 |
 |
 |
| |
Pegylated interferon alfa/ribavirin in persons who failed to prior interferon based therapy
Written for NATAP by Mark Sulkowski, MD, Johns Hopkins University School of Medicine
|
| |
| |
The majority of patients, particularly those with HCV genotype 1, treated with standard interferon alfa-2b plus ribavirin fail to achieve a sustained virologic response. Thus, there is considerable interest in the efficacy of pegylated interferons plus ribavirin in the treatment of HCV infection in persons who failed prior combination therapy. Several studies at the 52nd AASLD meeting in beautiful Dallas, TX addressed this important question.
Abstract 277. Analyses of 40 KDA Peginterferon Alfa-2a (Pegasys) in Combination With Ribavirin, Mycophenolate Mofetil, Amantadine, or Amantadine Plus Ribavirin In Patients That Relapsed or Did Not Respond To Rebetron_ Therapy: A Report Of Two Randomized, Multicenter, Efficacy And Safety Studies
Dr. Nid Afdhal presented on-treatment and end-of-treatment data from two randomized, controlled studies which retreated patients who had previously failed to suppress HCV RNA ("non-responders" or had experienced a re-emergence of HCV RNA following discontinuation of therapy ("relapsers") with PEG-IFN alfa-2a (180 mcg SC weekly) plus the following adjuvant therapies: 1) ribavirin 1 - 1.2 grams/day (RBV); 2) mycophenlate mofetil (CellCept) 1 gram twice daily (MMF); 3) amantadine 200 mg twice daily (AMA); 4) ribavirin 1 - 1.2 grams/day and amantadine 200 mg twice daily (RBV +AMA).
|
|
| |
|
|
| |
 |
|
| |
|
|
| |
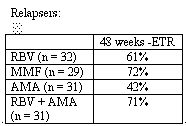 |
|
| |
|
|
| |
This ongoing study indicates that some patients who failed to respond to standard interferon/ribavirin may benefit from an additional course of therapy with pegylated interferon plus other agents. However, these results must be viewed with several caveats. First, it is anticipated the approximately 20% of EOT responders will experience a virologic relapse after a course of interferon plus ribavirin. Thus, the sustained response rates will be considerably lower for all treatment groups. In addition, ribavirin prevents relapse and there is little reason to believe that MMF or AMA will prevent relapse in a similar manner. Consequently, one may expect the sustained response rates to be considerably lower in the MMF and AMA groups than in the groups that received ribavirin. Furthermore, the medical rationale for the use of either MMF or AMA is dubious at best and, while new and better therapies are urgently needed, it remains doubtful that these agents will have any role in the future management of chronic HCV infections.
Abstract 279. Retreatment of Interferon And Interferon-Ribavirin Non-Responders with Peginterferon-alpha-2a (Pegasys) And Ribavirin: Initial Results from the Lead-In Phase of the Halt-C Trial
The HALT-C trial was designed to determine if long term maintenance interferon therapy, administered over 4 years, could prevent histologic progression, reduce the development of hepatocellular carcinoma and the need for hepatic transplantation in patients with chronic HCV and advanced fibrosis or cirrhosis who remain HCV-RNA (+) despite therapy. Since peginterferon plus ribavirin is more effective than standard interferon/ribavirin for treatment of chronic HCV all patients enrolled in the HALT-C trial were first treated with peginterferon plus ribavirin during the lead-in phase of this trial.
This study enrolled patients with advanced liver disease (Ishak Fibrosis stage 3) and no evidence of hepatic decompensation. All patients had previously failed to respond to interferon-based therapy administered for at least 12 weeks. After screening, all patients received pegylated interferon alfa-2a (180 mcg weekly) plus ribavirin (1 - 1.2 grams/day).
Dr. Adrian Di Bisceglie presented on-treatment virologic response data on the first 268 patients who completed at least 20 weeks of therapy within this ongoing study. For the purpose of this presentation, viral response was defined as an undetectable HCV-RNA level at treatment week 20. 15 (5.6%) patients discontinued therapy prior to week 20. At week 20, 107 of 268 patients (40%) (based on an intention-to-treat analysis) had an undetectable HCV RNA level. These patients will continue therapy for an additional 28 weeks and, per protocol, will not enter the "maintenance" phase of the study. There were several important "sub-group" analysis presented. Persons who had only failed prior interferon monotherapy were much more likely to achieve a response than those who failed prior interferon/ribavirin combination therapy (53% > 31%) and African-Americans were significantly less likely to respond than Caucasians (10% < 41%).
In addition, a large number of patients had dose reductions of the PEG-interferon (53%), ribavirin (44%) or both (47%). Thus, while the discontinuation rate was quite low (~5%), it remains uncertain if such dose reductions will impair the overall effectiveness of the therapy. Nonetheless, these results are encouraging and suggest that persons with advanced liver disease who failed to respond virologically to prior therapy may benefit from a course of pegylated interferon/ribavirin.
Abstract 987. Peg-Interferon alpha-2b Plus Ribavirin for Treatment of Patients with Chronic Hepatitis C Who Have Previously Failed to Achieve A Sustained Virologic Response Following Interferon alpha-2b or Interferon alph-2b Plus Ribavirin Therapy
In a poster session, Dr. Mark Sulkowski and colleagues presented preliminary data from an ongoing multicenter study of pegylated interferon alfa-2b (weight-based 1.5 mcg/kg weekly or fixed dose 100 mcg < 80 kg and 150 mcg > 80 kg weekly) plus ribavirin 800mg/day in patients who failed to achieve a sustained response following interferon-based therapy ("non-responders" and "relapsers").
Of 511 enrolled subjects, viral response data after 12-weeks of therapy was available on 481 patients. Overall, 35% of patients had an undetectable HCV RNA level after 12 weeks of therapy and no significant difference was observed in viral response or adverse events between patients who received fixed versus weight-based PEG-interferon dosing. The week 12 viral response rate was 26.5% among "non-responders" and 55.2% among "relapsers." As has been observed in other studies, persons with HCV genotype 1 and African-Americans were less likely to respond virologically. In this study patients will remain on therapy for an additional 36 weeks. It is anticipated that approximately 15-20% of the end-of -treatment viral responders will experience a "relapse." Thus, while the sustained viral response data is not yet available, it is likely that the observed rates will be lower than on-treatment results.
Conclusions:
Based on this and other data, persons who failed to achieve a sustained viral response to interferon alfa/ribavirin may consider a course of pegylated interferon alfa/ribavirin with the expectation of a response rate in the neighborhood of 20% for "non-responders" and 50-60% for relapsers. The response rates anticipated for "non-responders" is somewhat higher than might have been predicted and may reflect the increased effectiveness of pegylated interferon as well as dramatic improvements in the skill and expertise of the medical professional, who have learned how to use interferon/ribavirin more effectively since the introduction of combination therapy in 1998.
While encouraging, these data clearly indicate that many patients, particularly those with HCV genotype 1 and African-Americans, will not achieve HCV eradication with currently available treatments; new drugs are urgently needed for such patients.
Editorial note: 2 additional studies, one presented by ira Jacobson, MD, at Cornell-NY Hospital in NYC, reported similar findings.
|
|
| |
|
 |
 |
|
|