 |
 |
 |
| |
Switch to ATV/r Improves Lipids
|
| |
| |
Reported by Jules Levin
"EFFECTS OF SWITCHING TO RITONAVIR-BOOSTED ATAZANAVIR (ATV) ON HIV-INFECTED PATIENTS RECEIVING ANTIRETROVIRAL THERAPY WITH HYPERLIPIDEMIA"
(BMS EARLY ACCESS PROGRAM (EAP), SPANISH NESTED STUDIES)
poster #184, 12th CROI. Feb 22-25, 2005. Boston, MA
E Martinez1 , CJ Azuaje2 , A Antela3 , A Rivero4 , F Lozano5 , E Deig6 , D Fuster7 , O Serrano8
1-H Clinic, Barcelona; 2-H Vall D'Hebron, Barcelona; 3-H Ramon y Cajal, Madrid; 4-H Reina Sofa, Cordoba; 5-H de Valme, Sevilla; 6-H Granollers, Barcelona;
7-H Germans Trias i Pujol, Barcelona; 8-Bristol-Myers Squibb, Spain
The BMS ATV EAP is a multinational prospective trial for HIV treatmentexperienced patients who switch to ATV-based therapy due to treatment failure (not only immunovirological failure but also for toxicity, adherence or hyperlipidemia issues).
Spain enrolled 1621 patients from November 2002 to September 2004.
Nested subanalysis were proposed and carried out in Spanish centers
being 880 patients evaluated. For this specific analysis, only patients
recieving boosted ATV and showing hyperlipidemia at baseline defined
by at least one of the following criteria were selected:
--fasting triglycerides (TG) >500 mg/dL,
--total cholesterol (TC) >200 mg/dL,
--or LDL-cholesterol (LDL) >130 mg/dL
Clinical and fasting laboratory data were collected at baseline, 1, 3, 6,
9 and 12 months as long as the patient remained on therapy.
Introduction
Hyperlipidemia may be relatively prevalent among treated HIV-infected
patients, particularly with the use of protease inhibitors. Hyperlipidemia
is also a leading factor responsible of the increased risk for coronary
heart disease associated with the exposure to antiretroviral therapy.
Switching from nelfinavir to unboosted atazanavir in patients without
prior virological failure to protease inhibitors has been associated with
an improvement of lipid parameters without an increased risk for
clinically significant adverse effects associated with atazanavir and
for virological failure.
Atazanavir boosted with ritonavir (ATV/RTV) has shown a favorable
lipid profile and a durable virological response compared to twice daily
lopinavir/ritonavir (LPV/RTV) in patients with prior virological
failure.
This substudy was designed to assess the impact on lipid parameters
of switching to an antiretroviral regimen containing boosted ATV in
HIV-infected patients with dyslipidemia.
BRIEF SUMMARY OF RESULTS:
In the Spanish ATV/r EAP study, patients had at least one of the following: >500 fasting triglycerides, >200 total cholesterol, or LDL-cholesterol >130. After 12 months the average improvements were:
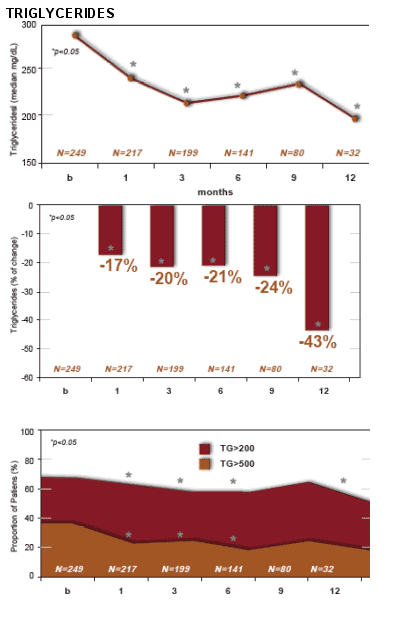
--triglycerides: -43%,
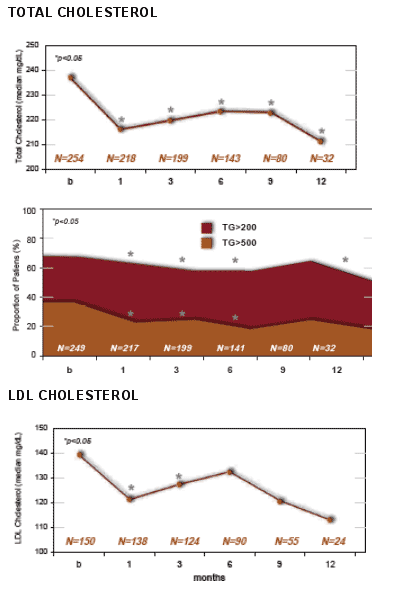
--total cholesterol: -22%,
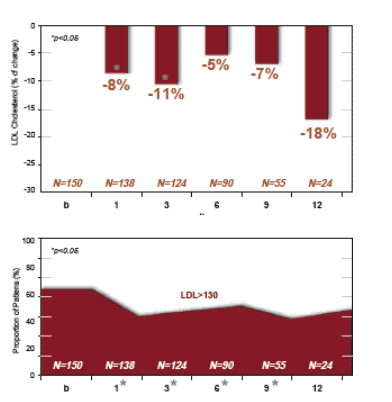
--LDL-chol: -18%
Half of patients were below 500 cp/mL when switched to ATV/RTV containing regimen. This percentage increased to 74% at 6 months (n=141) and to 75% at one year (n=32). Of 135 (53.4%) patients with HIV RNA <500 cp/mL at baseline, 96% at 6 months and 88% at 12 months remained below 500 cp/mL. Of 118 (46.3%) patients with HIV RNA 3500 cp/mL at baseline, 55% at 6 months and 60% at 12 months achieved undetectability below 500 cp/mL. CD4 cell counts (per mL) improved during follow up with increments of 45 cells at 6 months (n=142) and 48 at 12 months (n=32).
STUDY RESULTS
Jaundice was diagnosed in 14 (5.5%) patients but only 3 (1.2%) discontinued therapy. ALT values over 200 IU/L were rarely observed during follow-up and only one patient discontinued due to elevated transaminases, despite half patients were HIV-hepatitis B/C coinfected. Therapy was generally well tolerated. Only 8 (3.1%) of patients discontinued due to ATV-related adverse events.
Usage of lipid lowering therapy decreased from about 33% at baseline to 24% at month 12.
TG declined by 43% from baseline to month 12, from about 290 to 200. The percent with TG >500 appeared from the graph below to decrease from 39% to 20%; the percent with >200 TG decreased as well. Total cholesterol decreased by 22% from baseline to month 12, from about 240 to 210. LDL chol decreased from about 140 to 115 from baseline to month 12, about 18%. The percent of patients with LDL >130 decreased from about 65% at baseline to 43% at month 12. Non HDL chol decreased from about 182 at baseline to 170 at month 12, 24%. The percent with non HDL >160 decreased from about 80% at baseline to 50% at month 12. HDL chol decreased by 6% (NS) from baseline to month 12.
| |
| |
| |
 |
|
| |
| |
 |
|
| |
| |
 |
|
| |
| |
Baseline Characteristics of EAP Patients
Age: 41
77% men
CDC stage: 40% category C
HBV/HCV coinfection: 47%
HIV RNA medican: 4.4 log
HIV RNA <500 cp/ml: 53%
CD4 count median: 38%
65% with lipodystrophy
Cardiovascular Risk Factors
Forty percent of patients had multiple (>2) major risk factors or coronary heart disease (CHD) or equivalent. These numbers could be infraestimated since information about family history of early CV events was not collected.
Smokers: 43%
HTA (hypertension?): 5.3%
Age >45 males, >55 females: 30%
HDL <40: 42%
Diabetes: 8%
Previous cardiovascular event: 3.6%
0-1 risk factors: 59%
>2 risk factors: 32%
previous CV event/DM: 8.6%
TREATMENT HISTORY
790% previously used PIs; preceding therapy: 43% LPV/r, 8% NFV, 8% APV or APV/r, 4% IDV or IDV/r
Fasting TG >500 mg/dL: 29%
Total chol >200 mg/dL: 93%
LDL-chol >130 mg/dL: 64
REASONS FOR DISCONTINUATION
Adverse event: 3%
Jaundice/scleral icterus: 1.2%
Death: 1.2%
Viralogic failure: 1.6%
Other reasons: 5.5%
Total discontinuations: 11.4%
Most patients came from a previous PI regimen (mainly LPV/RTV) and drugs more frequently coadministered with ATV/RTV were TDF, DDI and 3TC.
The drugs most frequently administered with ritonavir boosted ATV were tenofovir (67%), didanosine (48%) and lamivudine (35%), abacavir (17%), d4T (15%) EFV (11%). Only 7 patients (4.3%) discontinued ritonavir-boosted ATV during the follow-up, one (0,6%) due to jaundice. At month six, 58% had <500 copies HIV-1 RNA/mL.
Out of 593 patients included receiving ATV/RTV, 255 met entry criteria and were prospectively followed for a cummulative followup of 1383 patient-months. Fifty six percent of patients completed at least 6 months of treatment.
| |
| |
| |
| |
|
 |
 |
|
|