 |
 |
 |
| |
Two Thirds Reach Sub-50 Viral Load Through 48 Weeks With Ibalizumab
|
| |
| |
HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow
Download the PDF here
Download the PDF here
Mark Mascolini
More than half of highly antiretroviral-experienced people who took an optimized regimen containing the monoclonal antibody ibalizumab for 48 weeks reached a viral load below 50 copies [1]. In a 48-week missing-data-equals-failure analysis, 16 of 27 people (59%) had an undetectable viral load. In a 48-week completer analysis, 16 of 24 people (67%) reached that mark.
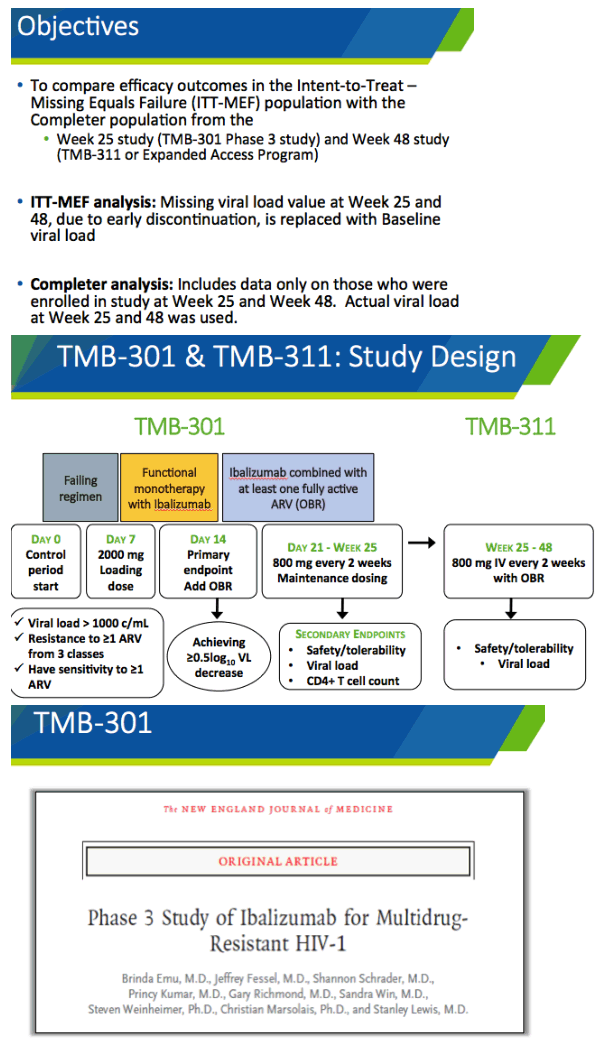
In March 2018 the FDA licensed ibalizumab in combination with other antiretrovirals for heavily pretreated people taking a failing regimen with multidrug-resistant HIV. Ibalizumab (Trogarzo) is a humanized monoclonal antibody that blocks HIV entry into CD4 cells. The novel agent is active against HIV that uses either CCR5 or CXCR4 receptors on CD4 cells. Ibalizumab is not cross-resistant with current antiretrovirals, and no drug-drug interactions with licensed antiretrovirals have come to light.
TMB-301 was a 25-week phase 3 trial of ibalizumab plus an optimized background regimen in heavily pretreated adults. The new analysis focuses on virologic response through week 48 in TMB-301 participants who entered an expanded-access program. On day 7 of TMB-301 participants got a 2000-mg intravenous loading dose of ibalizumab and continued their failing regimen for 7 days. On day 14 they replaced the failing regimen with an optimized regimen containing at least 1 fully active antiretroviral. On day 21 they started an intravenous ibalizumab maintenance dose of 800 mg every 2 weeks. After week 25 people who joined the expanded-access program continued that dose of the monoclonal antibody. This analysis assessed response at week 48.
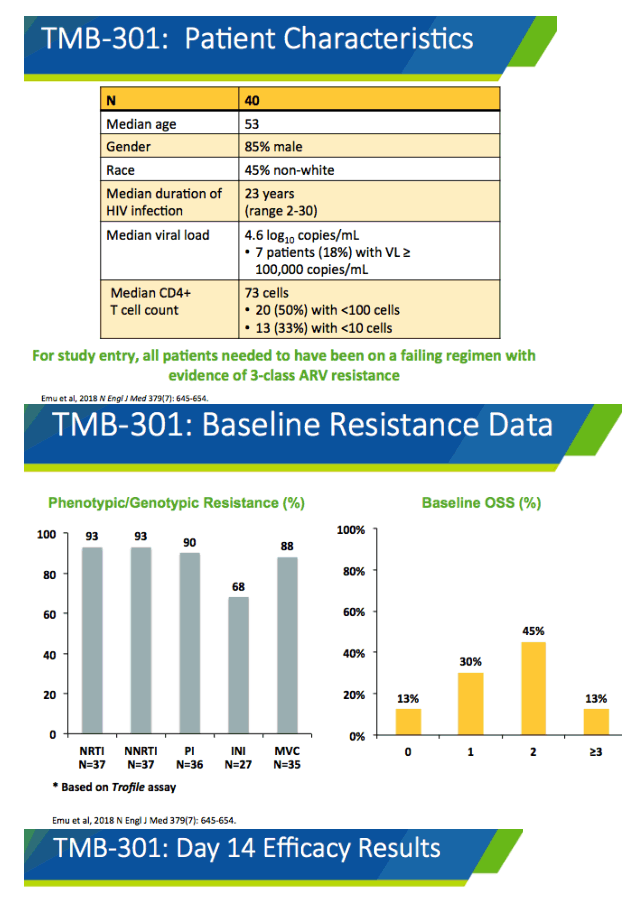
Among the 40 people who enrolled in the original trial, 85% were men and 45% nonwhite. Median age stood at 53, median HIV duration 23 years (range 2 to 30), median CD4 count 73, and median viral load 4.6 log10 copies (about 40,000 copies). Seven people (18%) had an initial viral load above 100,000 copies. All participants had evidence of HIV resistant to 3 antiretroviral classes.
At week 25 of the trial viral load dropped an average 1.7 log (50-fold) in a missing-equals-failure analysis and 2.2 log (about 160-fold) among 31 week-25 completers. Proportions with a week 25 viral load below 50 and 200 copies in the missing-equals-failure analysis were 43% and 50%, and in the completer analysis 55% and 65%. Among the 9 people who did not reach week 25, 4 died for reasons unrelated to study drugs, 4 stopped for reasons other than adverse events, and 1 had a drug-related serious adverse event.
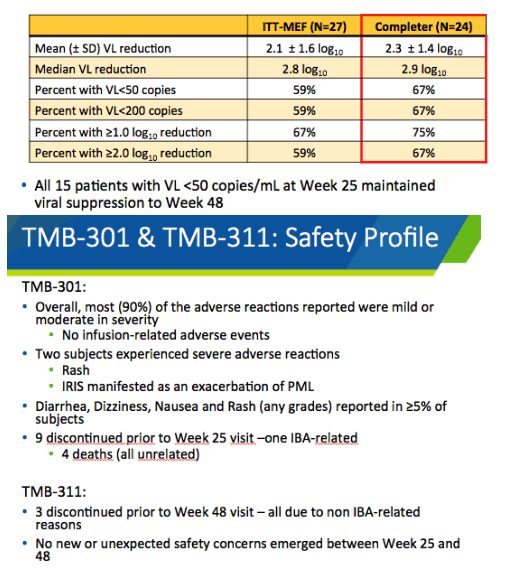
Twenty-seven people entered the expanded-access program. Their age averaged 53 years, 85% were men, 41% were nonwhite, and 59% had exhausted 3 or more antiretroviral classes. Twenty-four of these 27 people completed week 48 of follow-up on ibalizumab. At that point viral load dropped an average 2.1 log in a missing-equals-failure analysis and 2.3 log in a completer analysis. Respective median viral load drops were 2.8 and 2.9 log. Proportions of participants with a week-48 viral load below 50 copies were 59% in the missing-equals-failure analysis and 67% in the completer analysis. All 15 people with a week-25 viral load below 50 copies maintained that level through week 48.
Throughout the trial and expanded-access program, 90% of adverse reactions were mild or moderate. No infusion-related adverse events developed. The 2 severe adverse reactions were rash and immune reconstitution inflammatory syndrome (IRIS) involving progressive multifocal leukoencephalopathy. Among the 3 people who stopped treatment during expanded access, no one stopped for reasons related to ibalizumab.
References
1. Emu B, Weinheimer S, Cohen Z, Lewis S. Analysis of patients completing the ibalizumab phase III trial and expanded access program. HIV Drug Therapy, Glasgow 2018, October 28-31, 2018, Glasgow. Abstract O345.
2. Emu B, Fessel J, Schrader S, et al. Phase 3 study of ibalizumab for multidrug-resistant HIV-1. N Engl J Med. 2018;379:645-654.
https://www.nejm.org/doi/full/10.1056/NEJMoa1711460
--------------------------------
New Updated HHS ART Guidelines Issued Oct 25, 2018 say:
Ibalizumab (IBA), a CD4 post-attachment inhibitor, was recently approved for use in persons with multidrug-resistant HIV. A review of the results of a clinical trial on IBA use in this setting has been added to the section.
.......Patients with ongoing detectable viremia who lack sufficient treatment options to construct a fully suppressive regimen may be candidates for the recently approved CD4 post-attachment inhibitor ibalizumab (IBA).61 A single-arm, multicenter clinical trial enrolled 40 heavily ART-experienced participants who had multidrug-resistant HIV and who were experiencing virologic failure on an ARV regimen. Subjects received intravenous infusions of IBA every 2 weeks in addition to an optimized background regimen that included at least one additional agent to which the subject's virus was susceptible. At week 24, 43% of participants achieved HIV RNA <50 copies/mL, and 50% of participants achieved HIV RNA <200 copies/mL.62 Of the 27 participants who continued on to the 48-week follow-up study, 59% and 63% had HIV RNA <50 copies/mL and <200 copies/mL, respectively. All 15 patients who had HIV RNA <50 copies/mL at week 24 maintained viral suppression up to week 48.63


-----------------------------------------
both pdfs attached-
accompanying publication was FDA Perspective: Ibalizumab in Multidrug-Resistant HIV - Accepting Uncertainty
Abstract
Background
Ibalizumab, a humanized IgG4 monoclonal antibody, blocks the entry of human immunodeficiency virus type 1 (HIV-1) by noncompetitive binding to CD4.
Methods
In this single-group, open-label, phase 3 study, we enrolled 40 adults with multidrug-resistant (MDR) HIV-1 infection in whom multiple antiretroviral therapies had failed. All the patients had a viral load of more than 1000 copies of HIV-1 RNA per milliliter. After a 7-day control period in which patients continued to receive their current therapy, a loading dose of 2000 mg of ibalizumab was infused; the viral load was quantified 7 days later. Through week 25 of the study, patients received 800 mg of ibalizumab every 14 days, combined with an individually optimized background regimen including at least one fully active agent. The primary end point was the proportion of patients with a decrease in viral load of at least 0.5 log10 copies per milliliter from baseline (day 7) to day 14.
Results
A total of 31 patients completed the study. The mean baseline viral load was 4.5 log10 copies per milliliter, and the mean CD4 count was 150 per microliter. Of the 40 patients in the intention-to-treat population, 33 (83%) had a decrease in viral load of at least 0.5 log10 copies per milliliter from baseline (P<0.001 for the comparison with the control period). The mean viral-load decrease was 1.1 log10 copies per milliliter. During the control period, 1 patient, who received the optimized background regimen prematurely, had a decrease in viral load of 0.5 log10 copies per milliliter. At week 25, patients who had received ibalizumab plus an optimized background regimen had a mean decrease of 1.6 log10 copies per milliliter from baseline; 43% of the patients had a viral load of less than 50 copies per milliliter, and 50% had a viral load of less than 200 copies per milliliter. Among 10 patients who had virologic failure or rebound, in vitro testing identified 9 who had a lower degree of susceptibility to ibalizumab than at baseline. The most common adverse event was diarrhea (in 20% of patients). Four patients died from causes related to underlying illnesses; 1 had a serious adverse event (the immune reconstitution inflammatory syndrome) that was deemed to be related to ibalizumab therapy.
Conclusions
In patients with MDR HIV-1 infection who had advanced disease and limited treatment options, ibalizumab had significant antiviral activity during a 25-week study. Evidence of the emergence of diminished ibalizumab susceptibility was observed in vitro in patients who had virologic failure. (Funded by the Orphan Products Clinical Trials Grants Program of the Food and Drug Administration and TaiMed Biologics; TMB-301 ClinicalTrials.gov number, NCT02475629.)
----------------------------


|
| |
|
 |
 |
|
|