 |
 |
 |
| |
Tenofovir-diphosphate in PBMC following increasing
TAF vs. TDF dosing under directly observed therapy
|
| |
| |


program abstract:
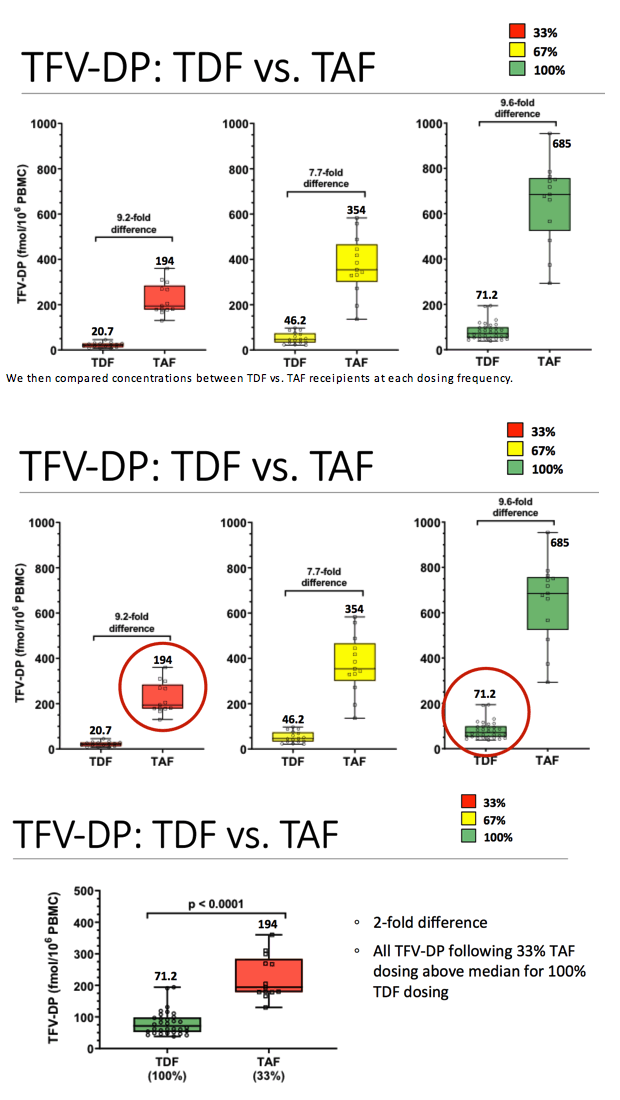
Conclusion: Across the three dosing arms, TFV-DP concentrations in PBMC were 9.2, 7.7, and 9.6-fold higher following TAF/FTC compared with TDF/FTC for 33%, 67%, and 100% dosing arms, respectively. Additionally, TFV-DP was twice as high in the 33% dosing arm for TAF as compared with the 100% dosing arm for TDF. Together, this study shows that the enhanced delivery of tenofovir to target cells with TAF compared to TDF occurs across dosing/adherence levels. These results are relevant for studying alternative dosing strategies and pharmacologic forgiveness for HIV treatment and prevention.
Results: Forty-four TDF/FTC participants contributed 63 data points; 15 from 33%, 16 from 67%, and 32 from 100% daily dosing. Twenty-eight TAF/FTC participants contributed 39 data points; 13 from 33%, 13 from 67%, and 13 from 100% daily dosing. Median (IQR) TFV-DP for TDF versus TAF in the 33% arm was: 21 (15, 28) versus 194 (179, 269) fmol/106 cells; in the 67% arm was: 46 (33, 71) versus 354 (329, 445) fmol/106 cells; and in the 100% arm was: 71 (53, 97) versus 685 (566, 751) fmol/106 cells, respectively (p<0.001 for all comparisons).

Alright, so to briefly go through some background. As we know, there are two prodrugs of tenofovir that are currently available. The first prodrug being tenofovir disoproxil or TDF, and more recently, tenofovir alafenamide or TAF. TAF is more stable in plasma compared to TDF and is primarily converted to tenofovir intracellularly by cathepsin A. Tenofovir is then phosphorylated intracellularly to its active form, tenofovir diphosphate. Due to the increased stability of TAF in plasma, and the selective cleavage of TAF by cathepsin A, more tenofovir is able to load target cells. Previous studies have looked at TFV and TFV-DP levels following TDF vs. TAF-containing regimens and found that this translates into lower plasma tenofovir and lower tfv-dp in DBS. Additionally, TAF results in higher TFV-dp in PBMCs, or target cells.

To our knowledge, TAF has not been directly compared to TDF in studies using directly observed therapy, or DOT. TAF and TDF have also not been directly compared in studies with less than daily dosing. Therefore, the objective of this study is to compare TFV-dp concentrations in target cells, or PBMCs, following increasing TDF/FTC vs TAF/FTC dosing under directly observed therapy.
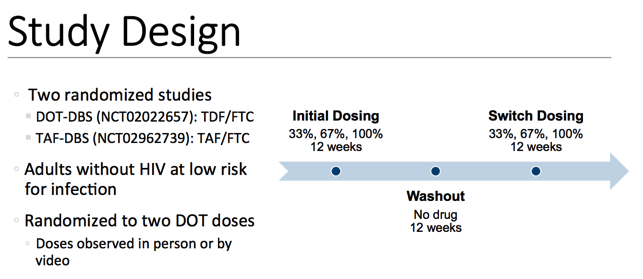
There were 2 randomized studies conducted. The first of which being DOT DBS, where participants received TDF/FTC. The second study was TAF DBS, where participants received TAF/FTC. In both studies, participants were adults without HIV at low risk for infection. These participants were randomized to two directly observed dosing sequences. Doses were observed either in person or by video. In both studies, the initial dosing regimen was either 33, 67, or 100% daily dosing for a 12 week duration, followed by a 12-week washout period, and a second dosing regimen that differed from the initial dosing regimen, but was again wither 33, 67, or 100% dosing.

For 33%, participants received a dose for one day, followed by two days without dosing. For 67%, participants received two days of dosing, followed by one day without dosing. And for 100% participants dosed daily throughout the 12-week dosing period.
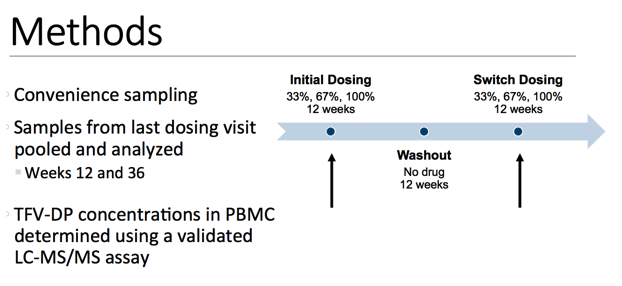
In both studies, convenience sampling was done throughout the 36-week study period. Samples from the last dosing visit, which would be weeks 12 and 36, were pooled and analyzed. Tenofovir diphosphate concentrations were then determined using a previously validated assay.
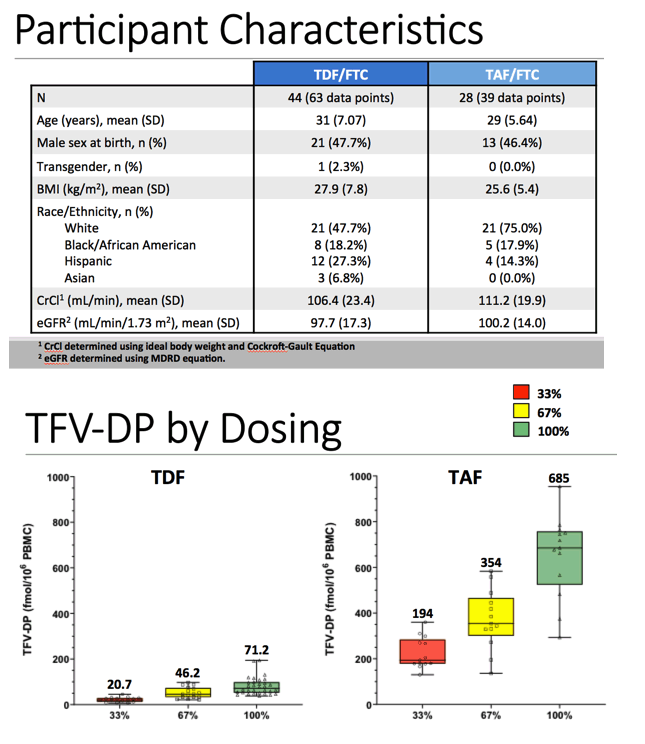
Here you can see tfv-dp concentrations in PBMCs following TDF (on the left) and TAF (on the right) after 33% in red, 67% in yellow and 100% daily dosing in green. You can see that among both those who received TDF and TAF, TFV-DP concentrations in PBMC increase with increasing dosing frequency. Specifically looking at those who received TDF on the left, median TFV-DP concentrations in PBMCs were aprroximately 21, 46, and 71 fentimols per million cells among those who received 33%, 67%, and 100% daily dosing. Then, for TAF, median concentrations were 194, 354, and 685 fentimols per million cells among those who received 33%, 67%, and 100% daily dosing.
We then compared concentrations between TDF vs. TAF receipients at each dosing frequency.

|
| |
|
 |
 |
|
|