 |
 |
 |
| |
PML-IRIS ensuing CNS and subsequent peripheral
viral escape of ARV-resistant variants
|
| |
| |
Reported by Jules Levin
CROI 2019 March 4-7 Seattle
Andrea Lisco1, Camille Lange2, Bryan Smith3, Maura Manion1, Lindo Natalie2, Dima Hammoud, Angela Kashuba, John Donhal, Alice Pau1, Safia Kuriakose1, Megan Anderson1, Pawel Murawnski5, Quan Yu5, Frank Maldarelli2 and Irini Sereti1
1Laboratory of Immunoregulation, National Institute of Allergy and Infectious Diseases, Bethesda, MD USA.2 HIV DRP Host-Virus Interaction Branch, National Cancer Institute Frederick, MD USA.3National Institute of Neurological Disorders and Stroke, Bethesda MD, USA.4 Center for Infectious Disease Imaging, Bethesda, MD USA.5Hematology Branch of the National Heart, Lung and Blood Institute, Bethesda MD USA

Program Abstract
CNS ESCAPE OF DRUG-RESISTANT HIV IN PML-IRIS AND CONSEQUENT PERIPHERAL DISSEMINATION
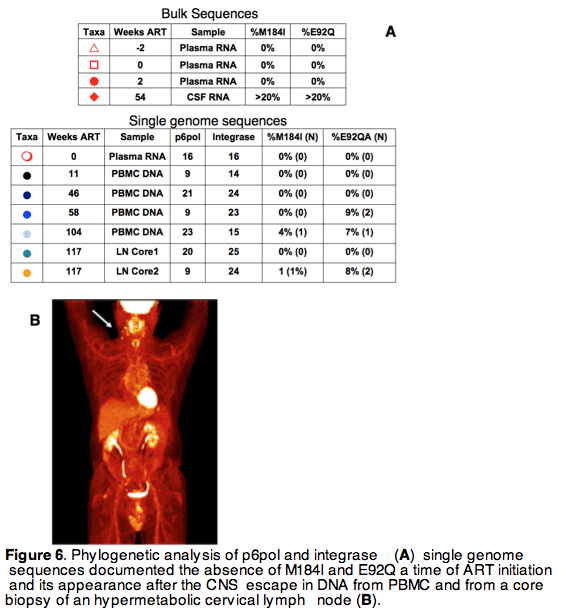
Inflammation and tissue influx of activated T cells affects the compartmentalization and dynamics of HIV viral replication, but the role of compartmentalized virus populations in the emergence of drug resistant mutations (DRMs) and virological failure is uncertain. Herein, we describe viral escape with high viral RNA levels and DRMs in CSF during an episode of unmasking PML-IRIS
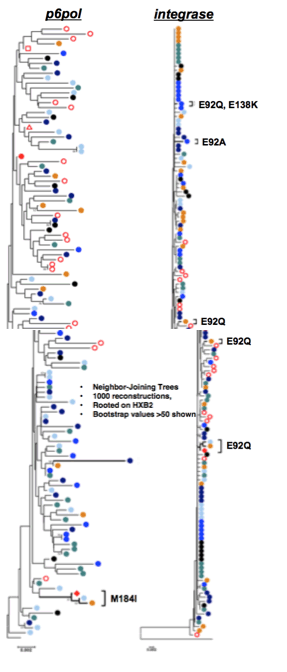
PBMCs and CSF mononuclear cells were used for immunophenotyping along with a flow cytometric T cells responses assay to JC virus (JCV), BKV and CMV. Plasma and CSF ARV concentrations were measured by tandem mass spectrometry. Phylogenetic analysis of CSF, plasma, rectal mucosa and cervical lymph node HIV variants was performed
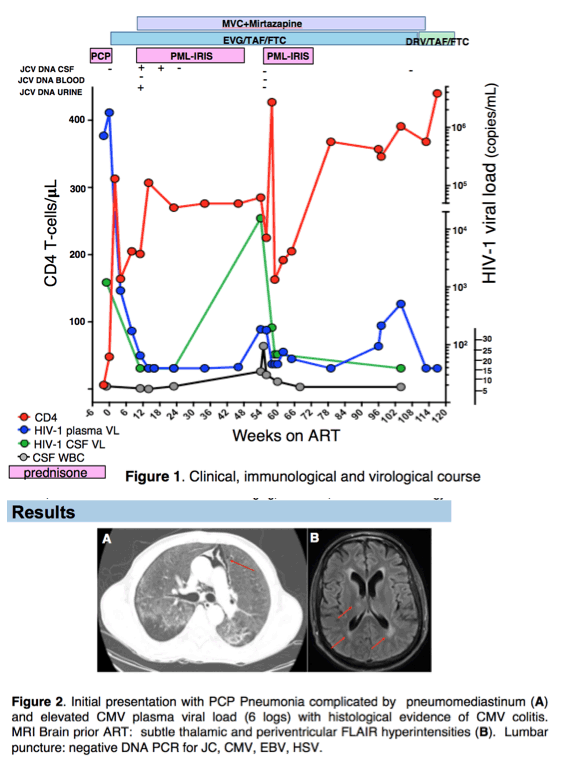
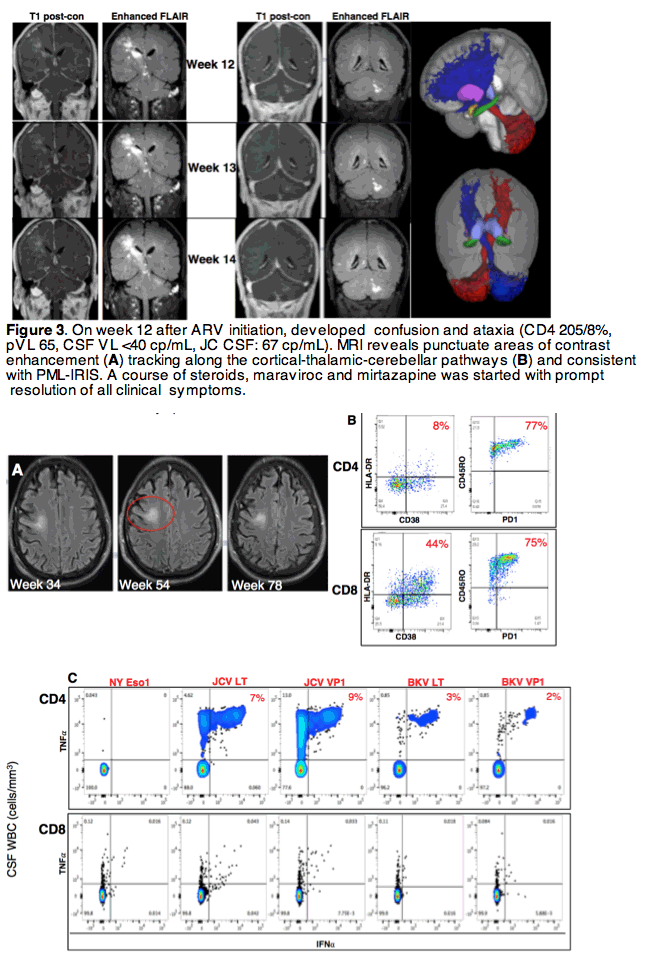
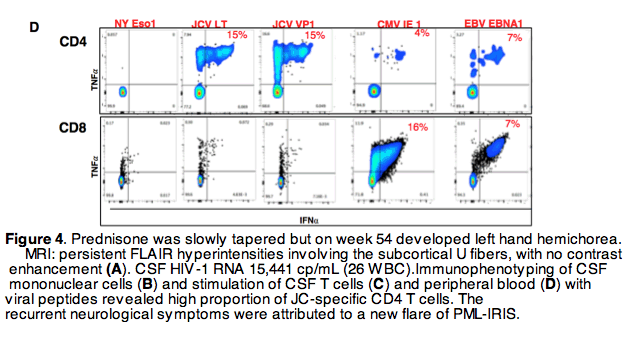
A man with HIV/AIDS [CD4: 6 cells/μL, HIV plasma viral load (VL): 716531 c/mL, CSF VL: 1200 c/mL, no DRMs] presenting with PCP, achieved prompt clinical improvement after TMP-SMX, Prednisone and cEVG/TAF/FTC initiation. 12 weeks after starting ARV, with suppressed HIV VL 300 fold lower than concurrent plasma EVG (1730 ng/mL). Robust specific CD4 T cells responses to JCV, but not to BKV or CMV were documented in CNS and peripheral blood at the time of PML-IRIS flare
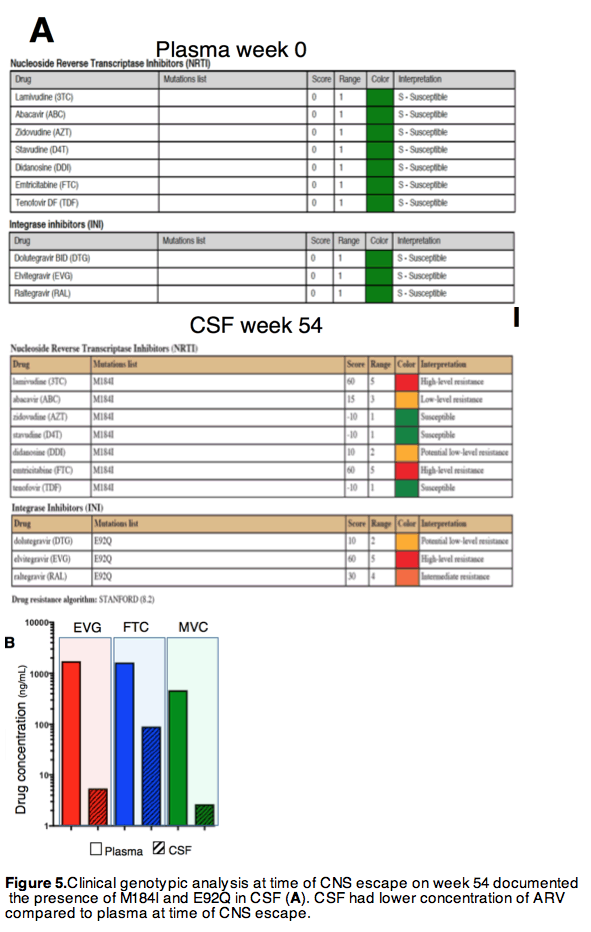
Rebound viremia with DRMs emerged in the setting of CNS immune activation from PML-IRIS and suboptimal CSF EVG levels. HIV-1 RNA was >100 fold higher in CSF than plasma, suggesting selection of resistant variants in CNS and consequent dissemination to peripheral blood and lymphnode. The case highlights the role of localized inflammatory processes and trafficking of immune cells in shaping HIV populations in CNS, plasma and lymphoid tissue

|
| |
|
 |
 |
|
|