| |
What is Cognitive impairment ?
|
| |
| |
Download the PDF here
Jules Levin
NATAP
Given that HIV-associated cognitive impairments span the scope of emotional changes, attention, executive dysfunction, and memory, we identify the various types of neuropsychiatric complications.....HIV reproduces in microglia, the brain's immune cells, causing central nervous system (CNS) inflammation and progressive cognitive and behavioral changes.... https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4319681/
In the general literature for HIV-uninfected -
Cognitive disorders often begin subtly but progress until they significantly impede the affected individual's quality of life. It is important to understand the various cognitive disorders, their symptoms and relevant treatment options.
Cognitive disorders are a part of the neurocognitive disorder classification in the fifth edition of the Diagnostic and Statistical Manual of Mental Disorders (DSM-V). Cognitive disorders are defined as any disorder that significantly impairs the cognitive function of an individual to the point where normal functioning in society is impossible without treatment. Some common cognitive disorders include:
• Dementia
• Developmental disorders
• Motor skill disorders
• Amnesia
• Substance-induced cognitive impairment
Like most mental disorders, cognitive disorders are caused by a variety of factors. Some are due to hormonal imbalances in the womb, others to genetic predisposition and still others to environmental factors. Common environmental causes of cognitive disorders include a lack of proper nutrients and interaction during vulnerable stages of cognitive development, particularly during infancy.
Other common causes of cognitive disorder include substance abuse and physical injury. When an area of the brain that determines cognitive function is damaged, either by the excessive use of drugs, by alcohol or from physical trauma, those neurophysiological changes can result in cognitive dysfunction.
Cognitive problems manifest in a variety of ways, with emotional imbalance being one of the most common symptoms. Cognitive impairment is frustrating, and those suffering from it often react with emotional outbursts, making it difficult for friends and family to help. Others may push people away in an attempt to isolate themselves, only making the problem worse. Other cognitive disorders have the opposite effect, causing the person to have dulled or nonexistent emotions.... .https://www.psychguides.com/guides/cognitive-problem-symptoms-causes-and-effects/
Cognitive disorders (CDs), also known as neurocognitive disorders (NCDs), are a category of mental health disorders that primarily affect cognitive abilities including learning, memory, perception, and problem solving. Neurocognitive disorders include delirium and mild and major neurocognitive disorder (previously known as dementia). They are defined by deficits in cognitive ability that are acquired (as opposed to developmental), typically represent decline, and may have an underlying brain pathology[1]. The DSM-5 defines six key domains of cognitive function: executive function, learning and memory, perceptual-motor function, language, complex attention, and social cognition.
Although Alzheimer's disease accounts for the majority of cases of neurocognitive disorders, there are various medical conditions that affect mental functions such as memory, thinking, and the ability to reason, including frontotemporal degeneration, Huntington's disease, Lewy body disease, traumatic brain injury (TBI), Parkinson's disease, prion disease, and dementia/neurocognitive issues due to HIV infection.[2] Neurocognitive disorders are diagnosed as mild and major based on the severity of their symptoms. While anxiety disorders, mood disorders, and psychotic disorders can also have an effect on cognitive and memory functions, the DSM-IV-TR does not consider these cognitive disorders, because loss of cognitive function is not the primary (causal) symptom.[3] Additionally, developmental disorders such as Autism spectrum disorder are typically developed at birth or early in life as opposed to the acquired nature of neurocognitive disorders.
Causes vary between the different types of disorders but most include damage to the memory portions of the brain.[4][5][6] Treatments depend on how the disorder is caused. Medication and therapies are the most common treatments; however, for some types of disorders such as certain types of amnesia, treatments can suppress the symptoms but there is currently no cure.[5][6] https://en.wikipedia.org/wiki/Cognitive_disorder
AIDS 2017 - Nightingale, Sama; Winston, Alanb
"The underlying mechanisms leading to cognitive impairment in HIV-positive populations are likely multifactorial and are outlined in Table 1. These proposed mechanisms are diverse and include HIV-viral factors, antiretroviral treatment factors and host factors.....Some risk factors for cognitive impairment increase with age and may be more prevalent in HIV-positive individuals. The interaction of HIV with cerebrovascular disease and age-related neurodegeneration may be particularly important. Cognitive impairment is found across all age groups studied and specific signals suggesting an accelerated ageing phenotype are currently lacking......Patients with HIV and hepatitis C coinfection are almost twice as likely to have cognitive impairment as HIV-positive individuals without hepatitis C infection.....The use of psychoactive drugs, particularly methamphetamine, has deleterious effects on cognition, which is more pronounced when combined with HIV-disease....
SUMMARY: Mild forms of cognitive impairment are prevalent in treated HIV-positive populations with access to effective antiretroviral therapy. Estimates of the burden of this problem are challenging due to different diagnostic criteria and differing patient characteristics in cohorts studies and vary from 5% to over 50%. The underlying pathogenesis of this cognitive impairment is poorly understood; however, this is likely to be multifactorial and includes a combination of immune-mediated factors such as monocyte/microglial activation despite antiretroviral therapy, neurotoxicity of antiretroviral therapy, the presence of infectious and noninfectious comorbidities, and lifestyle factors. Neuroimaging techniques such as MRI and MR spectroscopy can be useful when assessing patients with cognitive impairment. Soluble biomarkers may also have a role and require further evaluation, both when individual biomarkers are utilized and when combination of different biomarkers is utilized. Some patients with suppressed plasma HIV RNA have detectable virus in cerebrospinal fluid, termed cerebrospinal fluid escape. This can be associated with compartmentalized and resistant virus and has been associated with acute/subacute neurological presentations; however, most cases are asymptomatic or are associated with subtle impairment in cognitive function. In symptomatic patients with cerebrospinal fluid escape, antiretroviral therapy changes should be based on resistance profiles of cerebrospinal fluid virus and/or historic resistance in plasma. Evidence is relatively sparse for changing antiretroviral therapy in patients with cognitive impairment in the absence of cerebrospinal fluid escape or resistant virus. Such patients should be assessed for confounding conditions and potential antiretroviral neurotoxicity."
---------------------------
Cognitive impairment remains a frequently reported complaint in HIV-positive patients despite virologically suppressive antiretroviral therapy. Rates of cognitive impairment in antiretroviral treated HIV-positive cohorts vary and strongly depend on definitions utilized.
The underlying pathogenesis is likely to be multifactorial and includes immune activation, neuroinflammation, antiretroviral neurotoxicity, the presence of noninfectious comorbidities such as vascular disease and depression and patient lifestyle factors such as recreational drug use.
Contributing factors to cognitive impairment may change over time with ageing HIV-positive populations. Cerebrovascular disease and neurodegenerative causes of cognitive impairment may become more common with advancing age; how these factors interact with HIV-associated cognitive impairment is not yet known.
Cerebrospinal fluid HIV RNA escape may occur in up to 10% of patients undergoing lumbar puncture clinically and can be associated with compartmentalized and resistant virus.
Changes in antiretroviral therapy in patients with cognitive impairment should be based on current and historic resistance profiles of cerebrospinal fluid and plasma virus, or on potential antiretroviral drug neurotoxicity. Whether and how antiretroviral therapy should be changed in the absence of these factors is not known and requires study in adequately powered randomized trials in carefully selected clinical cohorts.
Cognitive impairment in antiretroviral therapy treated HIV-positive individuals continues to be reported and may become an increasingly important issue as this population ages [1,2]. The pathophysiology of this cognitive impairment is likely multifactorial with the underlying mechanisms incompletely understood. Current management is based predominantly on optimal strategies for best clinical practice rather than specific evidence base. Here we review the epidemiology, pathophysiology, clinical investigation and management of HIV-associated cognitive impairment.
Diagnosis of cognitive impairment
Several diagnostic criteria for cognitive impairment have been proposed with the HIV-associated neurocognitive disorders (HAND) criteria the most widely used [10]. The HAND criteria, also known as the Frascati Criteria, suggest neuropsychiatric testing of at least the following cognitive domains: verbal and language; attention and working memory; abstraction and executive function; memory (learning and recall); speed of information processing; and sensory-perceptual and motor skills. Criteria for HAND definition are met when at least two of these domains are more than one SD from normative test scores. For a diagnosis of HAND cognitive deficits must be acquired and not explained by comorbidities; however, in practice this can be difficult to determine and does not reflect the multifactorial causes of cognitive impairment seen in most clinical cohorts. Self-report functional assessments (such as Lawton and Brody's modified Activities of Daily Living scale or the Patients Assessment of Own Functioning Inventory) divide those with mild cognitive impairment into Mild Neurocognitive Disorder if cognitive impairment interferes with daily functioning or Asymptomatic Neurocognitive Impairment if it does not. The HAND criteria were created as a research tool and have limited applicability clinically, particularly as the significance of identifying patients with Asymptomatic Neurocognitive Impairment remains to be determined. These criteria have the potential for high false positive rate and the normative test scores upon which these criteria depend can be difficult to establish accurately [11]. Within recent studies utilizing more stringent criteria for the diagnosis of cognitive impairment, rates of impairment are substantially lower than when using the HAND criteria and are highly dependent on the methodology used [12]. In the Pharmacokinetic and Clinical Observations in People over Fifty (POPPY) study, three criteria for assessing cognitive impairment have been utilized: the HAND criteria, global deficit score and the multivariate normative comparison methods [13]. Here, the most stringent definition of cognitive impairment was defined as patients who met all three diagnostic criteria. In such patients, median global cognitive scores were lower and symptoms of cognitive complaints were higher compared with other definitions of cognitive impairment. Such an approach may help rationalize this diagnostic conundrum in the future.
When cognitive impairment is suspected in clinical practice, formal neuropsychometric testing by an experienced neuropsychologist is a crucial assessment. Such assessments typically use estimates of premorbid function to help determine whether an individual has significant cognitive impairment and take into account other factors such as mood. Mental health disorders are common in HIV-positive populations and can contribute to poor cognitive performance on testing [14]. Furthermore, a decline in cognitive performance may lead to depressive symptoms.
More recently computerized cognitive assessments have been evaluated in HIV. These are fast and inexpensive and can be easily administered in clinic. Their role requires further evaluation, in particular whether they may be a useful screening test [15].
Pathogenesis of HIV-associated cognitive impairment
The underlying mechanisms leading to cognitive impairment in HIV-positive populations are likely multifactorial and are outlined in Table 1. These proposed mechanisms are diverse and include HIV-viral factors, antiretroviral treatment factors and host factors.
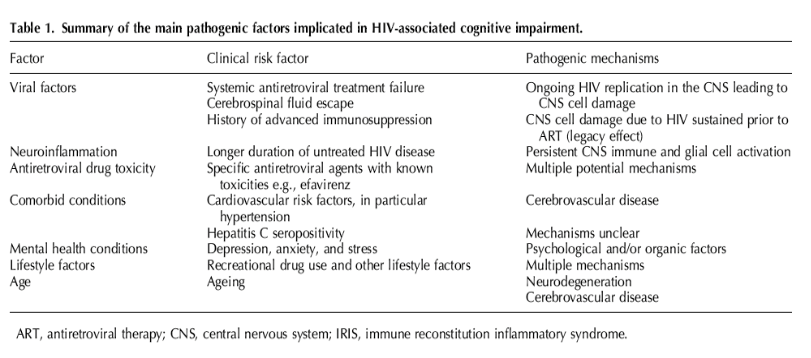
Viral factors
HIV replication in the CNS has been strongly linked to neuronal damage and the development of HAD. HIV enters the brain early in disease via migrating myeloid and lymphoid cells and establishes infection in perivascular macrophages and microglia. Direct infection may also occur in astrocytes. Direct neurotoxicity occurs from the HIV virus and its proteins, such as the envelope glycoprotein gp120 and the regulatory protein Tat. Chronic sustained immune activation in the CNS leads to the production of neurotoxic products, such as nitric oxide, arachidonic acid and proinflammatory cytokines [16].
Cerebrospinal fluid HIV RNA is closely correlated with plasma HIV RNA, and HIV replication in the CNS is often associated with systemic treatment failure [17]. In some individuals, HIV RNA in cerebrospinal fluid is higher than in blood and can be detected in cerebrospinal fluid when viraemia is otherwise fully suppressed in the plasma compartment. This so-called cerebrospinal fluid escape has been described in approximately 2-10% of patients depending on population studied and definition used [18-21]. In such cases, HIV-strains have been documented to be compartmentalized in the CNS with virus isolated from cerebrospinal fluid demonstrating different patterns of drug-associated resistance mutations to that observed in the plasma compartment [22]. Cerebrospinal fluid escape is associated with low nadir CD4+ and may be related to the establishment of a viral reservoir during advanced immunosuppression, which is only partially responsive to antiretroviral therapy [6,23].
In untreated patients in the preantiretroviral era, high levels of HIV in cerebrospinal fluid were associated with HAD [24-26]. In treated populations cerebrospinal fluid-escape is associated with markers of neuroinflammation; however, the associations with clinically relevant cognitive dysfunction are less clearly defined [6,25,27]. In case series, cerebrospinal fluid escape has been described in association with florid acute/subacute cognitive and neurological syndromes in patients on long-term suppressive antiretroviral therapy, but this appears to be uncommon [28-30].
Antiretroviral factors
Limited penetration of antiretroviral agents into the brain may contribute to cognitive impairment. Concentrations of many antiretroviral drugs are substantially lower in the cerebrospinal fluid than in plasma and concentrations in perivascular macrophages which harbour productive infection in the brain can fall below the IC50 for several drugs [31].
On the converse, there is growing evidence of neurotoxic effects of some antiretroviral agents, and there may be a therapeutic window between efficacy and toxicity in the CNS [32,33]. It has been suggested that antiretroviral drugs may lead to a chronic low-grade CNS immune reconstitution inflammatory syndrome whereby persistent immune and glial cell activation in the CNS established in advanced infection does not fully correct with antiretroviral therapy [34]. Whether anti-inflammatory treatment may have a role in such patients is the subject of ongoing study [35]. In some patients with a low nadir CD4+ cell count, cognitive impairment may relate to a legacy effect of CNS damage sustained before starting antiretroviral therapy, rather than an ongoing inflammatory process.
Premature ageing and neurodegeneration
Some risk factors for cognitive impairment increase with age and may be more prevalent in HIV-positive individuals. The interaction of HIV with cerebrovascular disease and age-related neurodegeneration may be particularly important. Cognitive impairment is found across all age groups studied and specific signals suggesting an accelerated ageing phenotype are currently lacking [12,13].
The prevalence of cerebrovascular disease increases with age. In HIV-positive populations, cerebrovascular disease can occur because of traditional risk factors or can result from the metabolic and systemic effects of HIV and antiretroviral therapy on endothelial function and cardiovascular risk factors [36,37]. A recent report has demonstrated that white matter hyperintensities, presumed to be of vascular origin, are more common in HIV-positive groups than lifestyle matched uninfected controls; these increase with age and are associated with increased rates of cognitive impairment [38].
Alzheimer's like pathology may contribute to the pathogenesis of HIV-associated cognitive impairment in ageing populations. Abnormal beta-amyloid deposition, a hallmark of Alzheimer's disease, has been observed in the brains of HIV-positive patients [39], and neurofibrillary tangles composed of phosphorylated tau occur at a younger age in people with HIV than in healthy controls [40]. To what extent Alzheimer's pathology contributes to neuronal damage and whether it increases the risk of HIV-positive patients developing Alzheimer's disease in later life is unclear. The ApoE ε4 genotype, an established risk factor for Alzheimer's disease, has been associated with cognitive impairment in HIV in some but not all cohorts [41,42]. This association may be restricted to older populations [43].
Host factors
When considering HIV-associated cognitive impairment, it is important to consider factors that are not directly related to HIV infection but are prevalent in HIV-positive populations [44,45]. Hepatitis C viral coinfection is associated with cognitive dysfunction independently of HIV infection and such effects may be compounded in patients with HIV. Patients with HIV and hepatitis C coinfection are almost twice as likely to have cognitive impairment as HIV-positive individuals without hepatitis C infection [46]. The use of psychoactive drugs, particularly methamphetamine, has deleterious effects on cognition, which is more pronounced when combined with HIV-disease [47,48]. As discussed above, cerebrovascular disease can contribute to cognitive impairment, and mechanisms underlying cerebrovascular disease become increasingly important as the HIV-positive population ages. For reasons that are unclear, some demographic groups, such as women and people of black African ethnicity, appear to be more vulnerable to cognitive impairment [45]. Educational attainment is protective against cognitive impairment, a general effect also observed in other causes of cognitive impairment such as Alzheimer's disease [44].
Assessing cognitive impairment
Assessments of patients with cognitive complaints include bedside screening assessments, formal neuropsychological assessments, biomarker assessments in both plasma and cerebrospinal fluid, and the use of neuroimaging techniques.
Screening for cognitive impairment
Screening for cognitive impairment out with the field of HIV-medicine is a controversial area due to concerns regarding overdiagnosing patients and lack of interventions when early stages of dementia are recognized. Similar concerns exist within the HIV-field with the added challenges of determining definitive diagnostic criteria for HIV-associated cognitive impairment, as discussed above, and interpreting cognitive function results from diverse population groups.
Although several screening algorithms have been proposed most guidelines now recommend cognitive testing only in symptomatic patients [49]. Most of the cognitive testing methods evaluated in HIV populations have been developed for dementia and are crude tools for the milder forms of cognitive impairment. Many tests such as the HIV Dementia Scale focus on motor and cognitive slowing; these are key features of the subcortical dementia phenotype found in HAD but are not seen in milder cognitive impairment in which deficits in learning, memory and executive function predominate [50].
Biomarkers
Biomarkers relating to immune and glial activation, neuronal injury and other mechanisms can be determined in cerebrospinal fluid and blood at abnormal concentrations in some patients with treated HIV-disease despite adequate HIV RNA suppression [27]. Many of these biomarkers correlate with impaired cognitive function to varying extents, but none has yet been shown to be clinically useful in isolation. It may be that a combination of biomarkers reflecting neuronal injury and glial cell or immune activation in combination with HIV RNA load in cerebrospinal fluid is a more useful tool than a single biomarker alone; however, to date, no such combination has been validated for clinical use, and this remains an area of ongoing research [51].
Traditional biomarkers in this field have focused on measurement of soluble markers from cerebrospinal fluid samples. In a recent study, the plasma biomarker neurofilament light chain protein has been associated with HAD and may prove a valuable tool in assessing cognitive function in HIV-positive patients whilst sparing the need for lumbar puncture examination [52].
As the HIV-positive population ages, differentiating cognitive impairment due to HIV from neurodegenerative conditions such as Alzheimer's disease becomes increasingly important. Decreased cerebrospinal fluid beta-amyloid concentrations associated with Alzheimer's disease have also been observed in HIV-positive patients with cognitive impairment [53,54], consistent with pathological reports of brain amyloid deposition in both conditions [39]. Cerebrospinal fluid tau is increased in Alzheimer's disease and HAD [53] but may be decreased in those with milder HIV-associated cognitive impairment [54]. Data are conflicting in this area and whether these biomarkers are useful clinically requires validation in larger cohorts. Other features may be more useful to distinguish Alzheimer's disease from HIV-associated cognitive impairment, such as hippocampal changes on imaging and the progressive nature of cognitive decline in Alzheimer's disease.
Clinical management
Antiretroviral therapy is a crucial component of the management of HIV-associated cognitive impairment. For patients diagnosed with HIV-associated cognitive impairment who are not receiving antiretroviral therapy, the initiation of treatment is recommended to prevent potential ongoing neuronal damage from uncontrolled HIV replication. Although this recommendation may appear less relevant subsequent to the findings of the INSIGHT-START study in which guidelines now recommend antiretroviral therapy to all persons with HIV [62], it remains crucial to consider cognitive impairment as a further indication for antiretroviral therapy for individuals who do not otherwise feel ready to commence treatment.
Regarding type of antiretroviral therapy to commence in patients with cognitive impairment, evidence base for the use of specific antiretroviral agents or the use of specific antiretroviral combinations with regard to improvements in cognitive function are relatively sparse. Although pharmacokinetic scoring systems have been proposed, which grade drug exposure in the cerebrospinal fluid [63], clinical data on the effectiveness of utilizing such a strategy are lacking. In general, standard antiretroviral regimens are recommended [64]. In one randomized controlled trial, no advantages were observed in cognitive function with the use of a maraviroc containing antiretroviral regimen [65] and in another randomized controlled trail, no differences in cognitive function were observed between nucleoside-reverse-transcriptase-inhibitor containing and sparing antiretroviral regimens [66].
Rather than inclusion of specific antiretroviral agents, recent guidelines have focused on specific antiretroviral agents to avoid in patients with cognitive impairment. For instance, avoidance of efavirenz use is generally recommended on the basis of results of cohort studies suggesting cognitive function to be poorer on patients currently receiving efavirenz-based antiretroviral regimens [67,68].
For patients receiving antiretroviral therapy, the management of cognitive impairment is based on the presence or the absence of detectable HIV RNA in the plasma and cerebrospinal fluid. Where HIV RNA is detected, it is generally recommended that antiretroviral therapy is optimized on the basis of the results of current and historical genotypic resistance tests from both the plasma and cerebrospinal fluid compartments, with a focus on determining if resistance is present in cerebrospinal fluid samples. Where HIV RNA is not detected, which is in the majority of patients, reassessment for other confounding conditions which may lead to or add to cognitive decline should be undertaken, and reassessment of antiretroviral therapies which may be associated with poorer cognitive function such as efavirenz should be given consideration.
Recommendations on the method and frequency of monitoring of patients with HIV-associated cognitive impairment are not evidenced based, and guidelines here are best practice algorithms. In general, close clinical monitoring is recommended with follow-up assessments of cognitive function 3-12 months after treatment initiation or modification is suggested. Follow-up with repeat cerebrospinal fluid examination is generally only recommended with patients in whom clinical improvements are not observed.
Nonpharmacological interventions should also be considered in the management of patients with cognitive impairment. Advice should be given on increasing physical activity, managing stress and low mood, sleeping well, eating healthily and challenging the mind, as well as general lifestyle advice on stopping smoking and limiting alcohol intake and drug use. Several nonpharmacological interventions are currently the subject of investigation in a large longitudinal study; these include cognitive training, self-management techniques, insomnia intervention and the effect of physical exercise [69].
Summary
Mild forms of cognitive impairment are prevalent in treated HIV-positive populations with access to effective antiretroviral therapy. Estimates of the burden of this problem are challenging due to different diagnostic criteria and differing patient characteristics in cohorts studies and vary from 5% to over 50%. The underlying pathogenesis of this cognitive impairment is poorly understood; however, this is likely to be multifactorial and includes a combination of immune-mediated factors such as monocyte/microglial activation despite antiretroviral therapy, neurotoxicity of antiretroviral therapy, the presence of infectious and noninfectious comorbidities, and lifestyle factors.
Neuroimaging techniques such as MRI and MR spectroscopy can be useful when assessing patients with cognitive impairment. Soluble biomarkers may also have a role and require further evaluation, both when individual biomarkers are utilized and when combination of different biomarkers is utilized. Some patients with suppressed plasma HIV RNA have detectable virus in cerebrospinal fluid, termed cerebrospinal fluid escape. This can be associated with compartmentalized and resistant virus and has been associated with acute/subacute neurological presentations; however, most cases are asymptomatic or are associated with subtle impairment in cognitive function. In symptomatic patients with cerebrospinal fluid escape, antiretroviral therapy changes should be based on resistance profiles of cerebrospinal fluid virus and/or historic resistance in plasma. Evidence is relatively sparse for changing antiretroviral therapy in patients with cognitive impairment in the absence of cerebrospinal fluid escape or resistant virus. Such patients should be assessed for confounding conditions and potential antiretroviral neurotoxicity.
|
|
| |
| |
|
|
|