 |
 |
 |
| |
Antiactivation activity with kinase inhibitor - JAK Inhbitor - added to suppressive ART / "Safety, tolerability and immunologic activity of ruxolitinib added to suppressive ART"
|
| |
| |
Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2019, Seattle
Mark Mascolini
Added to suppressive antiretroviral therapy (ART), the kinase inhibitor ruxolitinib significantly decreased sCD14, a monocyte activation marker, and boosted circulating T-cell counts in an AIDS Clinical Trials Group (ACTG) study [1]. Ruxolitinib did not affect levels of the inflammation and activation marker IL-6.
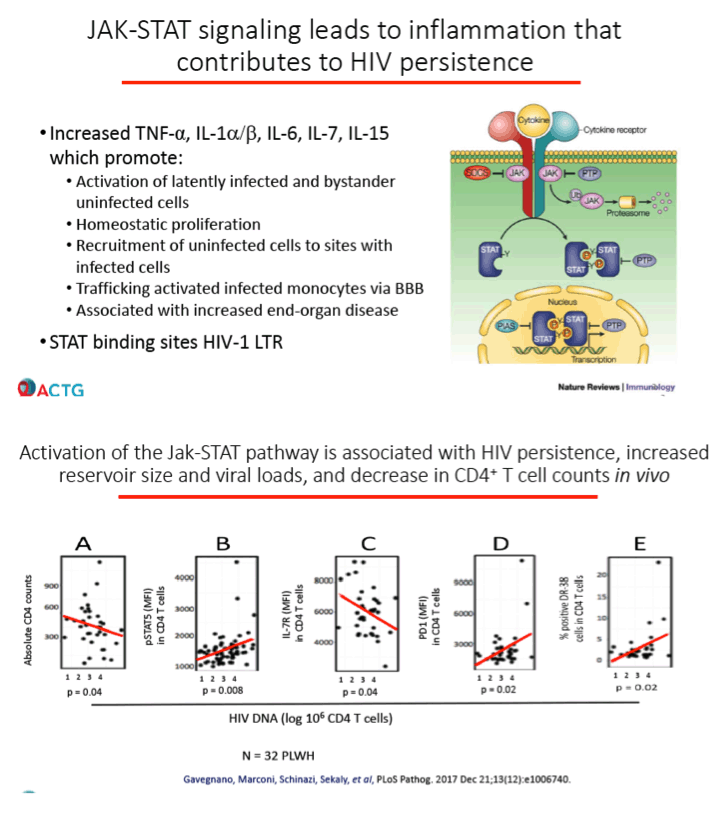
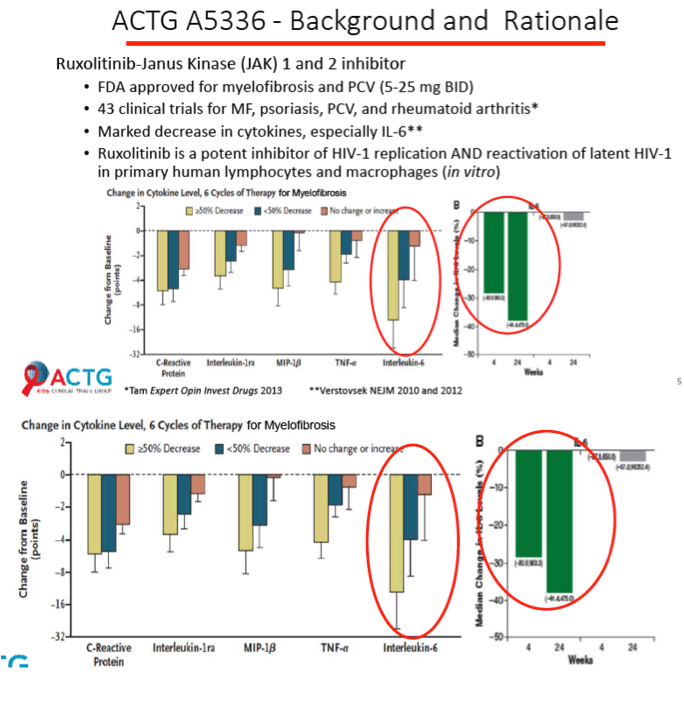
Ruxolitinib is a Janus-associated kinase (JAK) inhibitor licensed as Jakafi for treatment of polycythemia vera and myelofibrosis. Side effects include thrombocytopenia and anemia. ACTG A5336 investigators noted that JAK signaling promotes inflammation that can contribute to HIV persistence. Prior work showed that JAK-STAT activation fosters HIV persistence, increased HIV reservoir size, higher viral load, and lower CD4 count [2]. In vitro studies confirm that ruxolitinib potently inhibits HIV replication and reactivation of latent HIV in primary human lymphocytes and macrophages. CYP3A4 inhibitors boost ruxolitinib levels.
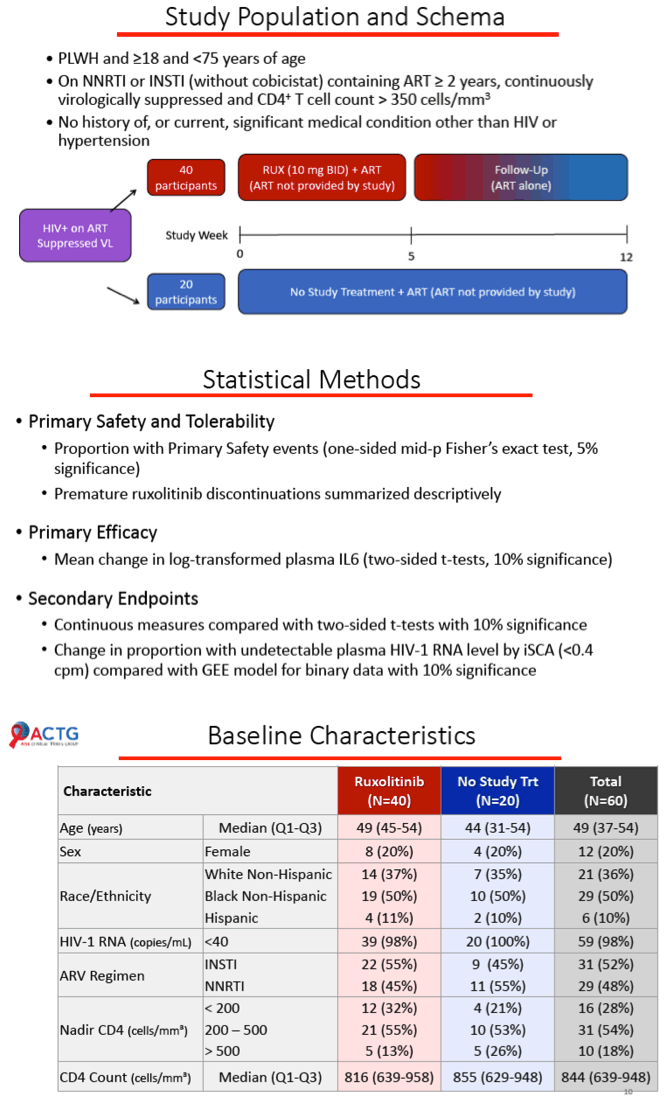
ACTG A5336 enrolled HIV-positive adults with at least 2 years of HIV suppression and a CD4 count above 350 while taking a nonnucleoside or integrase inhibitor (without cobicistat). They could have no significant medical conditions besides HIV infection or hypertension. Researchers randomized 40 people to add ruxolitinib to their ART regimen and 20 to continue ART without ruxolitinib. The JAK inhibitor continued for 5 weeks and follow-up for 12 weeks.
Median age stood at 49 in the ruxolitinib group and 44 in the control group. Half in each group were black, about one third white, and 20% women. Current median CD4 stood at 816 in the ruxolitinib arm and 855 in the control arm.
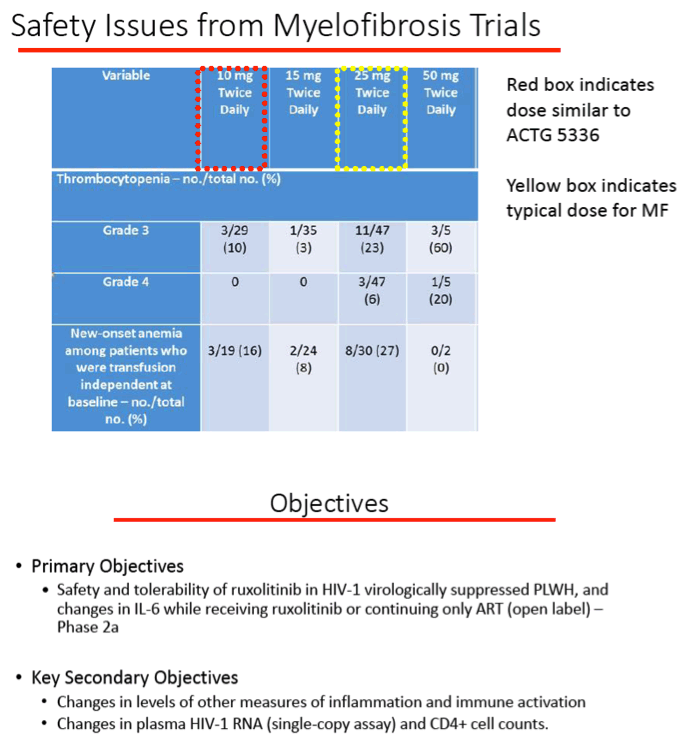
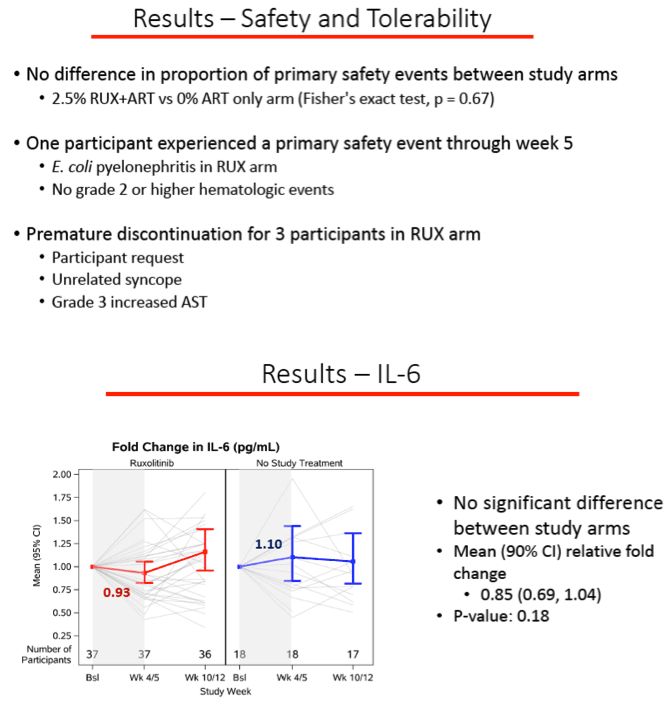
Primary safety endpoints arose in 1 person (2.5%) taking ruxolitinib and in none in the control group (P = 0.67). The 1 person who reached such an endpoint had E coli pyelonephritis and grade 2 or higher hematologic events. Three people stopped ruxolitinib, 1 with a grade 3 liver enzyme elevation, 1 with unrelated syncope, and 1 by request.
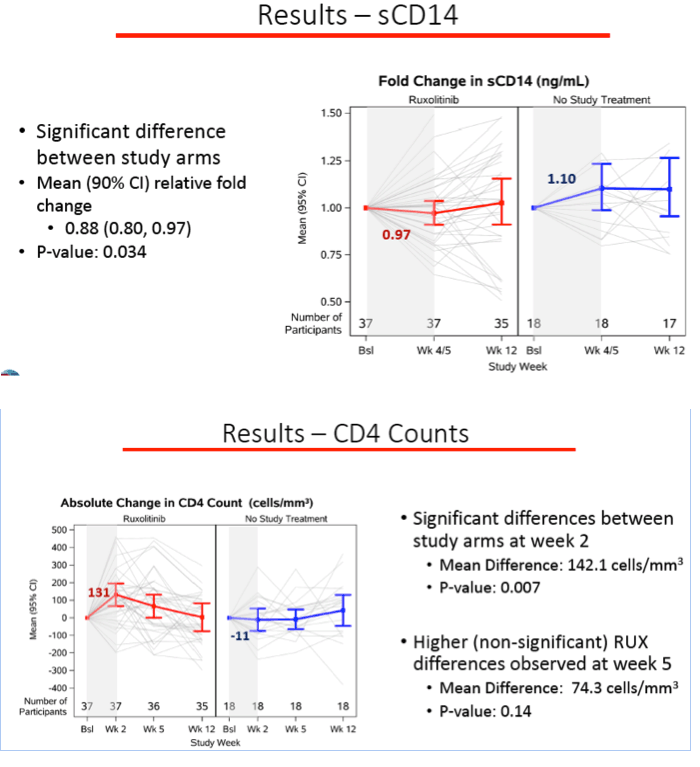
Initial IL-6 and sCD14 levels were normal in both study groups. From baseline to week 12, the inflammation marker IL-6 did not change significantly in either study arm, and the two arms did not differ in IL-6 change. sCD14, the cell activation marker, fell with ruxolitinib and rose in the control group, for an average relative fold change of 0.88 at week 5 (90% confidence interval 0.80 to 0.97) (P = 0.034).
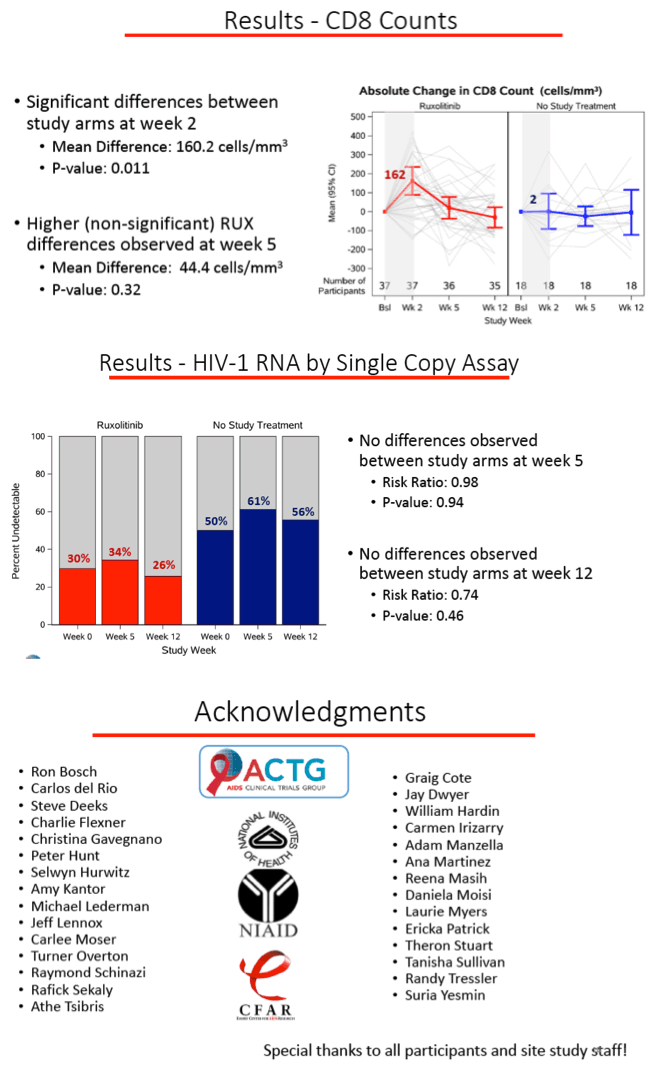
Through the first 2 weeks average CD4 count rose by 131 cells with ruxolitinib while falling 11 cells in the control group, a significant difference (average difference from controls 142 cells, P = 0.007). After 5 weeks of ruxolitinib, the CD4 count change remained nonsignificantly greater with ruxolitinib (average difference from controls 74 cells, P = 0.14). CD8 count, an inflammation signal, also rose significantly with ruxolitinib through 2 weeks (average difference from controls 160 cells, P = 0.011), but the difference had become nonsignificant by week 5 (average difference from controls 44 cells, P = 0.32). Proportion of participants with an HIV load undetectable by a single-copy assay did not change significantly from week 0 to week 5 or 12 in either study arm.
The ACTG team concluded that "in a select, stable cohort" of people with HIV, ruxolitinib proved safe and well tolerated. Participants taking the kinase inhibitor had significant drops in sCD14, a monocyte activation marker, but no change in IL-6. The investigators believe their findings provide "a rationale for future studies of JAK inhibitors in people with HIV who have residual inflammation or immune dysfunction despite long-term suppressive ART."
References
1. Marconi VC, Moser C, Gavegnano C, et al. Safety, tolerability and immunologic activity of ruxolitinib added to suppressive ART. Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2019. Seattle. Abstract 37LB.
2. Gavegnano C, Brehm JH, Dupuy FP, et al. Novel mechanisms to inhibit HIV reservoir seeding using Jak inhibitors. PLoS Pathog. 2017;13:e1006740. https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1006740

|
| |
|
 |
 |
|
|