 |
 |
 |
| |
GREATER WEIGHT GAIN AMONG TREATMENT-NAIVE
PERSONS STARTING
INTEGRASE INHIBITORS
|
| |
| |
Reported by Jules Levin
CROI 2019 March 4-7 Seattle
Kassem Bourgi1, Cathy Jenkins1, Peter F. Rebeiro1, Jordan E. Lake2, Richard
D. Moore3, W. C. Mathews4, Michael A. Horberg5, Amanda Willig6, Michelle
Floris-Moore7, Michael John Gill8, Angel M. Mayor9, Ronald Bosch10, Timothy R.
Sterling1, John R. Koethe1, for the North American AIDS Cohort Collaboration on
Research and Design (NA-ACCORD) for IeDEA,
1Vanderbilt University, Nashville, TN, USA, 2University of Texas at Houston, Houston,
TX, USA, 3Johns Hopkins University, Baltimore, MD, USA, 4University of California San Diego, San Diego, CA, USA, 5Kaiser Permanente Mid-Atlantic States, Rockville, MD, USA, 6University of Alabama at Birmingham, Birmingham, AL, USA, 7University of North Carolina at Chapel Hill, Chapel Hill, NC, USA, 8Alberta Health Services, Calgary, AB, Canada, 9Universidad Central del Caribe, Bayamon, Puerto Rico, 10Harvard University, Boston, MA, USA

Program abstract
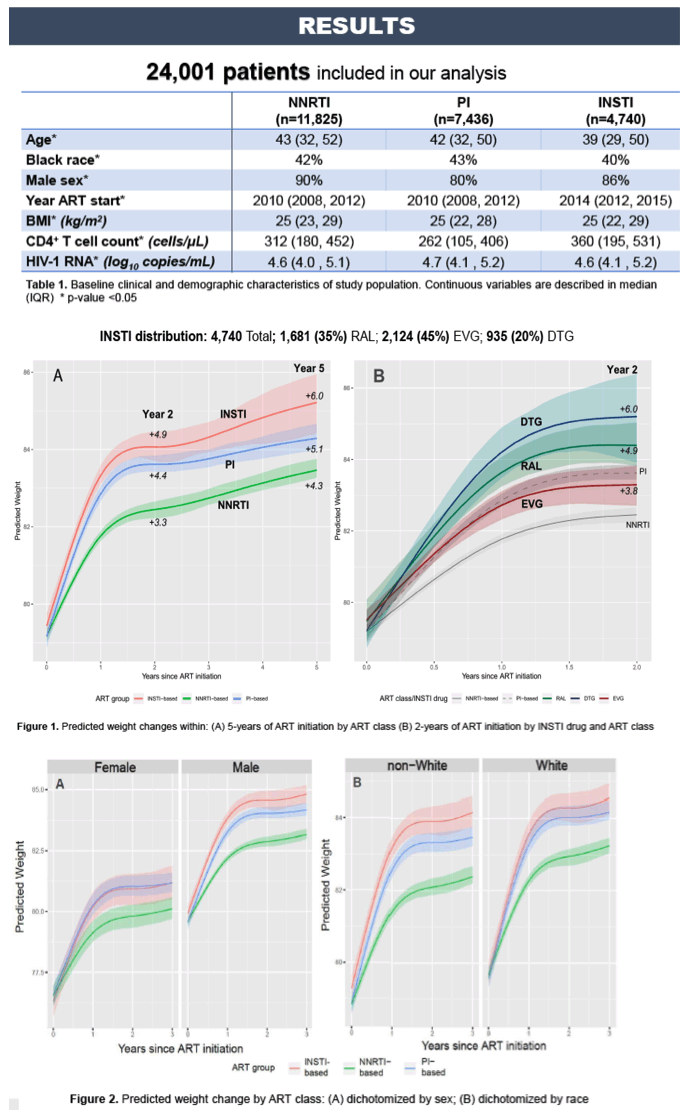
Obesity among persons living with HIV (PLWH) has steadily increased in the era of combination antiretroviral therapy (ART), and some PLWH experience substantial weight gain after initiating ART that may lead to metabolic comorbidities and poorer survival. To assess the influence of antiretroviral class and integrase strand transfer inhibitors (INSTI) on this phenomenon, we compared weight changes after initiating ART among treatment-naïve PLWH in the North American AIDS Cohort Collaboration on Research and Design (NA-ACCORD).
Adult, treatment-naïve PLWH in NA-ACCORD initiating INSTI, protease inhibitor (PI), and non-nucleoside reverse-transcriptase inhibitor (NNRTI)-based ART regimens after 01/01/2007 were included and followed through 12/31/2015. We used multivariate linear mixed effects models to generate marginal predictions of weights over time, adjusting for age, sex, race, cohort site, HIV acquisition mode, ART initiation year, and baseline weight, HIV-1 RNA, and CD4+ cell count. We used restricted cubic splines to relax linearity assumptions, multiple imputation for missing values, and bootstrapping to generate 95% confidence intervals. Predicted weights by ART class were reported at years 2 and 5. Due to shorter follow-up for newer INSTI drugs, predicted weights for raltegravir (RAL), elvitegravir (EVG), and dolutegravir (DTG) were reported at years 1 and 2.
Among 21,886 participants, 4,112 initiated INSTI-based regimens (2106 RAL, 1510 EVG and 477 DTG), 87% were male, and 43% were white. At ART initiation median age was 42 years, BMI was 25 kg/m², and CD4+ count was 303 cells/mm³. Weight gain was highest among PLWH starting INSTI (Figure1, A). At 2 and 5 years, PLWH on INSTI gained 4.4 and 5.8 kg, respectively, compared to 3.3 and 4.1 kg for NNRTI (pTreatment-naïve PLWH starting INSTI, especially DTG and RAL, are at higher risk of weight gain compared to older NNRTI-class regimens. This is clinically important as INSTI-based regimens are now recommended first line ART and PLWH are at increasing risk for obesity, metabolic comorbidities, and cardiovascular disease.

References:
1.KoetheJRetal.
RisingObesityPrevalenceandWeightGainAmongAdultsStarting
AntiretroviralTherapyintheUnitedStatesandCanada.
AIDSresearchandhumanretroviruses2016;32(1):50-8.
2. Grant PM et al. Long-term body composition changes in antiretroviral-treated HIV-infected individuals. AIDS 2016; 30(18): 2805-13.
3. Koethe JR et al. Higher Time-Updated Body Mass Index: Association With Improved CD4+ Cell Recovery on HIV Treatment. JAIDS 2016; 73(2): 197-204
4. Norwood J et al. Brief Report: Weight Gain in Persons With HIV Switched From Efavirenz-Based to Integrase Strand Transfer Inhibitor-Based Regimens. JAIDS 2017; 76(5): 527-31.
5. McComsey GA et al. Body composition changes after initiation of raltegravir or protease inhibitors: ACTG A5260s. CID 2016; 62(7): 853-62.
6. Reynes J et al. Lopinavir/ritonavir combined with raltegravir or tenofovir/emtricitabine in antiretroviral-naive subjects: 96-week results of the PROGRESS study. AIDS research and human retroviruses 2013; 29(2): 256-65
|
| |
|
 |
 |
|
|