| |
Comparison of the efficacy, safety and predictive value of HIV genotyping using distinct ritonavir-boosted protease inhibitors
|
| |
| |
Immediately below is a summary of this study and following that is the full text and data results from the study comparing Kaletra vs saquinavir 1000mg/100 mg ritonavir vs indinavir 800 mg/100 mg ritonavir, all taken twice daily plus nukes. The patients had extensive prior treatment experience and were considered "salvage".
ABSTRACT SUMMARY: The pharmacokinetic profile of protease inhibitors (PI) is improved significantly by adding low doses of ritonavir (rit). The use of ritonavir-boosted PI allows the reduction of pill burden, improves dosing schedules and enhances drug exposure, all factors that have been associated with a greater benefit of antiretroviral therapy.
All patients receiving ritonavir 100 mg bid together with saquinavir (SQV) soft gel 1000 mg bid, indinavir (IDV) 800 mg bid or lopinavir (LPV) 400 mg bid, as part of a salvage regimen, were retrospectively analyzed in a reference institution. Only subjects who had failed all three antiretroviral drug families in the past were included.
A total of 299 patients were included in the study (121 on SQV, 62 on IDV and 116 on LPV). Mean plasma HIV-RNA and CD4 lymphocyte counts at baseline were less favourable in the LPV group (4.4 logs, 275 cells/mm3) with respect to SQV (4.3 logs, 355 cells/mm3) and IDV (3.7 logs, 409 cells/mm3) groups (P < 0.05).
The proportion of subjects experiencing virological success (attainment of either < 500 HIV-RNA copies/ml or > 1 log reduction) in an intent-to-treat analysis at 24 weeks was 61% with LPV, 49% with SQV and 48% with IDV (NS). The CD4 count increased 48% with SQV, 45% with IDV and 37% with LPV. The proportion of subjects discontinuing therapy due to adverse events was higher using IDV (27%) than using either SQV (11%) or LPV (6%) (P < 0.05).
The presence of < 5 or > 5 PI resistance mutations at baseline discriminated significantly better who would respond to therapy in all instances: 90 vs. 48% with LPV, 95 vs. 21% with SQV and 90 vs. 23% with IDV. The efficacy of ritonavir-boosted PI combinations is greatly influenced by the extent of baseline PI resistance. Differences, not only in potency, but mainly in tolerance may favour the selection of one dual PI combination over others.
Int J Antimicrob Agents 2002 Dec;20(6):438-43
Barreiro P, Camino N, de Mendoza C, Valer L, Nunez M, Martin-Carbonero L, Gonzalez-Lahoz J, Soriano V.Service of Infectious Diseases, Instituto de Salud Carlos III, Madrid, Spain.
FULL ARTICLE
Introduction
The natural history of HIV infection changed dramatically since protease inhibitors (PI) became available for wide prescription in 1996 [1]. These compounds provide a 2-3 log reduction in plasma HIV-RNA in parallel with increases in CD4 counts of 100-150 cells/l during the first 3 months of treatment [2]. However, the initial enthusiasm generated by the reduction in morbidity and mortality in patients receiving PI-containing regimens has been blunted over time as long-term side effects (i.e. lipodystrophy) [3], difficulties related to high tablet burden and complicated dosing became manifest [4]. These limitations often result in poor drug compliance and consequently treatment failure [5 and 6].
The pharmacokinetic profile of PI deserves special attention. Both limited oral absorption and rapid hepatic clearance through cytochrome P450 enzymes
account for the poor bioavailability and short half-life of PI. These factors, together with a high affinity for plasma proteins, force the administration of most PI in bothersome three times daily schedules [7 and 8]. Even so, the relationship between trough and therapeutic levels, referred to as the inhibitory quotient (IQ), remains for many PI slightly above unity.
Many of these clinical and pharmacokinetic limitations of PI can be overcome by adding low doses of ritonavir (rit), which exert a potent inhibition of
cytochrome P450 3A [9 and 10]. In this form, the other PI can be administered twice or even once daily, using fewer tablets and improving their respective IQ.
Moreover, rit seems to inhibit membrane glycoprotein pumps that also may limit the oral absorption and distribution of other PI [11 and 12].
As adherence and pharmacokinetic factors rather than the selection of resistant viruses often cause the failure of classical PI-containing regimens [13, 14 and 15], rit-boosted PI combinations are currently among the preferred options for salvage therapy. Furthermore, since an enhanced IQ for some PI may allow low-level resistance to be overcome [16 and 17], rit-boosted PI combinations are also attractive options even when a loss of susceptibility to PI has developed or is clearly suspected.
We describe the performance of three different rit-boosted PI combinations in a large group of subjects experiencing virological failure after exposure to all
current antiretroviral drug families. Moreover, we report the predictive value of baseline HIV genotyping for each of these salvage interventions.
Patient choice
Clinical charts of all HIV-positive outpatients attending a reference centre for HIV/AIDS located in Madrid and receiving rit-boosted PI combinations, were
reviewed. The recruitment of patients was limited to a period beginning in May 2000 and ending in May 2001. Only subjects fulfilling the following criteria
were recruited: age >18 years-old, plasma HIV-RNA >500 copies/ml at the time of beginning rit-boosted PI therapy and past exposure to antiretrovirals from
all three drug families. In order to achieve study uniformity, only subjects in which rit was prescribed as capsules at doses of 100 mg bid were chosen.
Likewise, the study was limited to patients receiving the active PI as follows: saquinavir (SQV) soft gel 1000 mg bid, indinavir (IDV) 800 mg bid and lopinavir
(LPV) 400 mg bid--as part of Kaletra.
Demographic features (age, gender, risk behaviour), prior antiretroviral drug experience and the number of novel drugs in the rescue regimen were registered according to clinical records. Plasma HIV-RNA, CD4 cell counts, drug-related toxicities and treatment adherence measured using the pharmacist pill count record, were registered at baseline and 24 weeks after beginning the rit-boosted PI regimen.
Clinical assessment
Plasma HIV-RNA was measured using the third generation bDNA assay (Bayer, Barcelona, Spain), that has a detection limit of 50 HIV-RNA copies/ml [18].
A significant virological response was considered only when plasma HIV-RNA declined to <500 HIV-1 RNA copies/ml and/or >1 log with respect to baseline.
Virological success was considered using both on treatment (OT) and intent to treat (ITT) analyses, the latter in which missing subjects were considered as
failures.
The CD4 cell count was measured by flow cytometry (Coulter, Madrid, Spain). HIV genotyping was performed by protease gene sequencing using ABI 3100
(Applied Biosystems, Barcelona, Spain). Protease resistance mutations were identified according to the International AIDS Society 2001 update [19]. The
analysis of virological response in each treatment arm was performed taking into account the total number of PI-resistance mutations, both major and
compensatory changes.
Results
A total of 299 subjects were analysed: 121 on SQV, 62 on IDV and 116 on LPV. Although the main demographic features, such as age, gender and risk
behaviour, were similar along the three arms, other baseline characteristics differed, particularly when comparing subjects on SQV-rit and IDV-rit with those receiving Kaletra. Using Kruskal-Wallis, the following four variables were found to differ statistically in at least one of the treatment arms: mean duration of previous PI exposure, previous exposure to IDV, Abacavir (ABC) use and mean CD4 count at baseline. Patients receiving Kaletra had more prior abacavir use (p<0.01; 43% on Kaletra had prior ABC use vs 5% and 4% for patients receiving IDV/r or SQV/r regimens. 66% of patients receiving Kaletra used ABC in their regimen vs 51% in the IDV/r regimen and 30% in the SQV/r regimen (p<0.05). In more detail, prior PI exposure was broader and for a longer period of time in subjects who began Kaletra. They had received IDV, nelfinavir (NFV) and/or SQV in 90, 81 and 73% of cases, respectively. In contrast, subjects on SQV-rit had previously been exposed to IDV and/or NFV in 73 and 66% of instances, respectively. Likewise, patients who began IDV-rit had been exposed to SQV and NFV in 32 and 76% of cases, respectively. Mean duration of prior PI exposure was 40 months for patients getting Kaletra vs 24 months for IDV/r and 32 months for SQV/r (p<0.05). None of the patients who began SQV- or IDV-rit had been exposed to Kaletra previously. On the other hand, subjects on IDV-rit showed a lower mean plasma HIV-RNA at baseline than those who began Kaletra or SQV-rit. Finally, the mean CD4 cell count at baseline was lower in patients who began Kaletra (275 cells) compared with the other two groups (409 cells in IDV/r and 355 cells in SQV/r). The mean number of PI resistance mutations: 4.5 for Kaletra patients, 3.4 for IDV/r, and 3.7 for SQV/r, there was no significant difference. Tenofovir was not available at the time the study was performed. In almost all subjects, dual PI therapy was prescribed with another two drugs (one to two nucleoside analogues and/or one NNRTI).
Clinical efficacy
All study subjects completed 24 weeks of follow-up. In the ITT analysis, patients on Kaletra tended to show greater improvement, although the difference did not reach statistical significance. It should be noted that the greater rate of response seen in the Kaletra arm was not due to a beneficial effect of ABC (mean response rate of 63% with ABC and 57% without ABC), a drug which was widely prescribed with Kaletra and less frequently with the other PIs.
|
|
 |
| |
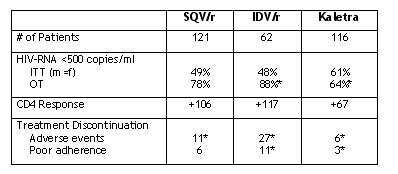
*differences statistically significant (p<0.05)
ITT is intent-to-treat analysis; m=f is missing equals failure; OT is on-treatment analysis
In order to estimate the intrinsic antiviral potency of each dual PI combination, an OT analysis was also performed. In this case, IDV-rit showed the better
performance. Up to 88% of subjects still on IDV-rit at 24 weeks showed virological response. This figure was 78% on SQV-rit (P=n.s.) and 64% on Kaletra
(P<0.05).
The median CD4+ cell increase in patients on rit-boosted combinations at 24 weeks was significantly lower among those on Kaletra (+67 cells/l) than in
subjects receiving either SQV-rit (+106 cells/l) or IDV-rit (+117 cells/l). However, when CD4+ cell variations were assessed, taking into account the mean percentage of variation with respect to baseline, the differences among groups were minimized: +37% on Kaletra, +45% on IDV-rit and +48% on SQV-rit. The difference in mean percent of variation was significanr between SQV/r and Kaletra but not between IDV/r and Kaletra.
Safety
Adverse reactions leading to drug discontinuation and attributable to the dual PI therapy occurred more frequently in subjects on IDV-rit (16/62, 27%) (P<0.05), than in those receiving SQV-rit (13/114, 11%) or Kaletra (7/116, 6%). Kidney stones and gastrointestinal intolerance were the main side effects recorded in subjects who discontinued IDV-rit, whereas nausea/vomiting and diarrhoea were the main side effects in subjects who stopped SQV and Kaletra, respectively.
Poor treatment adherence was recorded in 11% (six of 56) of subjects on IDV-rit, 6% (six of 104) of those on SQV-rit and in 3% (three of 106) of those on
Kaletra, for whom tablet count records were available. Only differences between subjects on IDV-rit and Kaletra achieved statistical significance (P=0.02).
Predictive value of HIV genotyping
Virus amplified material and nucleotide sequence could be obtained from a total of 207 individuals who completed 24 weeks on therapy. By groups, protease sequences were available for 53 subjects who began SQV-rit, 28 on IDV-rit and 62 on Kaletra. On average, the number of baseline PI resistance mutations tended to be greater among subjects who began Kaletra (4.5±3) than among those under SQV-rit (3.7±2.4) or IDV-rit (3.4±1.9).
Overall, subjects experiencing virological failure at 24 weeks with any of the rit-boosted combinations were those carrying a greater number of PI resistance
mutations at baseline. The mean number of PI mutations in subjects experiencing virological response with respect to those who failed was: 2.7±1.8 vs. 6.7±1.7 using SQV-rit (P<0.05); 3.1±1.9 vs. 5.8±1.6 taking IDV-rit (P<0.05); and 3.7±2.7 vs. 7.2±2.7 receiving Kaletra (P<0.05).
An estimation of the best break point in the number of PI resistance mutations to discriminate who will obtain virological response under rit-boosted combinations showed that five or less PI mutations was the most appropriate cut-off. Accordingly, rates of response were 95 vs. 21% with SQV-rit, 90 vs. 23% with IDV-rit and 90 vs. 48% with Kaletra (P<0.001 in all instances). However, as noted, the response to Kaletra tended to be higher in respect to SQV- or IDV-rit in subjects harbouring greater than five PI mutations, although the differences did not reach statistical significance.
An accurate analysis of the genotypic profile found specific mutations at baseline, which could be associated more consistently with dual PI failure. For instance, subjects harbouring I54V, G73S, I84V and/or L90M before beginning SQV-rit had a greater chance of presenting with virological failure at 24 weeks (P<0.05). With respect to IDV-rit, the presence of A71V and/or L90M substitutions markedly reduced the chance of response, although without reaching statistical significance. Finally, in respect to Kaletra, only the presence of the V82A substitution was statistically associated with a greater rate of virological failure at 24 weeks.
Discussion By Authors
We compared the efficacy and safety of three different rit-boosted PI combinations in heavily pre-treated HIV-infected patients. Our results confirm their high potency as part of salvage interventions, irrespective of the rit-boosted active PI in use. The virological superiority of Kaletra in the ITT analysis over SQV- and IDV-rit, although not statistically significant, was of interest considering the more unfavourable pre-treatment characteristics of the group of patients assigned to Kaletra.
Other studies exploring the efficacy of dual PI regimens in multi-treated subjects have recently been published. The use of the previous hard-gel formulation of SQV with rit 100 mg bid lead to a 48% virological response rate at 24 weeks [20], which is slightly higher than that seen in our current study. However, the limited bioavailability of SQV using the hard gel formulation was counterbalanced by the narrower antiretroviral experience of that selected cohort. Zolopa et al. [21] reported a 56% efficacy of SQV-rit at 24 weeks in a group of subjects who had failed NFV. Again, the limited antiretroviral experience of this cohort most likely contributed to the success of that salvage intervention. In another study in which subjects with longer PI experience were included, a 38% success rate was seen using SQV hard gel with rit [22].
With respect to IDV-rit, we previously found a 22% virological success rate at 24 weeks in a similar patient population [23]. However, in that trial the poor results were partially due to the prescription of rit in liquid formulation, which was badly tolerated by a substantial proportion of patients. The higher virological response rate seen in our current study could be partially attributed to the improved tolerance of rit capsules.
Lastly, the performance of Kaletra in the current study is in the range of preliminary reports from different European expanded access programmes. In these trials, the rates of virological response were homogenous, varying from 56% in the German and Spanish trials [24 and 25] to 58% in the British and Italian studies [26 and 27].
Although the gastrointestinal tolerance of IDV-rit improved significantly with the advent of rit capsules, the high incidence of kidney stones seen in our trial was worrying and was substantially increased over the incidence of IDV without rit [28 and 29]. This observation is not new and has already been emphasised by others [30] who have established a direct correlation between IDV peak levels and the risk of nephrotoxicity.
The intrinsic antiviral potency of the three dual PI regimens was comparable. In the case of IDV-rit and Kaletra, it is well established that tiny doses of rit increase trough levels 30-fold although with a wide inter-individual variability. As a result, the Cmin exceeds the EC50 for wild type and low-level resistant viruses in most instances [31 and 32]. With respect to SQV, the boosting effect of rit provides different benefits, mainly increasing the drug absorption [33]. Also, SQV seems to have the longer antiviral activity, with an intracellular half-life approaching 24 h [34].
When PI are used at full doses without rit boosting, the emergence of specific mutations at the protease gene seems critically to affect the sensitivity to each of these compounds [35]. However, when these drugs are boosted by rit, high PI levels diminish any influence of single mutations on drug sensitivity, so that their activity is really compromised only when multiple PI resistance mutations are present. This phenomenon is well established for Kaletra, for which combinations from a set of 11 protease mutations have been involved in causing significant resistance [17]. Taking into account this information, we identified a critical threshold in five PI resistance mutations above which the response to any rit-boosted PI was significantly compromised. The usefulness of this mutation score to predict response has already been proven with Kaletra [17, 36 and 37], but not with other dual PI combinations. It is noteworthy that in all instances, the virological response to each of the different dual PI combinations was greatly affected when more than five PI resistance mutations were present. However, the residual activity of Kaletra on viruses carrying PI mutations over this break point was still significant, most likely reflecting its advantageous pharmacokinetic profile.
In conclusion, rit-boosted PI regimens are suitable for rescue interventions in multi-drug experienced patients. They provide a significant virological response rate, which is greatly dependent of the baseline number of protease mutations. Differences not only in potency, but mainly in tolerance, may also favour the selection of one dual PI combination over others.
References
1. F. Palella, K. Delaney, A. Moorman et al., Declining morbidity and mortality among patients with advanced HIV infection. New Engl. J. Med. 338 (1998),
pp. 853-860.
2. R. Gulick, J. Mellors, D. Havlir et al., Treatment with indinavir, zidovudine and lamivudine in adults with HIV infection and prior antiretroviral therapy. New Engl. J. Med. 337 (1997), pp. 734-739.
3. A. Carr, K. Samaras, S. Burton et al., A syndrome of peripheral lipodystrophy, hyperlipidaemia and insulin resistance in patients receiving HIV protease inhibitors. AIDS 12 (1998), pp. F51-F58.
4. G. Friedman and L. Andrews , Adherence to antiretroviral therapy. AIDS Rev. 3 (2001), pp. 111-120.
5. B. Ledergerber, M. Egger, M. Opravil et al., Clinical progression and virologic failure on HAART in HIV-1 patients: a prospective cohort study. Lancet 354
(1999), pp. 863-868. Abstract | Full Text + Links | PDF (168 K)
6. R. Rodriguez-Rosado, I. Jimenez-Nacher, V. Soriano, P. Anton and Gonzalez-Lahoz , Virological failure and adherence to antiretroviral therapy in
HIV-infected patients. AIDS 12 (1998), pp. 1112-1113.
7. M. Barry, S. Gibbons, D. Back and F. Mulcahy , Protease inhibitors in patients with HIV disease: clinically important pharmacokinetic considerations. Clin.
Pharmacokinet. 32 (1997), pp. 194-209.
8. C. Flexner , HIV protease inhibitors. New Engl. J. Med. 338 (1998), pp. 1281-1292.
9. G. Kumar, A. Rodrigues, A. Buko and J. Denissen , Cytochrome P450-mediated metabolism of the HIV-1 protease inhibitor ritonavir (ABT-538) in human liver microsomes. J. Pharmacol. Exp. Ther. 277 (1996), pp. 423-431.
10. D. Kempf, K. Marsh, G. Kumar et al., Pharmacokinetic enhancement of inhibitors of the HIV protease by coadministration with ritonavir. Antimicrob.
Agents Chemother. 41 (1997), pp. 654-660.
11. R. Kim, M. Fromm, C. Wandel et al., The drug transporter P-glycoprotein limits oral absorption and brain entry of HIV-1 protease inhibitors. J. Clin.
Invest. 101 (1998), pp. 289-294.
12. R. Van Praag, G. Weverling, P. Portegies et al., Enhanced penetration of indinavir in cerebrospinal fluid and semen after addition of low-dose ritonavir.
AIDS 14 (2000), pp. 1187-1194.
13. D. Havlir, N. Hellmann, C. Petropoulos et al., Drug susceptibility in HIV infection after viral rebound in patients receiving indinavir-containing regimens. J. Am. Med. Assoc. 283 (2000), pp. 229-234.
14. D. Descamps, P. Flandre, V. Calvez et al., Mechanisms of virologic failure in previously untreated HIV-infected patients from a trial of
induction-maintenance therapy. J. Am. Med. Assoc. 283 (2000), pp. 205-211.
15. O. Gallego, C. de Mendoza, M.J. Perez-Elias et al., Drug resistance in patients experiencing early virological failure under a triple combination including indinavir. AIDS 15 (2001), pp. 1701-1706.
16. J. Condra, C. Petropoulos, R. Ziermann, W. Schleif, M. Shivaprakash and E. Emini , Drug resistance and predicted virologic response to HIV type 1
protease inhibitor therapy. J. Infect. Dis. 182 (2000), pp. 758-765.
17. D. Kemp, J. Isaacson, M. King et al., Identification of genotypic changes in HIV-protease that correlate with reduced susceptibility to the protease inhibitor lopinavir among viral isolates from PI-experienced patients. J. Virol. 75 (2001), pp. 7462-7469.
18. M. Collins, B. Irvine, D. Tyner et al., A branched DNA signal amplification assay for quantitation of nucleic acid targets below 100 molecules/ml. Nucleic
Acid Res. 25 (1997), pp. 2979-2984.
19. International AIDS Society-USA Panel, Antiretroviral resistance mutations. HIV Clin. Trials 2 (2001), pp. 346-355 Revised April .
20. R. Rodrguez-Rosado, V. Soriano, P. Barreiro, C. Dona and J. Gonzlez-Lahoz , What dual protease inhibitor combination for salvage therapies?. AIDS 13 (1999), pp. 2180-2181.
21. Zolopa A, Keiser P, Tebas P, et al. Predictors of response to ritonavir/saquinavir antiretroviral therapy in patients for whom nelfinavir failed: a multi-centered clinical cohort study. Proceedings of the Thirteenth International AIDS Conference, Durban, 2000 (abstract WePeB4173).
22. P. Chavanet, L. Piroth, M. Grappin et al., Randomized salvage therapy with saquinavir-ritonavir versus saquinavir-nelfinavir for highly protease
inhibitor-experienced HIV-infected patients. HIV Clin. Trials 2 (2001), pp. 408-412.
23. P. Barreiro, V. Soriano, V. Oller and J. Gonzlez-Lahoz , Potent activity but limited tolerance of ritonavir plus indinavir in salvage interventions. J. Acquir.
Immune Defic. Syndr. 24 (2000), p. 488.
24. Rockstroh J, Lohrengel B, Rode R, Reitmayer R. Efficacy and safety results from the ABT-378/R early access program in Germany. Proceedings of the
Eighth European Conference on Clinical Aspects and Treatment of HIV-Infection, Athens, 2001 (abstract P232).
25. Clotet B, Moreno S, Gonzlez-Lahoz J, et al. Efficacy and safety results of lopinavir/r based HAART in expanded access program in Spain through W24.
Proceedings of the Eighth European Conference on Clinical Aspects and Treatment of HIV-Infection, Athens, 2001 (abstract P235).
26. Gilleece Y, Qazi N, Pozniak A, Gazzard B, Nelson M. The efficacy of lopinavir in PI-failing regimes and the safety of switching from a non-failing
antiretroviral regime to lopinavir. Proceedings of the Eighth European Conference on Clinical Aspects and Treatment of HIV-Infection, Athens, 2001 (abstract P60).
27. Germinario A, di Luzio U, Chevallier P. Initial efficacy and safety results from the lopinavir/ritonavir (ABT-378/R) early access program in Italy. Proceedings of the Eighth European Conference on Clinical Aspects and Treatment of HIV-Infection, Athens, 2001 (abstract P233).
28. P. Bonfanti, L. Valsecchi, F. Parazzini et al., Incidence of adverse reactions in HIV patients treated with protease inhibitors: a cohort study. J. Acquir.
Immune Defic. Syndr. 23 (2000), pp. 236-245.
29. P. Barreiro, V. Soriano, N. Cabello and J. Gonzalez-Lahoz , Is there a role for indinavir twice-daily in clinical practice?. Clin. Microbiol. Infect. 6 (2000),
pp. 152-154.
30. Cahn P, Casiro A, Puentes T, et al. Continuing indinavir (800 mg tid) versus switching to indinavir plus ritonavir (800/100 mg bid) in HIV patients having
achieved viral load suppression. A randomized study: the bid efficacy and safety trial. First IAS Conference on HIV Pathogenesis and Treatment. Buenos Aires, 2001 (abstract 60).
31. D. Burger, P. Hugen, R. Aarnoutse et al., A retrospective, cohort-based survey of patients using twice-daily indinavir+ritonavir combinations:
pharmacokinetics, safety, and efficacy. J. Acquir. Immune Defic. Syndr. 26 (2001), pp. 218-224.
32. A. Hsu, G. Granneman, D. Kempf et al., The Ctrough inhibitory quotient predicts virologic response to ABT-378/ritonavir therapy in treatment experienced patients. AIDS 14 Suppl 14 (2000), p. 12.
33. A. Veldkamp, R. van Heeswijk, J. Mulder et al., Steady-state pharmacokinetics of twice-daily dosing of saquinavir plus ritonavir in HIV-1 infected individuals. J. Acquir. Immune Defic. Syndr. 27 (2001), pp. 344-349.
34. M. Nascimbeni, C. Lamotte, G. Peytavin, R. Farinotti and F. Clavel , Kinetics of antiviral activity and intracellular pharmacokinetics of HIV type 1 protease inhibitors in tissue culture. Antimicrob. Agents Chemother. 43 (1999), pp. 2629-2634.
35. R. Shafer, R. Kantor and M. Gonzales , The genetic basis of HIV-1 resistance to reverse transcriptase and protease inhibitors. AIDS Rev. 2 (2000), pp.
211-228.
36. V. Calvez, I. Cohen-Codar, A. Marcelin et al., Analysis of the correlation between baseline genotypic profile and virologic response to lopinavir/ritonavir in 104 multiple PI-experienced patients treated under Expanded Access Program. Antivir. Ther. 6 Suppl 1 (2001), p. 69.
37. B. Masquelier, S. Lawson-Ayayi, D. Neau et al., HIV-1 genotypic determinants of the virologic response to lopinavir/ritonavir-containing therapy in PI-experienced patients. Antivir. Ther. 6 Suppl 1 (2001), p. 84.
|
|
| |
| |
|
|
|