 |
 |
 |
| |
Effect on Glucose Metabolism by Adding Lo-Dose RTV to Reyataz: In Vitro study
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
Reported by Jules Levin
"Maintenance of Favorable In Vitro Metabolic Profile of atazanavir When Combined With Low-Dose Ritonavir"
M.A. Noor and colleagues from Bristol-Myers Squibb reported these study results in an oral presentation at the Intl AIDS Conference in Bangkok.
BRIEF SUMMARY
In this study Noor and BMS conducted an in vitro examination of the effect of atazanavir and lopinavir, and these two drugs boosted with low dose ritonavir (as they are in fact used in patients), on glucose metabolism. Previous studies have shown that unboosted atazanavir (Reyataz) has not in general had a negative impact on patients taking Reyataz-based HAART. Since Reyataz is often used in a regimen boosted with 100 mg RTV once daily, there has been some concern that the benefits seen with unboosted Reyataz may not apply when 100mg of RTV is added. So this study addresses this question with an in vitro examination. Noor concluded from study findings: ATV when used in combination with low dose RTV (<= 3 µM) maintains its favorable profile in this cell model of glucose uptake. The data are consistent with the current hypothesis that RTV boosted ATV used clinically will maintain the favorable metabolic profile of unboosted atazanavir. Clinical data, studies in humans/patients, are needed to confirm these findings. There is speculation that impaired glucose metabolism may play a role in the development of body changes. At the Lipodystrophy Workshop in Paris in Summer 2003, a lipodystrophy substudy of the 034 study comparing efavirenz to atazanavir (unboosted) was presented showing that on average patients receiving atazanavir did not develop body changes. In fact patients receiving efavirenz fared well also. At the Bangkok conference there is a poster from BMS addressing these findings and I will report on that. In this report, immediately below, you'll find summaries of the published studies examining indinavir and lopinavir/r and their affect on glucose/insulin resistance.
Background from Noor.
Treatment of HIV with some protease inhibitor (PI) drugs is associated with metabolic complications including insulin resistance and type 2 diabetes. A proposed mechanism is by blockade of insulin-responsive glucose transporter (GLUT4). Atazanavir (ATV) is a new PI that is not associated with insulin resistance and does not block glucose uptake through GLUT4 in vitro. Clinically, increasing number of patients are being treated with combination of low-dose ritonavir (RTV) (mean Cmax 0.5 to 1.5 µM) added on to ATV to take advantage of enhanced pharmacokinetic profile. We tested the hypothesis whether low dose RTV would negate the favorable metabolic profile of ATV previously reported in vitro.
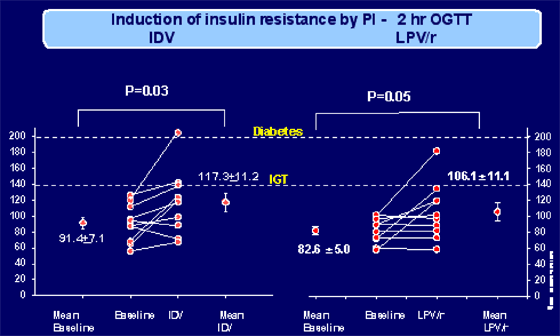
In the picture of the Table 1 below (if you can't see it or additional pictures of tables and graphs below, this article with all pictures will be posted on NATAP website), this is a depiction of the data from studies of indinavir and Kaletra given to individuals to evaluate the induction of insulin resistance by performing 2 hr OGTT. These studies have been previously reported. Healthy volunteers were given one dose of indinavir. (Noor et al, AIDS, 2001 May 4). In the second study several individuals received Kaletra and one individual developed glucose intolerance approaching diabetes. (Lee et al, AIDS March 5 2004). Results of these two studies are summarized immediately below.
In the presence of protease inhibitors, studies have found that the normal update, storage and transport of glucose is inhibited.
RESULTS OF STUDY OF INDINAVIR (Noor et al, AIDS, May 4, 2004), "Metabolic effects of indinavir in healthy HIV-seronegative men". IDV 800 mg thrice daily was given to HIV-negative healthy volunteers to determine the effects of this PI drug in the absence of HIV infection and immune dysregulation. They studied glucose and lipid metabolism before and after 4 weeks of treatment to optimize detection of direct metabolic effects and minimize changes in body composition. Subjects were instructed to take indinavir 800 mg three times daily with two glasses of water. Adherence was monitored. Ten subjects completed the study. Subjects ranged in age from 30 to 65 years.
Fasting glucose, insulin, I : G ratio and insulin resistance index by HOMA all increased significantly with indinavir. Fasting lactate levels did not change. Seven subjects had an increase in HOMA index while three had no change. During OGTT, glucose and insulin levels at 2 h also increased. Of note, based on glucose levels at 2 h on OGTT, one subject developed diabetes (2 h glucose 11.3 mmol/l), one became glucose intolerant (2 h glucose 7.9 mmol/l), and one nearly developed impaired glucose tolerance (2 h glucose 7.7 mmol/l). A trend towards an increase in AUC for glucose was observed. Insulin levels at 2 h were significantly increased.
During hyperinsulinemic euglycemic clamp, a steady-state insulin level of >500 pmol/l was achieved after 30 min. Steady-state glucose levels of >4.5 mmol/l were reached at 60 min and were maintained for 180 min. After 4 weeks of indinavir treatment, insulin-mediated glucose disposal rate per unit of insulin from 60 to 180 min declined in nine out ten subjects by an average of 1.4 mg/kg.
The authors concluded: The metabolic effects of indinavir in HIV-seronegative subjects was studied to distinguish direct drug effects from those caused by the host response to HIV that occurs in the context of immune reconstitution. Following 4 weeks of treatment, there was a small decrease in total fat by DEXA but no increase in abdominal visceral fat and subcutaneous fat or the ratio of visceral to total adipose tissue by computed tomographic scanning. Consequently the effects on intermediary metabolism cannot be attributed to an increase in visceral fat, although they could be part of a more complex syndrome that subsequently would lead to changes in body composition.
Indinavir caused insulin resistance in these HIV-negative subjects. Genetic factors may play a role in susceptibility to the effects of indinavir. Three subjects had first-degree relatives with diabetes. These included the subject who developed diabetes and the subject who developed near impaired glucose tolerance by OGTT criteria. The third subject with a family history of diabetes had the greatest decline in glucose disposal rate (M value) during hyperinsulinemic euglycemic clamp on indinavir.
Peripheral insulin resistance has been reported as a complication of therapy with PI in HIV-infected patients, but the mechanism is not completely understood. In this study there was a 20% decrease in sensitivity of the peripheral tissues to insulin action (i.e. insulin-mediated glucose disposal) during a hyperinsulinemic euglycemic clamp. Insulin-mediated glucose disposal occurs primarily in muscle and adipose tissues. Insulin signaling induces translocation of the glucose transporter vesicles (predominantly GLUT4) to the plasma membrane. In vitro study of the effect of PI on glucose uptake in cultured adipocytes has shown that indinavir at near-peak therapeutic concentration of 10 umol/l (7120 ng/ml) caused inhibition of the insulin-stimulated glucose uptake. This 26% decrease is similar to our finding of 20% decline in insulin-mediated glucose disposal, although findings in cultured cells cannot be directly extrapolated to results in vivo. Moreover, our data do not exclude another independent effect of indinavir on the liver that causes insulin resistance.
RESULTS OF LOPINAVIR/R STUDY. (Lee et al, AIDS, March 5, 2004). A dose of 400 mg lopinavir/100 mg ritonavir was given twice a day to 10 HIV-negative men. Fasting glucose and insulin, lipid and lipoprotein profiles, oral glucose tolerance, insulin sensitivity by euglycemic hyperinsulinemic clamp, and body composition were determined before and after lopinavir/ritonavir treatment for 4 weeks.
Ten male subjects ranged in age from 29 to 66 years; one subject was Hispanic whereas the other nine were Caucasian. After 4 weeks of lopinavir/ritonavir treatment, fasting triglyceride levels increased nearly twofold, whereas VLDL cholesterol levels increased by 33%. Eight out of 10 subjects showed an increase in triglyceride levels. There were no changes noted in fasting LDL, HDL, IDL, lipoprotein (a), total cholesterol, or HDL. Among the VLDL subclasses, VLDL2 levels significantly increased and there was a smaller trend in VLDL3 levels. A small decrease in LDL subspecies cannot be ruled out. FFA increased by 30% after lopinavir/ritonavir treatment. Eight out of 10 subjects showed an increase in FFA levels (Fig. 1). The clearance time of triglyceride levels as measured by an intravenous fat tolerance test showed a trend towards a small 14% delay in fat clearance.
In contrast to what we found with 4-week treatment with indinavir, fasting glucose, insulin, and insulin resistance index by HOMA did not change after 4 weeks of treatment with lopinavir/ritonavir. Likewise, insulin resistance as measured by euglycemic hyperinsulinemic clamp was not significantly altered. During the euglycemic hyperinsulinemic clamp, a steady state insulin level of approximately 360 pmol/l was achieved after 30 min and maintained until 180 min. Steady-state glucose levels of approximately 4.5 mmol/l were achieved at 60 min and were maintained to 180 min. The insulin-mediated glucose disposal rate per unit of insulin (M/I) from 120 to 180 min remained unchanged with lopinavir/ritonavir treatment (15.9 ± 2.1 mg/kg per min per uU/ml at baseline versus 16.2 ± 2.3 at 4 weeks; P = 0.81).
During the OGTT, glucose levels were increased at 2 h after 4 weeks of treatment. Eight out of 10 subjects showed an increase in 2-h glucose levels and one subject developed impaired glucose tolerance (2 h glucose of 10.1 mmol/l). The AUC for glucose showed a trend towards an increase. There was a trend towards a decrease in insulin levels early in the OGTT (30 min), and insulin levels also increased slightly by 2 h. FFA were suppressed to the same level both before and during treatment with lopinavir/ritonavir.
After 4 weeks treatment with lopinavir/ritonavir, there were no significant changes in body composition, including weight, BMI, body cell mass by bioelectric impedence analysis, lean body mass, bone mineral content, or fat mass by DEXA, or visceral or subcutaneous fat by CT.
Author Discussion.
The most notable effect of 4 weeks of lopinavir/ritonavir treatment on HIV seronegative men was the 83% increase in triglyceride levels with the greatest increase seen in VLDL particles. These results are similar to but quantitatively less than the threefold increase in triglyceride levels seen in HIV-negative subjects treated with ritonavir for 2 weeks. Ritonavir is the likely agent causing the increase in triglyceride levels in the lopinavir/ritonavir combination. The effect of lopinavir cannot be studied in isolation, as ritonavir is needed as a pharmocokinetic booster by blocking cytochrome CYP 3A4; therefore, one cannot determine whether lopinavir alone contributes to the elevation in triglyceride levels seen in this study.
In contrast to the effects on VLDL and triglyceride levels, lopinavir/ritonavir did not increase LDL cholesterol levels. Increases in LDL have been observed in HIV-positive patients on HAART with several PI. However, HAART with a non-nucleoside reverse transcriptase inhibitor (NNRTI), such as nevirapine, raises LDL cholesterol to similar levels as indinavir, ritonavir, nelfinavir and saquinavir. In contrast, the treatment of HIV-negative individuals with four different PI: ritonavir, indinavir, amprenavir, and lopinavir/ritonavir (this study), did not lead to an increase in LDL levels. These data suggest that PI therapy does not directly cause alterations in LDL metabolism. Whether these changes in LDL levels seen in HIV-positive patients on PI and NNRTI represent a restoration to health, or an interaction between HAART and HIV remains to be determined. Preliminary data indicate that atazanavir does not increase LDL in HIV-positive patients. Therefore, multiple mechanisms for alterations in LDL levels may be involved.
Although lopinavir/ritonavir had marked effects on triglyceride, FFA and VLDL cholesterol metabolism, less dramatic effects were seen on glucose metabolism and insulin resistance. Unlike indinavir, lopinavir/ritonavir had no effect on fasting glucose, fasting insulin, or HOMA, an assessment of fasting insulin resistance. No difference in insulin sensitivity or the induction of insulin resistance was seen in 10 patients using the euglycemic hyperinsulinemic clamp. However, there was a small, but significant, decrease in glucose tolerance at 120 min of OGTT. A similar impairment in glucose tolerance during OGTT, but with little insulin resistance by the minimal model, was found with amprenavir treatment of HIV-positive patients. We also previously found an increase in insulin and glucose at 120 min with indinavir 4-week treatment of healthy normal volunteers.
There are several potential explanations for the different effects of lopinavir/ritonavir on glucose metabolism compared with amprenavir and indinavir. First, the changes seen during the OGTT may be independent of GLUT4 blockade, and may be caused by impaired first and second-phase insulin secretion. Woerle et al. showed in 13 HIV-infected patients starting various PI including lopinavir/ritonavir and indinavir that first-phase insulin secretion was decreased by 25% during the hyperglycemic clamp. Second-phase insulin secretion did not decrease, but was inappropriately reduced in the setting of peripheral insulin resistance, as reflected in a decreased disposition index. Lopinavir/ritonavir may thus impair both first and second-phase insulin secretion more than peripheral resistance, resulting in impaired glucose tolerance on OGTT, with little evidence of impaired insulin-mediated glucose disposal on euglycemic hyperinsulinemic clamp. The decreased insulin levels at 30 min during the OGTT are consistent with impaired first-phase insulin secretion. An effect on insulin clearance is less likely, as levels during the clamp were not affected. Alternatively, the 10% decrease induced by lopinavir/ritonavir in glucose tolerance by AUC during the OGTT is small, and it is possible that a small change may have been missed on euglycemic hyperinsulinemic clamp or fasting glucose and insulin levels. The finding that 2 h insulin levels on OGTT increased in those with the largest increase in glucose supports this hypothesis. Although insulin levels at 120 min are elevated, second-phase insulin secretion may not be adequately increased in the setting of insulin resistance. A combination of mild insulin resistance coupled with impaired secretion preventing an adequate compensatory increase in insulin is therefore likely. Although we have not performed a randomized trial of lopinavir/ritonavir versus indinavir, it is of note that on lopinavir/ritonavir one patient developed impaired glucose tolerance, whereas on indinavir, one developed diabetes and two developed impaired glucose tolerance. The clinical significance of the small impairment of glucose tolerance on lopinavir/ritonavir is uncertain.
The lack of significant insulin resistance seen with lopinavir/ritonavir treatment during the clamp differs from the current in-vitro data on PI. Indinavir, amprenavir, and ritonavir have been shown to inhibit glucose uptake in 3T3-L1 adipocytes, and indinavir has been shown to inhibit glucose uptake by the acute blockade of GLUT4 transporters in a Xenopus laevis oocyte GLUT4 expression system. Of note, the effect of lopinavir on GLUT4 in vitro remains to be studied. One consideration in comparing the in-vivo effects of indinavir with other PI is the lower protein binding of indinavir. Whereas indinavir is only 60-65% protein bound, ritonavir and lopinavir are over 98% protein bound. The in-vitro studies of PI did not use normal concentrations of serum proteins, and therefore may have had higher free drug levels that do not account for the possible in-vivo effects of protein binding seen with lopinavir and ritonavir. Indinavir may thus achieve higher serum unbound drug concentrations in vivo than other PI.
Another difference between indinavir and lopinavir/ritonavir treatment was their effects on the FFA level. Four-week treatment with indinavir did not increase FFA, whereas lopinavir/ritonavir raised FFA by 30%. Although the stimulation of lipolysis with elevated FFA could be considered a mechanism for the insulin resistance of PI, this is not likely to be the case for several reasons. First, treatment with indinavir induced more insulin resistance than lopinavir/ritonavir by euglycemic hyperinsulinemic clamp, and yet did not increase fasting FFA levels, but tended to decrease them. Second, both indinavir and lopinavir/ritonavir showed FFA suppression during the OGTT. Third, indinavir acutely induced insulin resistance during a euglycemic hyperinsulinemic clamp with normal suppression of FFA. Finally, lopinavir/ritonavir induced a robust increase in fasting FFA without inducing insulin resistance. Although some have speculated that insulin resistance in HIV infection occurs secondary to the increased release of FFA, such studies demonstrate an uncoupling of FFA release and an induction of insulin resistance.
Likewise, it might be postulated that PI-induced insulin resistance might be the cause of the observed hypertriglyceridemia after lopinavir/ritonavir treatment. However, indinavir induced insulin resistance without increasing triglyceride levels, and lopinavir/ritonavir increased triglyceride levels without inducing significant insulin resistance. These data demonstrate that the effects of the drugs on each metabolic pathway are independent. They also emphasize the need to study other PI drugs on multiple metabolic pathways.
There are several potential limitations to the current study. There were no significant changes in body composition after 4 weeks of treatment with lopinavir/ritonavir. There was a trend towards a small decrease (4%) in visceral fat by CT scanning, but no decrease in subcutaneous fat, or total fat by DEXA. We cannot rule out the possibility that this small decrease in visceral adipose tissue may have dampened the induction of insulin resistance, but it might also have dampened the increase in FFA and triglyceride levels. The lack of increase in visceral adipose tissue suggests that changes in lipid and glucose metabolism seen with lopinavir/ritonavir are independent of increased central fat accumulation.
The drug combination of lopinavir/ritonavir limits the ability to identify which of the individual drugs or whether the combination of both are the causative agents of the effects on glucose and lipid metabolism. However, the combination is currently used to treat HIV patients and may be more relevant clinically. Four-week treatment with lopinavir/ritonavir may not have been taken long enough to induce significant changes in body composition. Only men were enrolled in the current study, and pre and postmenopausal women may have different metabolic outcomes.
In summary, 4-week treatment with lopinavir/ritonavir increased plasma triglyceride levels and worsened glucose tolerance during OGTT; however, insulin sensitivity as measured by the euglycemic hyperinsulemic clamp was not impaired. These results contrast with those seen with indinavir, which had little effect on lipid metabolism but significantly increased insulin resistance. The metabolic effects of PI thus appear to be drug specific and not class specific. Individual PI need to be studied with respect to their effects on lipid and glucose metabolism in vivo and in the presence and absence of HIV. As of yet, no PI has been shown to increase LDL significantly in HIV-negative individuals. Further exploration of the mechanism of altered lipid metabolism with PI may lead to a better understanding of lipoprotein production, and long-term studies may offer a better insight into the risk of coronary artery disease in patients with HIV infection.
TABLE 1.
|
|
| |
| |
 |
|
| |
| |
Noor presented a slide showing the results of a BMS study (Parker et al, 2nd IAS Meeting, Paris July 2003, Poster no 708) examining the effect of various protease inhibitors and their effect on glucose uptake in vitro. He made two points, based on these study results: there is a concentration dependent decrease in glucose uptake for all protease inhibitors examined (lopinavir, ritonavir, saquinavir, nelfinavir, indinavir), and "not all protease inhibitors are created equal", protease inhibitors vary in their effect on inhibiting glucose update; he said that lopinavir & ritonavir, at concentrations therapeutically used in patients (5-15 mm) strongly inhibit GLUT 4 mediated glucose uptake, while NFV & SQV has lesser effect; indinavir was in the middle. Atazanavir had significantly lesser effect in inhibiting insulin stimulated glucose uptake.
Noor said they've taken this a step further & validated this. Noor then showed a slide from a study that appears to be about to published (Noor et al, AIDS 2004, in press) using the hyperinsulinemic clamp to examine the effect of ATV, LPV/r and IDV on insulin sensitivity. He compared ATV to LPV/r in healthy volunteers after 5 days of treatment. Noor reported that compared to placebo, the insulin-stimulated glucose disposal rate on ATV was no different. He showed that when giving healthy volunteers LPV/r for 5 days, the insulin-stimulated glucose disposal rate decreased by 24% compared to healthy volunteers (p=0.008). He said this compares with what has been seen in a separate study with IDV given for 4 weeks to healthy volunteers, where the insulin-stimulated glucose disposal rate decreased by an average of 17% (p=0.009), (Noor et al, 2001 May 4). He also included a reference to what happens to diabetics, where non-diabetic healthy subjects were compared to diabetics. On average the insulin-stimulated glucose disposal rate decreased by 38% (p<0.01), (Menelly et al, Diabetes Care, 1998 Aug)
Noor went on to say that since the majority of doctors are prescribing ATZ in combination with low-dose RTV (100 mg), concerns have been raised that RTV might negatively impact the beneficial effects BMS has observed and reported regarding the effect of ATZ on glucose. To answer this question Noor said we hypothesized that ATZ combined with low concentrations (<=3 mM, which is the Cmax observed in patients) of ritonavir would demonstrate an in vitro profile similar to that of ATZ alone in models of glucose uptake.
METHODS: GLUCOSE UPTAKE IN PRIMARY ADIPOCYTES
GLUT activity was assayed as [3H]2-deoxyglucose uptake following insulin stimulation in the mouse or rat primary adipocytes cell models as described previously. Glucose uptake was quantified in the presence of atazanavir in combination with ritonavir at various doses to approximate concentrations expected in patients. PIs and insulin (1 mM) added in glucose-free KRBH buffer with 3% BSA to adipocytes from fasted rat epididymal fat pad or human SAT, add labeled deoxyglucose (30 minutes), reaction stopped with 400 mM Cyto B (35 minutes).
RESULTS
EFFECT OF LOW CONCENTRATIONS OF RTV (<=2 mM) ON GLUCOSE UPTAKE IN RAT PRIMARY ADIPOCYTES
Noor reported that at concentrations up to 3 mM their was no effect of RTV on insulin-stimulated glucose.
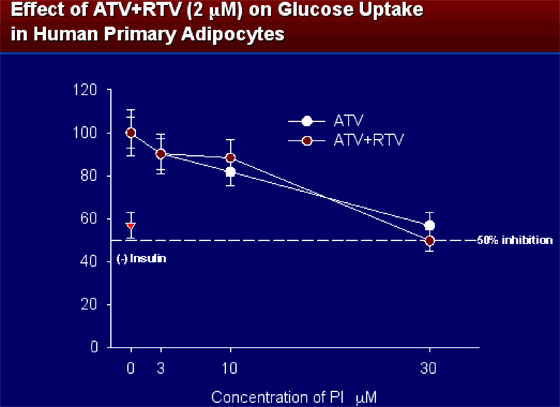
ATV, at therapeutic concentration, alone and in combination with RTV (2 mM) at concentrations observed with pharmacologically enhanced ATV did not inhibit glucose uptake in human primary adipocytes. At higher concentrations (doses) of 3-10 mM (concentrations seen in patients) there appeared to be a slightly greater affect on reducing glucose uptake but similar effect, and when concentration was increased to 30 mM there was only a less than 50% inhibition.
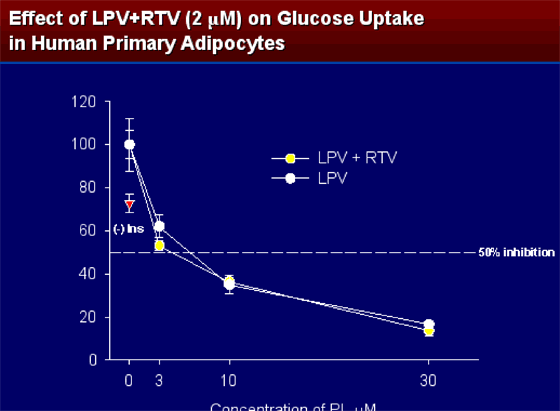
At concentrations of 3 mM or higher lopinavir strongly inhibited insulin-stimulated glucose uptake. When RTV was added (2 mM) to lopinavir no further inhibitory affect was seen.
|
|
| |
| |
 |
|
| |
| |
|
|
| |
| |
 |
|
| |
| |
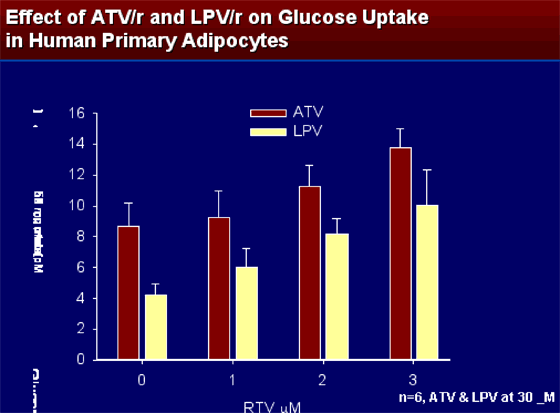
EFFECT OF ADDING RTV TO ATV AND LPV ON GLUCOSE UPTAKE IN HUMAN PRIMARY ADIPOCYTES
At concentrations of RTV of 0, 1, 2, and 3 mM when added to ATZ and LPV, ATZ had higher insulin-stimulated glucose uptake at all of these concentrations than LPV/r. (n=6, ATV & LPV at 30 mM.
|
|
| |
| |
 |
|
| |
| |
Noor concluded:
RTV at concentrations of <=3 mM dose does not affect insulin-stimulated glucose uptake. Inhibitory effect is observed at 10 or more mM.
ATV as a single drug at <=10 mM or combined with low concentration RTV up to 3 mM, which is about the Cmax observed in patients treated with boosted ATV, has very little effect on insulin stimulated glucose uptake, whereas LPV as single drug at <=3 mM or combined with low concentration RTV up to 3 mM affects insulin stimulated glucose uptake in vitro.
Clinical correlation and confirmation studies in patients are needed and planned.
|
| |
|
 |
 |
|
|