Reported by Jules Levin
"Increasing Serologic, Virologic and Biochemical Response Over Time to Adefovir (ADV) 10 mg in HBeAg+ Chronic Hepatitis (CHB) Patients"
P Marcellin1, TT Chang2, SG Lim3, W Sievert4, M Tong5, S Arterburn6, S Xiong6, CL Brosgart6, and G Currie6 for the 437 Study Group1Hopital Beaujon, Clichy, France; 2National Cheng Kung University Hospital, Tainan, Taiwan,
Republic of China; 3National University Hospital, Singapore; 4Monash Medical Centre, Victoria, Australia;
5Huntington Medical Center Research Institute, Huntington, California, USA;6Gilead Sciences, Inc., Foster City, California, USA
Marcellin reported this data and information at the EASL meeting, April 13-17, Paris, 2005.
Investigator summary:
The proportion of patients with clinical benefit increased over time:
--Undetectable HBV DNA increased (48% week 144)
--ALT normalization increased (80% week 144)
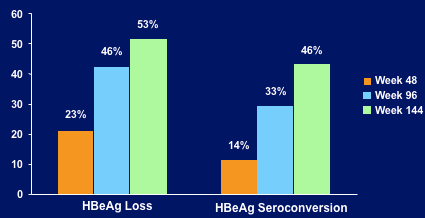
--HBeAg loss and seroconversion increased (53% & 46% week 144)
Safety profile over 144 weeks consistent with first 48 weeks:
--safety profile similar to placebo at 48 weeks
Resistance to adefovir is delayed and infrequent in this study population.
INTRODUCTION
Nucleotide analogue of adenosine monophosphate
Activity against wild-type and lamivudine-resistant (LAM-R) HBV1-5
10 mg once daily orally
Durable HBV DNA suppression with a high threshold for the development of resistance
Safety profile similar to placebo in 48 week controlled trials
KEY INCLUSION CRITERIA
HBeAg+ , anti-HBe- and HBsAg+
Compensated liver disease
HBV DNA ³
106 copies/mL
ALT x ULN ³
1.2 —10
Adequate renal function
STUDY DESIGN*
*misallocation: patients received significant periods of intermittent dosing in Yr 2.

HBV DNA UNDETECTABLE & ALT NORMALIZED
Kaplan-Meier Estimates

HBeAg LOSS & SEROCONVERSION OVER TIME
Kaplan-Meier Estimates
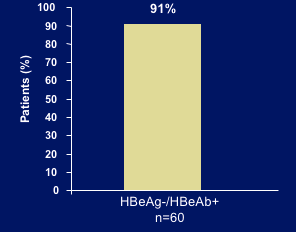
DURABILITY of HBeAg SEROCONVERSION OFF THERAPY
Median duration off ADV therapy: 55 weeks (range 5-114)
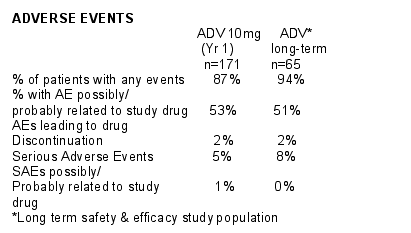
No patient who discontinued treatment with adefovir had a serum creatinine elevation >0.5 mg/dL above baseline or a serum phosphorous <2.0 mg/dL.
No patient had an ALT elevation accompanied by signs of worsening liver function.
RESISTANCE EVALUATION
All samples with detectable serum HBV DNA by PCR (>1000 copies/ml) at weeks 48 & 144* were analyzed.
--Incidence of ADV resistance at week 144* (3%, 2/65)
