| |
Fat Distribution in Men With HIV Infection
|
| |
| |
JAIDS Journal of Acquired Immune Deficiency Syndromes: Volume 40(2) 1 October 2005 pp 121-131
From the Study of Fat Redistribution and Metabolic Change in HIV Infection (FRAM)
Received for publication May 17, 2005; accepted August 4, 2005.
Supported by National Institutes of Health grants RO1-DK57508, HL74814, HL 53359, and AI 027767 and NIH GCRC grants M01-RR00036, RR00051, RR00052, RR00054, RR0636, and RR00865. The funding agency reviewed the study design but played no role in collection, management, analysis, or interpretation of the data or in preparation of the manuscript. NIH personnel participated as members of the CARDIA Publications Committee that reviewed and approved the manuscript.
Reported by Jules Levin
In response to the publication of the FRAM Study I've received emails from patients expressing concern. They do not feel that the study results capture the experiences of patients developing belly fat accumulation. Perhaps the point is correct, that FRAM's study design was not able to evaluate the experiences of many patients who experienced fat accumulation with or without lipoatrophy..
The study authors do however say (see below) that some study patients in FRAM self-reported that over time they experienced increased belly fat. At the same time the study authors found the overall prevalence of self-reported increased visceral or belly fat was similar between the Controls & HIV+ study participants in FRAM.
One of the study limitations is that it's a cross-sectional study, which means they take a one-time look at the participants. They do not follow people over time. FRAM did not follow patients from when they started HAART in 1995 when many patients first developed belly fat & lipoatrophy simultaneously. FRAM's cross-sectional look I believe lumps into one study many different situations including patients who may have been on HAART for years & patients who may have just started HAART. Here is what the author says about study limitations.
"....There are several limitations to our data. We report only on men here. This is a cross-sectional study, so further changes in adipose tissue distribution may occur over time. However, the study was done approximately 5 years after the introduction of highly active ARV therapy, which is more than adequate time to see the syndromes studied by others. It is not possible to determine what regional body composition would have been in the HIV-infected group in the absence of HIV infection or the syndrome of lipoatrophy...."
If the FRAM study design was unable to capture the full lipodystrophy experience of many patients, can the study correctly conclude as the authors do "There is no association of peripheral lipoatrophy with central lipohypertrophy or increased VAT." The study authors identify perhaps the main concern regarding lipodystrophy, that fat loss, also called lipoatrophy, is a hallmark problem. The authors say: "Peripheral lipoatrophy was more frequent in HIV-positive men than in controls (38.3% vs. 4.6%, P < 0.001) The also conclude: "we find evidence for a syndrome of subcutaneous lipoatrophy in HIV-infected men, with lower body depots affected more than upper body depots". The authors also report an interesting finding that even if patients do not cross the threshold of clinical lipoatrophy, many patients seem to be experiencing a generalized fat loss: "Compared with controls, the similar pattern of decreased fat seen in HIV-infected men both with and without the clinical syndrome of lipoatrophy suggests that the clinical syndrome of lipoatrophy represents only the most severely affected group of the spectrum".
I've taken extracts from the study report to highlight them & they were specifically chosen to respond to questions I've received regarding the study.
Factors Associated With Leg SAT (subcutaneous adipose tissue, or fat just under the surface of the skin) and VAT (visceral adipose tissue, or fat deep inside the belly)
Finally, we assessed whether similar factors were associated with leg SAT (the most affected subcutaneous depot) and VAT (an important central depot that is associated with metabolic changes) in HIV-infected men by a multivariate model that included CD4 cell count, HIV RNA level, and ARV therapy duration, as well as the non-HIV-related predictors used above. The effects of race, smoking, and physical activity all followed known patterns in the general population.43 Of note, age was associated with less leg SAT, but more VAT, in HIV-infected subjects. Higher current HIV RNA levels were associated with more leg SAT but did not appear to be associated with VAT. Duration of use of stavudine was strongly associated with less leg SAT, as was that of indinavir. There was a trend for an association of duration of use of didanosine with less leg fat. However, stavudine and indinavir were not associated with more VAT. No ARV was estimated to have a substantial positive association with VAT; the largest point estimate for an ARV was much smaller in magnitude than those for race, age, or physical activity. In contrast, duration of nevirapine use was associated with less VAT. There were no apparent associations with any ARV class or HAART after adjusting for stavudine and indinavir for leg SAT or nevirapine for VAT.
Here is what the study publication says about the study participants:
Subjects
The protocol was approved by institutional review boards at all sites. HIV-infected subjects were selected from coded lists of patients seen in 16 HIV or infectious disease clinics or cohorts during 1999. Each list was randomly ordered and grouped into blocks of 5 potential subjects. Sites were given sets of blocks; only after extensive recruitment efforts by multiple methods were completed for a block was a new block provided. Analyses of female participants are incomplete and will be reported subsequently. Of 2094 male patients active in the HIV clinics, 414 did not answer multiple contact attempts; 154 were dead; 269 were ineligible (institutionalized, n = 23; communication difficulty, n = 37; primary care provider refused, n = 13; or excluded by study criteria, n = 196 [age <18 years; factors prevented imaging: weight >136 kg, height >6'5", metal in body, claustrophobia]). The remaining 1257 eligible men were approached for informed consent. Of those, 279 declined participation and 163 did not return for data collection. Examinations were completed from June 2000-January 2002 on 825 men (65.6% of 1257 contacted and eligible).
Control subjects were recruited from 2 centers of the Coronary Artery Risk Development in Young Adults (CARDIA) study.39,40 CARDIA subjects were originally recruited as a population-based sample of healthy 18- to 33-year-old white and African American men and women from 4 cities in 1986 for a longitudinal study of cardiovascular risk factors. Participants in the CARDIA study were stratified for the 2 races and genders. Of 377 participants who agreed to the year 15 CARDIA examination who were approached to participate in FRAM, 339 (90%) agreed to participate; 28 (7%) did not return for examination; and 14 (4%) were excluded from FRAM for factors preventing imaging. HIV testing was not done in the CARDIA cohort, but the prevalence of HIV infection was estimated to be <1% based on previous data, and no CARDIA participant was taking an ARV medication.
Here we report findings for the 425 HIV-infected men and 152 control men in the 33- to 45-year age range; we excluded 11 HIV-infected men who had an opportunistic infection or malignancy within the same or previous calendar month as the examination, to remove confounders of acute changes in fat.
TABLE 1. Demographics of Men Between the Ages of 33 and 45
Lipoatrophy and Lipohypertrophy by Self-Report, Examination, and Concordance
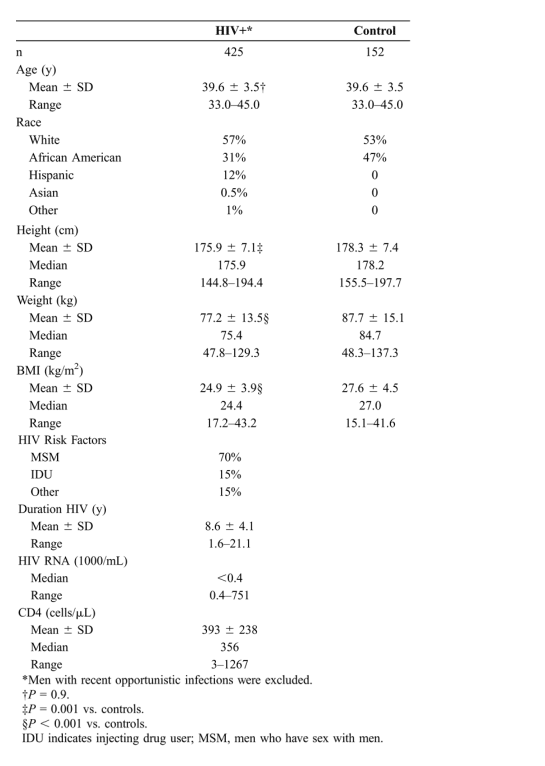
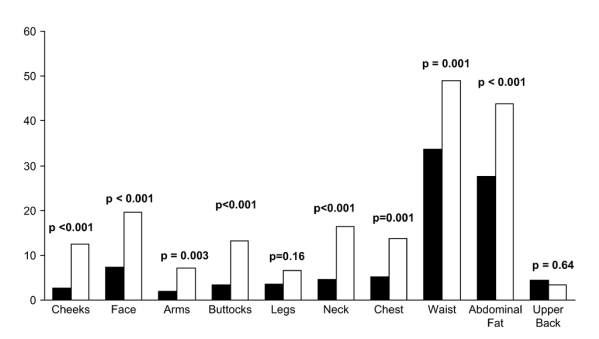
Figure 1A shows that more HIV-infected men than control subjects reported fat losses in the peripheral sites: cheeks, face, arms, buttocks, and legs (all P < 0.001). Half of fat loss changes reported in HIV-infected men were mild. Likewise, for central sites, higher proportions of HIV-infected men than control subjects reported decreases in waist size and chest fat and possibly neck and upper back fat. These results indicate that a significantly greater proportion of HIV-infected men lost fat in peripheral and central sites compared with control men.
Some HIV-infected men reported gain in fat (Fig. 1B). However, the prevalence of fat gain was significantly lower in HIV-infected men than controls in most central and peripheral sites.
Findings on examination were similar to those from self-report (data not shown). Using the commonly accepted standard of self-report confirmed by physical examination, clinical lipoatrophy was more prevalent in all peripheral sites in HIV-infected men than in controls (Fig. 2A). HIV-infected men were also more likely to have lipoatrophy in the central site of abdominal fat and possibly neck, but the prevalence of central lipoatrophy was much lower than in the periphery. With regard to clinical lipohypertrophy defined as concordance of self-report and examination, HIV-infected men had less lipohypertrophy in the neck, chest, abdomen, waist, cheeks, face, arms, and buttocks compared with controls (Fig. 2B).
FIGURE 1. Prevalence of self-report of change in body fat over the prior 5 years. HIV-infected men (solid bars) and control subjects (open bars) were surveyed for self-reported changes in fat in multiple anatomic areas and in waist size that occurred in the 5 years prior to study. A, Decreased fat. B, Increased fat.
% Reporting fat Loss (figure A)
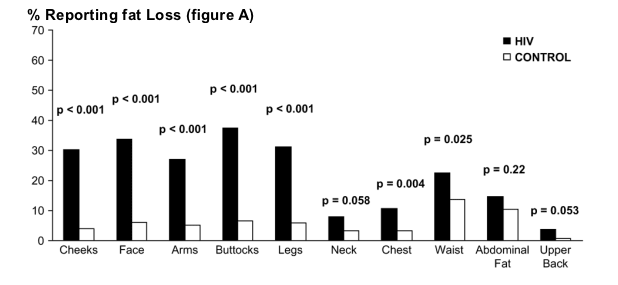
% Reporting fat Gain (figure B)
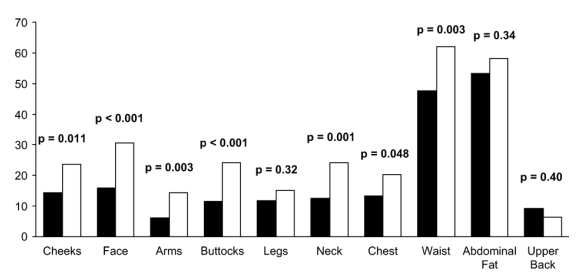
FIGURE 2. A, Prevalence of lipoatrophy by concordance. Subjects who reported loss of fat and had less fat than normal on examination were designated as having clinical lipoatrophy. B, Prevalence of lipohypertrophy by concordance. Subjects who reported gain of fat and had more fat than normal on examination were designated as having clinical lipohypertrophy. HIV-infected (solid bars); control subjects (open bars).
% with Clinical Lipoatrophy (Figure A)
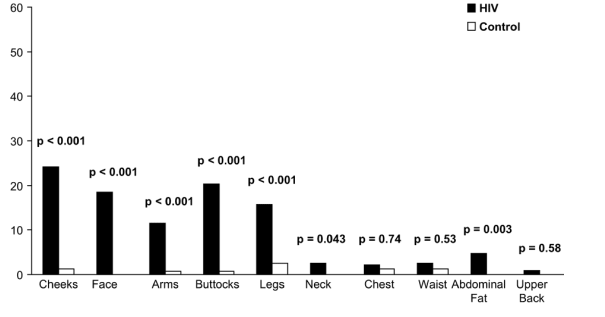
% with Clinical Lipohypertrophy (Figure B)
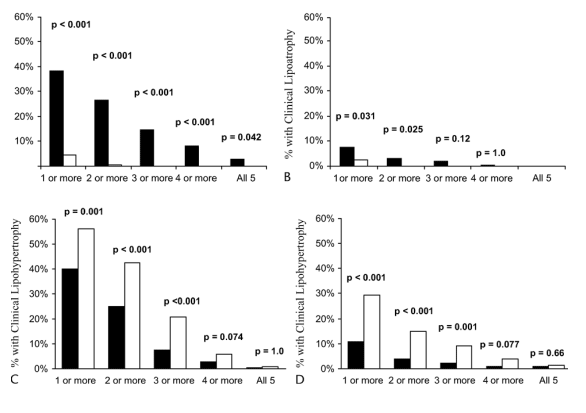
We then separately assessed the prevalence of lipoatrophy and lipohypertrophy at any peripheral or central site (Fig. 3). HIV-infected men were more likely to have peripheral lipoatrophy in at least one site compared with controls (38% vs. 5%; P < 0.001, Fig. 3A). HIV-infected men commonly had multiple sites of peripheral lipoatrophy, whereas this was uncommon in controls. More HIV-infected men than controls had clinical central lipoatrophy (8% vs. 3%, P = 0.03, Fig. 3B), although this finding was less common than peripheral lipoatrophy. In contrast, controls were more likely to have lipohypertrophy at one or more sites, be they central or peripheral (Figs. 3C and D).
FIGURE 3. Prevalence of lipoatrophy (A and B) and lipohypertrophy (C and D) by number of anatomic sites affected in HIV-infected (closed bars) and control subjects (open bars). The percentage of subjects with each number of anatomic sites affected is presented. A and D, Peripheral sites (cheeks, face, arms, buttocks, leg). B and C, Central Sites (neck, chest, upper back, waist, abdominal fat).
% with Clinical Lipoatrophy (A)
MRI Measurement of Regional Adipose Tissue Volumes
Because peripheral lipoatrophy was the most common characteristic that distinguished HIV-infected men from control subjects and because peripheral lipoatrophy has been reported as a distinct clinical syndrome, we compared adipose tissue volume measured by MRI in HIV-infected men with and without clinical peripheral lipoatrophy and controls. Compared with HIV-infected men without peripheral lipoatrophy, HIV-infected men with clinical peripheral lipoatrophy had less SAT in peripheral depots of legs and arms, but they also had less SAT in central depots of the lower trunk/abdomen and upper trunk/chest, and possibly VAT. The findings were similar when adipose tissue volume was adjusted per lean body mass rather than height (data not shown).
|
|
| |
| |
|
|
|