. Median time to viral suppression was significantly faster with an integrase inhibitor (62.6 days) than with a nonnucleoside (140.5 days) or a PI (154.5 days) (P = 0.002). Viral suppression was faster with integrase inhibitors, even though the integrase group had a significantly higher pre-ART viral load (109,000 copies) than people starting a nonnucleoside (56,035 copies) or a PI (49,550 copies) (P = 0.032).
The researchers noted that many people attained viral suppression after 6 months, and the low suppression rate in the PI arm could be better with a suppression cutoff above the stringent 20 copies used in this analysis. Adherence does not explain the different response rates with the three antiretroviral classes because the investigators excluded people with evidence of poor adherence.
The Yale investigators stressed that this is a preliminary analysis. They are working on a fuller report they aim to publish. The researchers noted that current guidelines typically define virologic failure as a viral load above 200 copies 24 weeks after treatment begins. Because of the faster response to integrase inhibitors, the investigators suggested that people starting ART with this class "should be suspected as having virologic treatment failure if they have not achieved viral suppression as early as 12 weeks after ART initiation."
Reference
1. Jacobson K, Ogbuagu O. Comparison of time to viral suppression among treatment-naive HIV-infected adults initiating combination antiretroviral therapy by antiretroviral regimen class. IDWeek2017/IDSA. October 4-8, 2017. San Diego. Abstract 1394.



|
| |
|
 |
 |
|
|