 |
 |
 |
| |
Kick-and-kill with vaccines plus vorinostat has no impact on CD4 reservoir
|
| |
| |
22nd International AIDS Conference, Amsterdam, Netherlands, July 23-27, 2018
Mark Mascolini
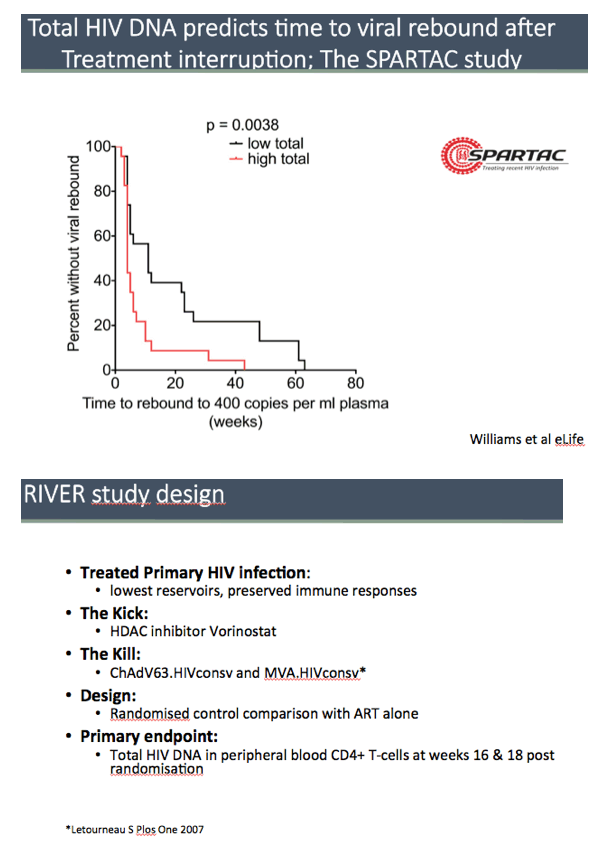
A randomized trial found that giving people with recently diagnosed HIV two vaccines and vorinostat plus standard antiretroviral therapy (ART) did not drain the HIV reservoir in CD4 cells faster than standard ART alone [1]. This first randomized study of a kick-and-kill approach to mopping up HIV reservoirs underlines the difficulty of reactivating latent virus and killing it with ART or--in this case--with an immune system trained by a double-barreled prime-boost HIV vaccine.
RIVER trial researchers in the UK planned to deliver the kick-and-kill to people who responded to antiretroviral treatment of primary HIV infection and thus should have the lowest HIV reservoirs and the best-preserved immune responses. They aimed to kick latent HIV into an activated state by giving the well-studied and generally well-tolerated HDAC inhibitor, vorinostat. The kill would come from HIV-specific T-cell responses stirred by two vaccines, ChAdV63.HIVconsv and MVA.HIVconsv.
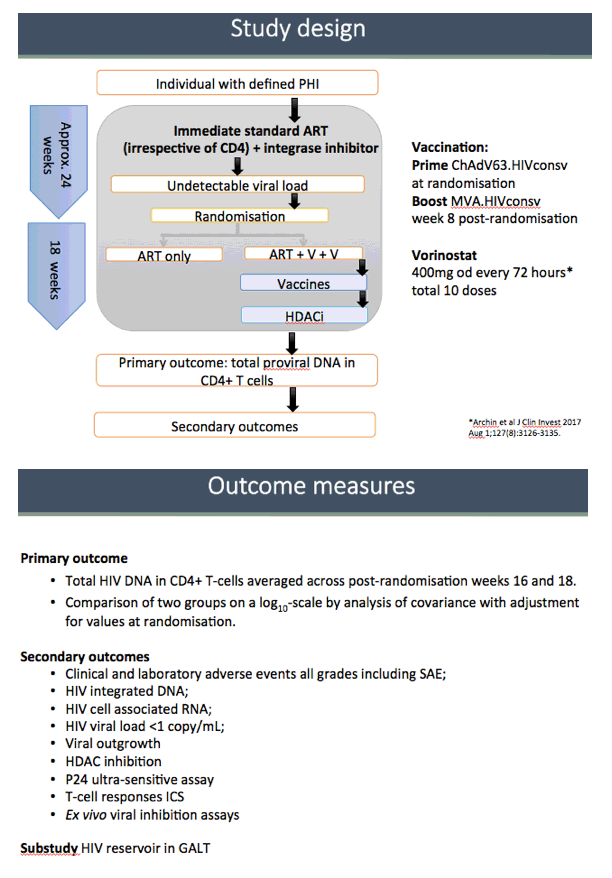
RIVER enrolled people who reached an undetectable viral load with standard ART including an integrase inhibitor for primary HIV infection. Researchers randomized them to 18 weeks of continued ART alone or 18 weeks of continued ART plus the two vaccines and vorinostat. The primary study aim was to compare total proviral HIV DNA in CD4 cells between the two treatment arms averaged across weeks 16 and 18 after randomization. The investigators compared HIV DNA in the two groups on a log scale by analysis of covariance with adjustment for values at randomization.
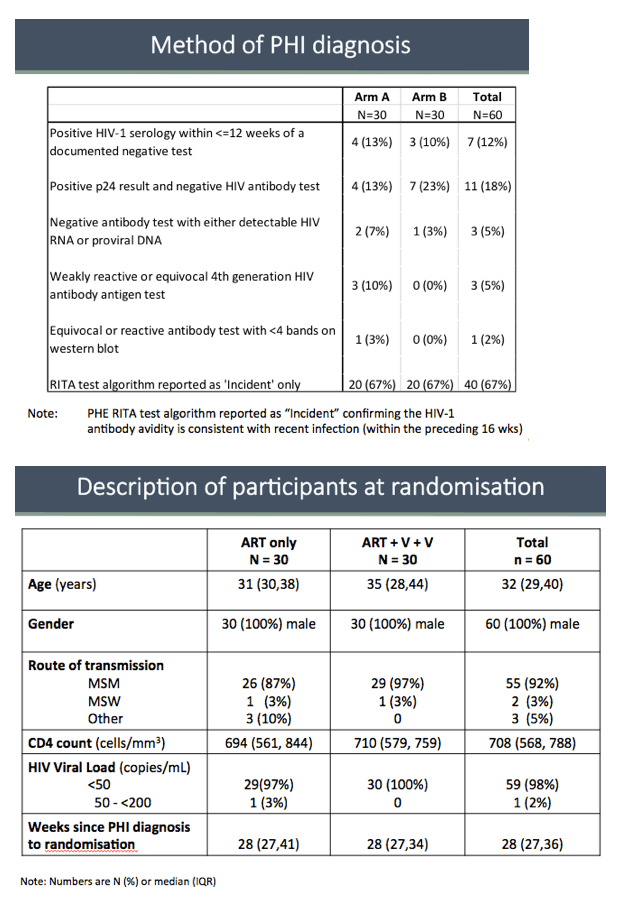
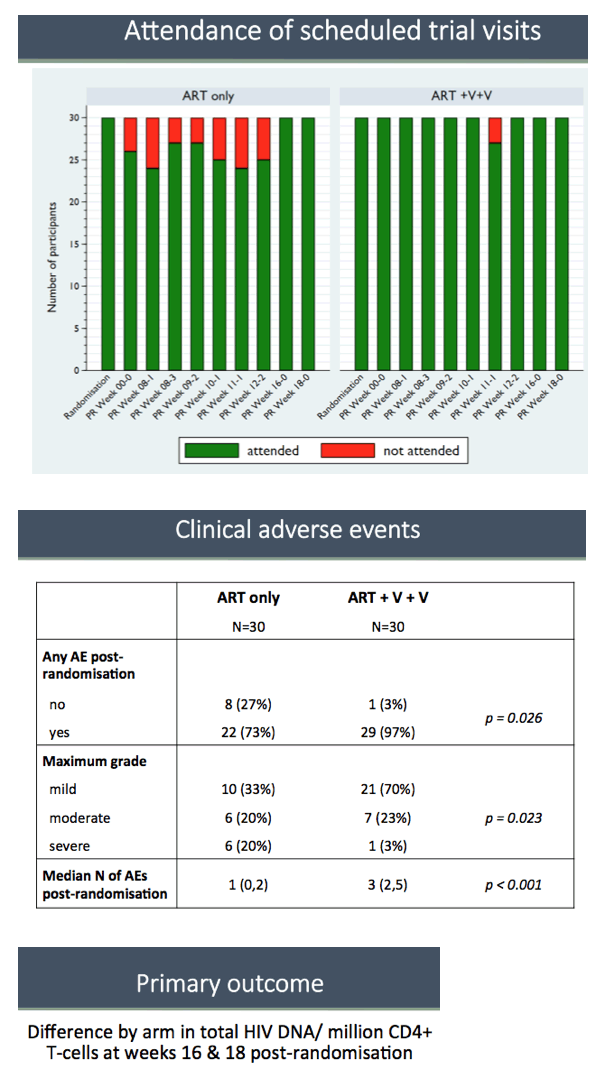
Each study arm included 30 participants with a total median age of 32 years. Everyone in both arms was male and 92% picked up HIV during sex between men. Median CD4 count measured 710 in the kick-and-kill arm and 694 in the ART-only arm. One person in the ART-only arm and none in the kick-and-kill arm had a baseline viral load between 50 and 200 copies; everyone else had a sub-50-copy baseline load. Median time between primary HIV diagnosis and randomization was 28 weeks in both study arms. No one dropped out of the study and 97% of participants adhered to their intervention.
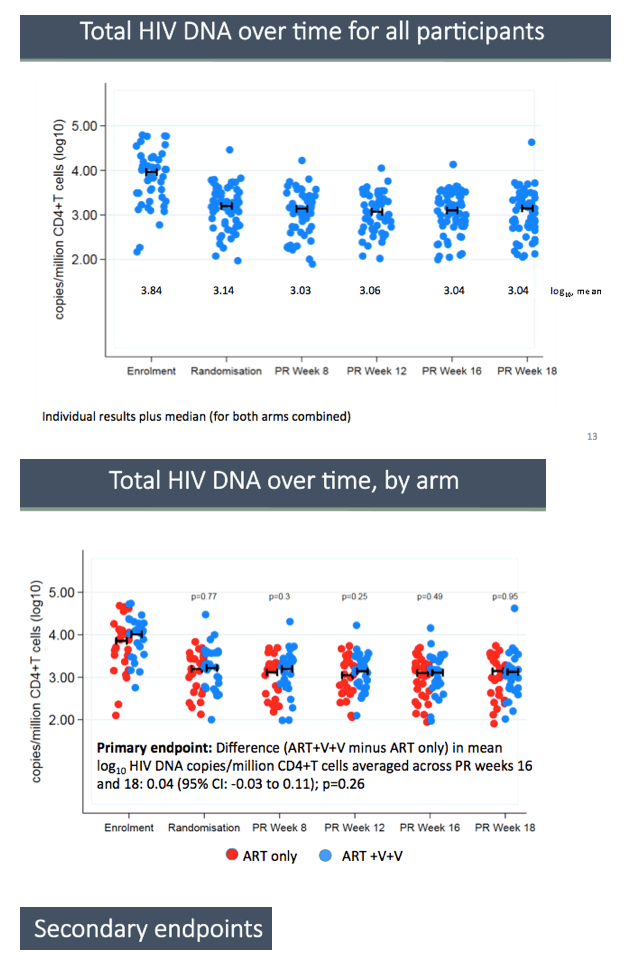
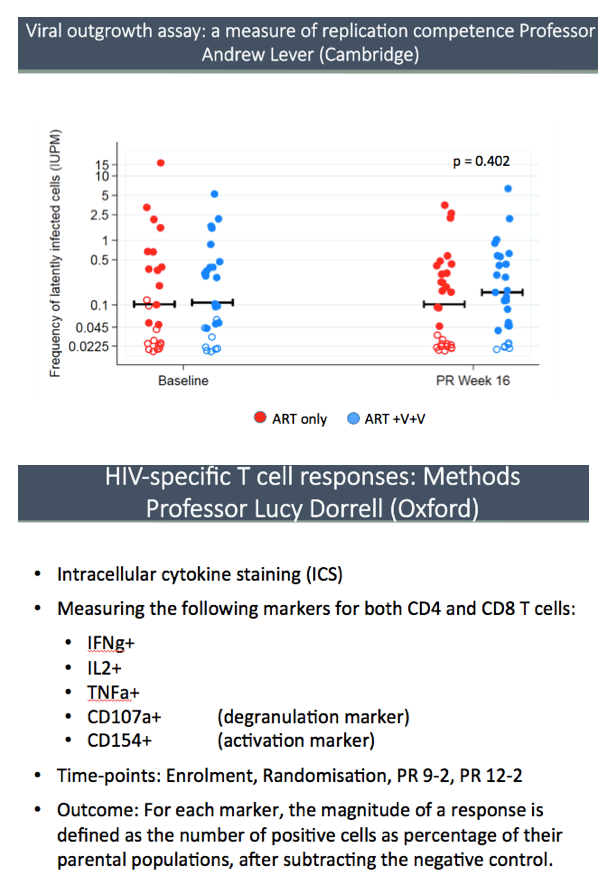
Total HIV DNA in CD4 cells proved virtually identical in the two study groups at enrollment, randomization, and 8, 12, 16, and 18 weeks after randomization. The between-arm difference averaged across weeks 16 and 18 was 0.04 copies/million CD4s (95% confidence interval -0.03 to 0.11, P = 0.26). This analysis adjusted for baseline total HIV DNA and stratum, with multiple imputation of missing baseline values. A viral outgrowth assay, which measures viral replication competence, found no difference between the two study arms 16 weeks after randomization.
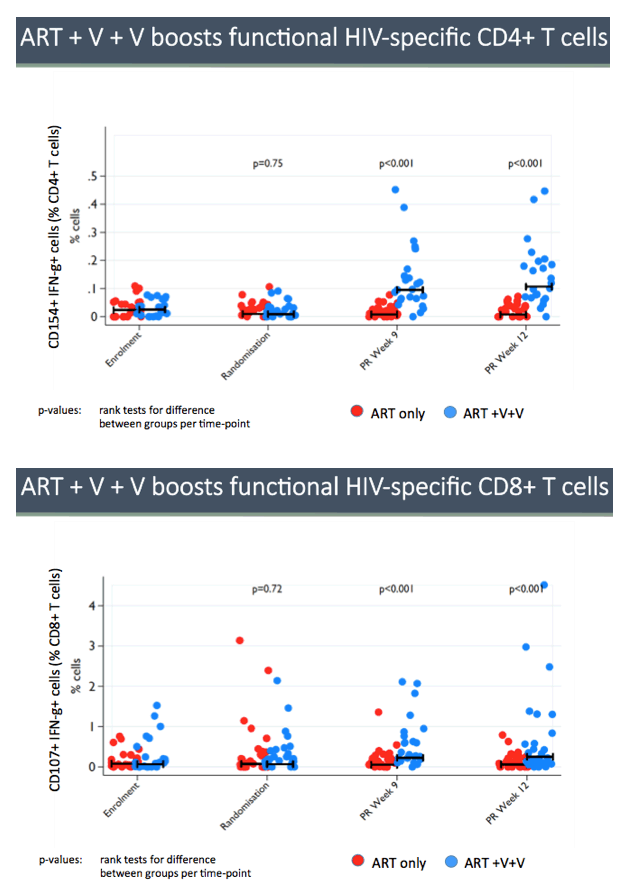
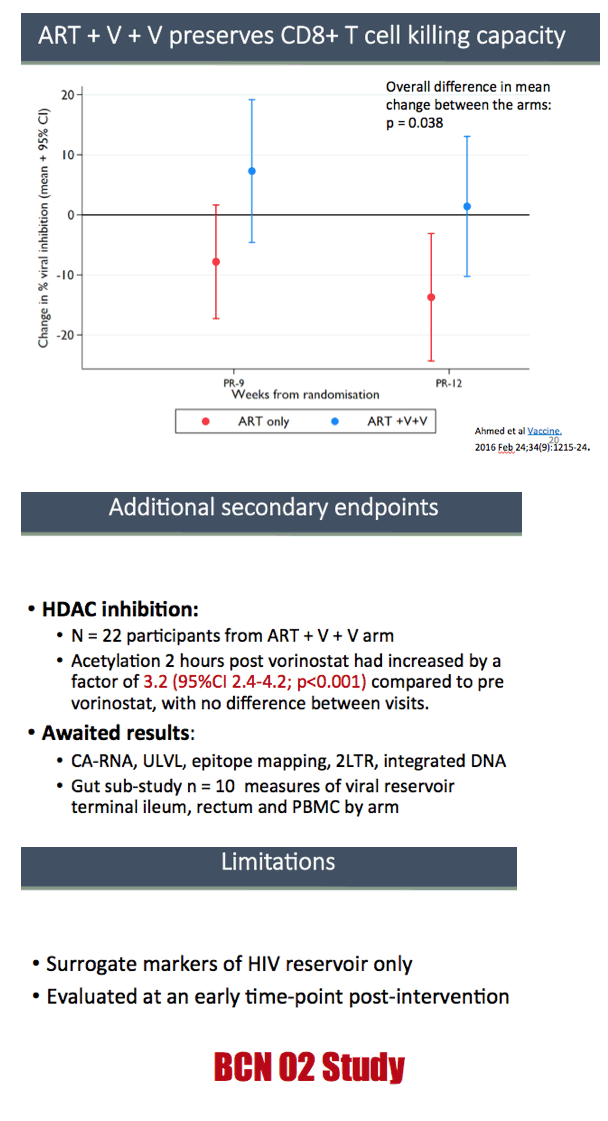
The kick-and-kill strategy did significantly boost levels of functional CD8 T cells 9 and 12 weeks after randomization (P < 0.001). And kick-and-kill preserved CD8-cell killing capacity 12 weeks after randomization whereas standard ART did not (P = 0.038).
Significantly more kick-and-kill recipients had any adverse event after randomization (97% versus 73%, P = 0.026), but most adverse events in the kick-and-kill group were mild (70% versus 33%, P = 0.023).
In a press release principal investigator Sarah Fidler (Imperial College London) noted that "this particular set of treatments together didn't add up to a potential cure for HIV, based on what we've seen so far." But co-principal investigator John Frater (University of Oxford) stressed that the findings do not mean no kick-and-kill approach will work. More effective agents, he argued, might yield different results.
Reference
1. Fidler S, Stohr S, Pace M, et al. A randomised controlled trial comparing the impact of ART with a 'kick-and-kill' approach to ART alone on HIV reservoirs in primary HIV infection: RIVER trial. AIDS 2018: 22nd International AIDS Conference, Amsterdam, Netherlands, July 23-27, 2018. Abstract TUAA0202LB.
------------------------------------



--------------------------------
RIVER trial press release - First randomised trial of 'kick and kill' approach to HIV cure leaves puzzles to be solved
Issued on behalf of the RIVER Study
http://www.cherub.uk.net/river-trial-press-release/
Tuesday 24 July 2018
First randomised trial of 'kick and kill' approach to HIV cure leaves puzzles to be solved
Researchers have reported the results of the first randomised clinical trial to test a novel strategy involving waking up and then killing the 'sleeping' HIV that is hiding in the body using an experimental approach known as 'kick and kill'.
Led by Imperial College London, the University of Oxford, MRC Clinical Trials Unit at UCL, and the University of Cambridge, the RIVER study aimed to improve on current care by eradicating HIV from the body, which would represent a major step forward in the search for a cure.
Although investigators found no difference in effect between those participants who received the active 'kick and kill' therapy and those who had standard treatment, they say the trial paves the way for testing different combinations of therapies to tackle the HIV that persists in patients who receive antiretroviral treatment (ART). The trial demonstrates how researchers are looking beyond current therapies which, despite revolutionising the lives of people living with HIV, are not a cure and have to be taken for life.
The RIVER study, which ran from 2015 to 2018 in London and Brighton, tested the 'kick and kill' approach in 60 men recently diagnosed with HIV and who had the virus under control by taking ART. Despite having undetectable levels of HIV in their blood, patients on ART are not cured. If they stop taking ART, the virus returns from hiding places in the body known as 'reservoirs'. This has led researchers to propose that a possible cure for HIV would be to target these reservoirs, force HIV out of hiding and then kill it.
To test this idea, RIVER used two medicines on top of the standard ART treatment. First, two vaccines to coach the body's immune system to recognise and destroy HIV, and second, a drug called vorinostat (also used for managing cancer) that would 'wake up' the reservoir cells that HIV is hiding in and force the virus to reveal itself and face the immune system. This approach to an HIV cure is called 'kick and kill' - the kick is delivered from vorinostat, and the kill comes from the body's own immune system killer cells that have been trained by the vaccines.
To claim success in the RIVER proof of concept trial, the researchers said that those receiving the kick and kill drugs should experience a significant fall in levels of HIV in the reservoir cells, known as CD4 T cells.
However, when the results were first unveiled to researchers in April 2018, they found that the half of trial participants given the kick and kill drugs in addition to ART had similar levels of infected reservoir cells compared to those who only received standard ART therapy.
The RIVER study leaders report these initial findings today in a 'Late Breaker' session at the International AIDS Society's annual meeting in Amsterdam, Netherlands (AIDS 2018). The results of the study will be submitted for publication in a peer-reviewed journal.
RIVER Chief Investigator, Professor Sarah Fidler of Imperial College London said:
"In the RIVER study, we found that all the separate parts of the kick and kill approach worked as expected and were safe. The vaccine worked on the immune system, the kick drug behaved as we expected it to, and the ART worked in suppressing viral load in the body, but the study has shown that this particular set of treatments together didn't add up to a potential cure for HIV, based on what we've seen so far."
"For those who have access to it, ART brings amazing success in managing HIV but people have to take it for most of their lives. We have to think about other, sustainable alternatives, and a cure or at least some form of remission is a key goal.
"Finding an HIV cure means orchestrating a lot of things. We have to generate new ideas and turn them into trials that will give meaningful results, we have to agree what research tests will tell us whether things are working or not, we need to very carefully monitor study participants and most importantly we need to make sure any trial intervention is safe. RIVER achieved all these things but sadly not the evidence of a possible HIV cure yet."
Based on the findings, the study doctors have told participants that they cannot recommend that all study participants receive the kick and kill drugs as well as their ART and that the trial did not find evidence to recommend that any participants could safely stop taking their ART.
Looking forward to possible next steps, the RIVER co-principal investigator and scientific lead, Professor John Frater of the University of Oxford said:
"We need to think about why we didn't see an effect. The important thing to realise is that despite these disappointing results, it does not mean that the basis of the approach is wrong. This is the very first randomised study of the 'kick and kill' concept in humans and the field now needs to work together to explore how better and more effective agents can have an impact on the HIV reservoir while remaining safe.
"It is possible that the combinations of drugs we used weren't quite right, but for this first study we didn't want to compromise on safety by using stronger agents that might work better but could cause toxicity to the participants. It is possible that vorinostat was not quite potent enough to wake up as much HIV as was needed for the newly trained immune system to recognise. Equally, it is possible that a different sort of immune response to the one we induced is needed to target the HIV reservoir. All of these possibilities need to be teased out and considered to guide our next move in searching for an HIV cure."
The vaccines used in RIVER, known as ChAdV63.HIVconsv and MVA.HIVconsv, were highly effective at inducing HIV specific immune responses in the active group. Follow-up research may include giving further boosts of this vaccination together with a different kick drug or possibly using the 'next generation' of these vaccines that are now being trialled in other studies.
RIVER was run by 'CHERUB', a UK collaboration of five universities pooling their expertise to develop an HIV cure. CHERUB is supported by the National Institute for Health Research and brings together HIV researchers at Imperial College London, the University of Oxford, the University of Cambridge, Medical Research Council Clinical Trials Unit at UCL, and King's College London. RIVER was funded by the Medical Research Council, UK and the industry partners, MSD and GSK.
RIVER was the first HIV cure study to use a randomised controlled approach, and the authors urge that this should be repeated in future HIV cure trials. Professor Abdel Babiker of the MRC Clinical Trials Unit at UCL, said:
"Although the results are disappointing, they are unambiguous because of the randomisation and completeness of follow up assessments. Because ART is so effective at reducing viral load, without the randomised control group of participants taking ART alone to compare against, we couldn't have been so confident in knowing whether the kick and kill drugs had made any impact. It's important that future HIV cure trials follow this approach and compare their outcomes to an ART-only group."
During its three years, RIVER benefitted from outstanding commitment from its 60 participants. There was 100% attendance at the primary endpoint study visits, and no participants were lost from the study. In the experience of the doctors and researchers, this was extraordinary and reflects new and deeper levels of commitment from participants.
"They take time off work, they come for lots of visits and tests, and they are an amazingly committed group of people," said Professor Fidler. "They are not just volunteers, they are active advocates for support and they push us to go further all the time. They are helping to define where this research can go next and they are the real pioneers of new treatments."
A participant in the trial said:
"You can be hopeful, but you must be realistic when you take part in a trial. The results from previous trials, the way RIVER was designed and the people running and supporting it, all gave me confidence that it was the right thing to do to take part. So, the initial results are disappointing because of course everyone would have liked to have seen some difference between the control and the active groups. We will have to wait and see what the next move is, but we can't hold back now in the search for a functional cure."
"It's remarkable that nobody dropped out of the trial. It had outstanding commitment from participants because people want to see an HIV cure happen and can see that being involved is the way we are going to push the boundaries back."
Damian Kelly, RIVER community advisory board leader and Director of Patient Advocacy Alliance, said:
"The cost of HIV treatment globally and the new infection rates globally mean that we have to drive towards a cure for HIV or some in-between point such as remission.
"Everyone involved in RIVER - the participants, the clinic staff who have such a critical role in keeping participants engaged and supported, the doctors, the lab technicians and the scientists - everyone should be proud that they were involved in the first randomised controlled HIV cure trial."
"I'm confident that not an ounce of effort that has gone into the RIVER trial will be wasted as these results will help to direct and inform the design of future trials and move us closer towards the goal of a cure."
-ends-
For more information:
Chris McIntyre
University of Oxford
christopher.mcintyre@admin.ox.ac.uk
+44 (0)1865 270 046
Kate Wighton
Imperial College London
k.wighton@imperial.ac.uk
+44 (0)20 7594 2409 / +44 (0)7561 451780
Notes to Editors
1. The RIVER main findings will be presented as Late-breaker oral abstract session at the 22nd International AIDS Conference (AIDS 2018), in Amsterdam, the Netherlands, on Tuesday 24 July 2018, during the session titled 'Strategies for cure: Pitfalls, possibilities and promise'
The AIDS2018 reference is: Abstract TUAA0202LB "A randomised controlled trial comparing the impact of antiretroviral therapy (ART) with a 'Kick-and-Kill' approach to ART alone on HIV reservoirs in individuals with primary HIV infection (PHI); RIVER trial"
Professor Sarah Fidler and Professor John Frater will present the results at a press conference hosted by AIDS2018 at 1000-1045 CEST on Tuesday 24 July. The press conference (titled 'Treatment & Cure: Special Spotlight on Dolutegravir') will be held in Press Conference Room 1 located on level 1 in rooms G-106/G-107, directly above the media centre.
Media information for HIV2018 can be found here: http://www.aids2018.org/Media-Centre
An FAQ for the main findings of RIVER is here:
http://www.cherub.uk.net/faq-for-the-main-findings-of-the-river-trial/
The RIVER participants results briefing - provided to all participants in the study and available for download here: http://www.cherub.uk.net/river-participants-results-briefing/
The RIVER initial analysis scientific report is available for download here: [URL TO FOLLOW]
2. Funding of RIVER: RIVER was funded by the Medical Research Council (Grant ref MR/L00528X/1) in collaboration with support from industry partners. Raltegravir and Vorinostat are manufactured by Merck Sharp and Dohme (MSD). MSD agreed to supply the study drugs free of charge for participants participating in the study. The manufacture of ChAdV63.HIVconsv and MVA.HIVconsv vaccines and all pre-clinical and clinical work to date for these vaccines have been provided free of charge to the RIVER study by Lucy Dorrell and Tomáš Hanke of the University of Oxford utilising their existing MRC grants. GSK own the ChAd vector and have reviewed the RIVER protocol and have approved the use of the ChAdV63.HIVconsv. in the RIVER study.
About the RIVER study
The RIVER trial opened for recruitment in 2015 and concluded in 2018. It is one of the first clinical trials to test a new idea of how to cure HIV, through waking a 'reservoir' of cells infected with sleeping virus and killing them using the immune system. This idea has been called 'Kick and Kill' and this study will investigate if it might work.
The RIVER study is being conducted by the CHERUB collaboration, an alliance of HIV researchers at University of Oxford, Imperial College London, the University of Cambridge, UCL, and King's College London. The study is funded by the Medical Research Council with support from industry partners MSD and GSK.
There are six centres participating in RIVER: Royal Free Hospital, Royal Free London NHS Foundation Trust; St Mary's Hospital, London, Imperial College Healthcare NHS Trust; Mortimer Market Centre, Central and Northwest London NHS Foundation Trust; Chelsea and Westminster Hospital, Chelsea and Westminster Hospital NHS Foundation Trust; St Thomas' Hospital, London, Guy's and St Thomas' NHS Foundation Trust; and Royal Sussex County Hospital, Brighton and Sussex University Hospitals NHS Trust.
http://www.ctu.mrc.ac.uk/our_research/research_areas/hiv/studies/river/
About CHERUB
The CHERUB collaboration ('Collaborative HIV Eradication of Reservoirs UK BRC') is a UK network of internationally recognised doctors and researchers from the NIHR Biomedical Research Centres at Oxford, Cambridge and London working together with patients to find a cure for HIV infection.
The aims of the CHERUB collaboration are to:
• Develop and promote a coordinated UK-wide response to the need to explore strategies targeting HIV eradication and remission.
• Create a biobank of samples for use in scientific research by the collaborative parties, according to agreed study protocols.
• Undertake internationally competitive clinical and scientific research within a framework for sharing of data, resources and consumables.
Website: www.cherub.uk.net
|
| |
|
 |
 |
|
|