 |
 |
 |
| |
Factors associated with therapeutic failure of 2-drug regimens, DAT’AIDS cohort: Two-antiretroviral maintenance failure--raltegravir regimens seem most vulnerable
|
| |
| |
Conference on Retroviruses and Opportunistic Infections (CROI), March 4-7, 2019, Seattle
Mark Mascolini
Two-antiretroviral maintenance regimens failed virologically in only 3.5% of 3484 people in a 2010-2017 French analysis [1]. Raltegravir/etravirine (RAL/ETR) and raltegravir plus darunavir/ritonavir (DRV/r/RAL) appeared to be the duos most vulnerable to virologic failure. Other failure risk factors included highest viral load above 100,000 copies and a previous virologic failure.
Two-drug maintenance combinations have gained popularity because of their simplicity, lower toxic potential, and preservation of other antiretroviral classes. But data on real-life safety and virologic failure risk remain sparse. To address those issues, researchers working with the big French Dat'AIDS cohort (NCT02898987) conducted this study.
Dat'AIDS combines data from 22 major French HIV centers. This analysis focused on Dat' AIDS participants taking a 2-antiretroviral maintenance regimen between January 2010 and December 2017 with a viral load below 50 copies. Defining virologic failure as consecutive viral loads above 50 copies during 2-drug maintenance therapy, the Dat' AIDS team considered the five most frequently prescribed 2-drug combos: dolutegravir plus rilpivirine (DTG/RPV), RAL/ETR, DTG/3TC or FTC (XTC), DRV/r/RAL, and DRV/r/XTC. The investigators used Cox proportional hazards models adjusted for sociodemographics, HIV variables, and antiretroviral variables to estimate risk of stopping a 2-drug regimen for virologic failure.
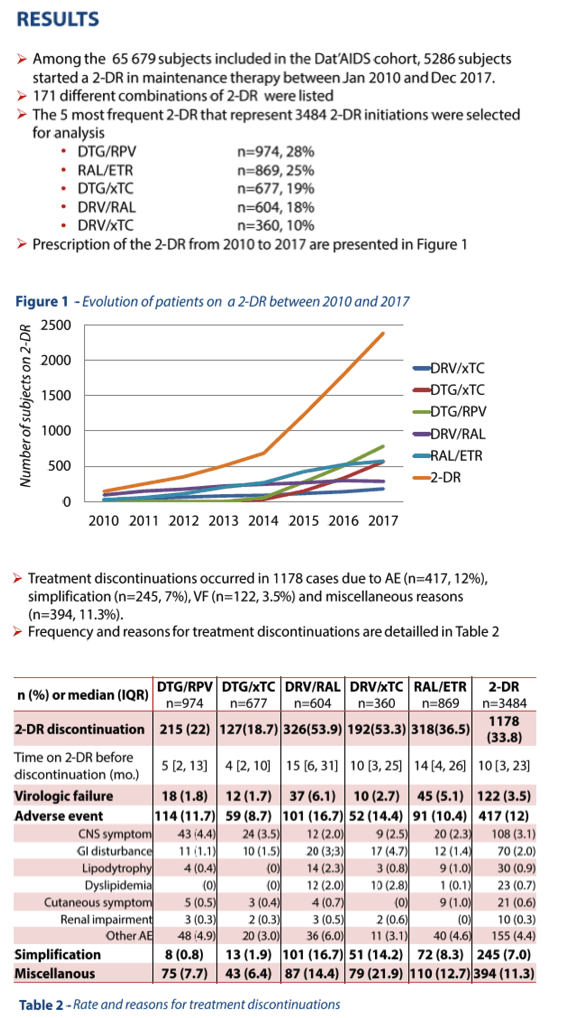
The analysis included 3484 people, 28% taking DTG/RPV, 25% RAL/ETR, 19% DTG/XTC, 18% DRV/r/RAL, and 10% DRV/r/XTC. During the study period only 122 people (3.5%) stopped their 2-drug regimen because of virologic failure. In contrast, 417 people (12%) stopped for adverse events. Quitting for virologic failure proved most frequent with DRV/r/RAL (6.1%), followed by RAL/ETR (5.1%), DRV/r/XTC (2.7%), DTG/RPV (1.8%), and DTG/XTC (1.7%). Quitting for adverse events was also most frequent with DRV/r/RAL (16.7%), followed by DRV/r/XTC (14.4%), DTG/RPV (11.7%), RAL/ETR (10.4%), and DTG/XTC (8.7%).
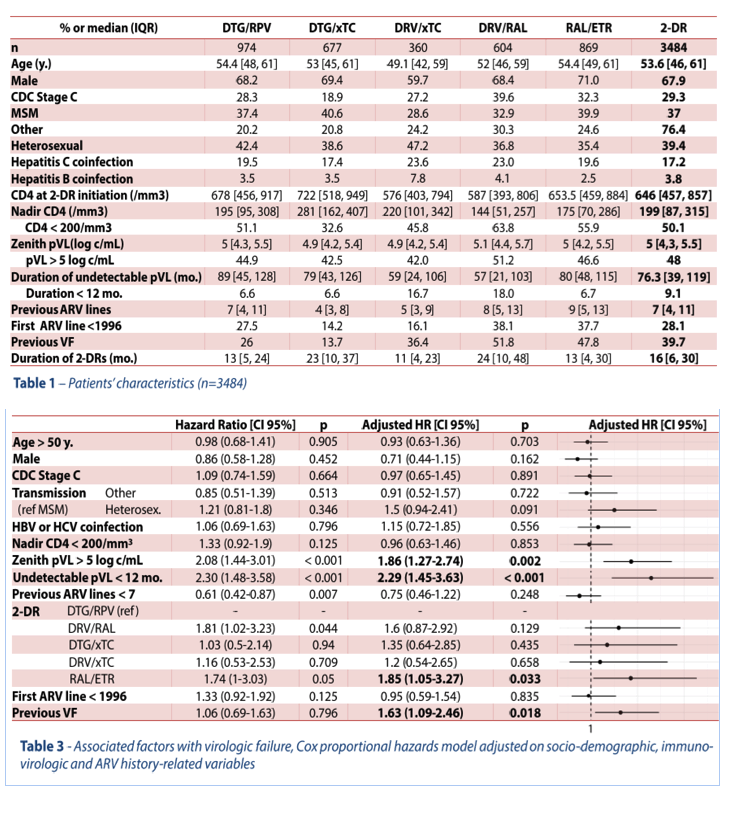
The multivariable analysis singled out RAL/ETR as the only 2-drug regimen associated with virologic failure, almost doubling the risk (see list below). There was a trend toward an association between DRV/r/RAL and virologic failure. Three other independent failure risk factors emerged: highest viral load above 100,000 copies, viral load undetectable for fewer than 12 months, and previous virologic failure, at the noted adjusted hazard ratios (aHR) and 95% confidence intervals (CI):
Risk factors for virologic failure with 2-drug maintenance therapy:
-- RAL/ETR (vs DTG/RPV): aHR 1.85, 95% CI 1.05 to 3.27, P = 0.033
-- DRV/r/RAL (vs DTG/RPV): aHR 1.6, 95% CI 0.87 to 2.92, P = 0.129 (not significant)
-- Highest viral load above 100,000 copies: aHR 1.86, 95% CI 1.27 to 2.74, P = 0.002
-- Viral load undetectable fewer than 12 months: aHR 2.29, 95% CI 1.45 to 3.63, P < 0.001
-- Previous virologic failure: aHR 1.63, 95% CI 1.09 to 2.46, P = 0.018
Age, gender, nadir CD4 count, first antiretrovirals before 1996, and other variables did not predict virologic failure in this analysis.
The Dat'AIDS team underlined the nonsignificant association between DRV/r/RAL and failure, suggesting that regimen "could be associated with more virologic failure" but that the association failed to reach significance because of insufficient statistical power. Among the 5 regimens analyzed, DRV/r/RAL had the highest proportion of people quitting because of failure (6.1%).
Reference
1. Allavena C, Deschanvres C, Peytavin G, et al. Factors associated with therapeutic failure of 2-drug regimens, DAT’AIDS cohort. Conference on Retroviruses and Opportunistic Infections (CROI). March 4-7, 2019. Seattle. Abstract 493.

Clotilde Allavena1, Colin Deschanvres1, Gilles Peytavin2, David Rey3, Marc-Antoine Valantin4, Firouze Bani-Sadr5, Olivier Robineau6, Claudine Duvivier7, Andre Cabie8, Laurent Hocqueloux9, Lise Cuzin8, Veronique Joly2, Antoine Cheret10, Jacques Reynes11
1CHU de Nantes, Nantes, France,2AP-HP, Hopital Bichat-Claude Bernard, Paris, France,3Hopitaux Universitaires de Strasbourg, Strasbourg, France,4AP-HP, Hopitaux Universitaires Pitie Salpetrière, Paris, France,5CHU de Reims, Reims, France,6Centre Hospitalier de Tourcoing, Tourcoing, France,7Necker Hospital, Paris, France,8CHU Fort de France, Fort de France, Martinique,9Centre Hospitalier Regional d'Orleans, Orleans, France,10Hopital Bicetre, Le Kremlin-Bicetre, France,11CHU de Montpellier, Montpellier, France

|
| |
|
 |
 |
|
|