 |
 |
 |
| |
Atazanavir Use in Pregnancy
|
| |
| |
Reported by Jules Levin
3rd IAS Conference
July 24-27, 2005
Rio de Janeiro
Poster # TuPe5.2P01
Morris A.1; Juethner S.2; Theroux E.3
1Community Research Initiative of New England, Springfield, MA; 2Bristol Myers Squibb;
3Baystate Medical Center Children's Hospital, Springfield, MA
Background
In the United States, most HAART regimens used during pregnancy include a protease inhibitor in order to meet two goals:
1. Optimal treatment for the health of the woman and
2. To prevent perinatal HIV transmission
Data are available in the published literature on nelfinavir and several other PIs, however, no data exist on some of the newer agents.
Atazanavir has a pregnancy category B label. While unconjugated hyperbilirubinemia is commonly noted with atazanavir use in non-pregnant adults, it is not known if the administration of this drug during pregnancy is safe in
mothers or will exacerbate physiological hyperbilirubinemia in neonates. At this time, there are no published studies evaluating atazanavir in pregnant women. Prospective trials employing various HAART regimens during pregnancy are
underway, however, additional observational data are needed to inform the field until these trials are complete.
Methods
--This is a retrospective, observational study of all pregnant women treated with atazanavir in one clinical practice through June 2005.
--Institutional review board approval was obtained.
--Data were abstracted from charts on maternal CD4 cells, viral loads, bilirubins, and jaundice.
--Infant weights, heights, the presence or absence of hyperbilirubinemia, and infant HIV infection status were recorded.
--Adverse effects were also recorded in mothers and infants.
Results
--To date nine women have been treated with atazanavir during pregnancy. Six of nine were on boosted atazanavir.
--ZDV/3TC was used in six women; tenofovir in the other three.
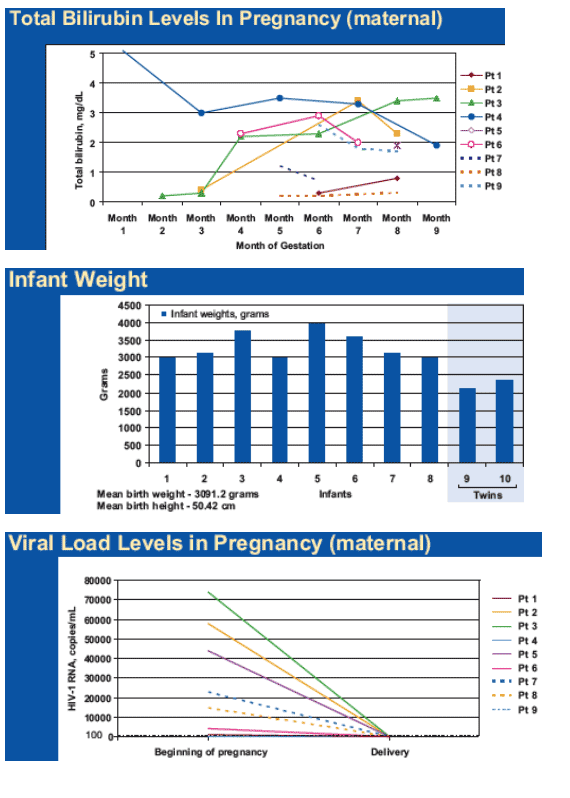
--At the onset of pregnancy, the mean viral load of women not on ART (n=7) was 31,133 copies/mL. The two women on ART at the time of conception had undetectable viral loads < 50 copies/mL.
--At delivery, all had HIV-1 RNA levels <100 copies/mL, with 7/9 having <50 copies/mL.
--The mean CD4 count increased from 366 to 450 cells/mm3 during the course of pregnancy.
--Maternal total bilirubin at time of delivery ranged from 0.3-3.5 mg/dL.
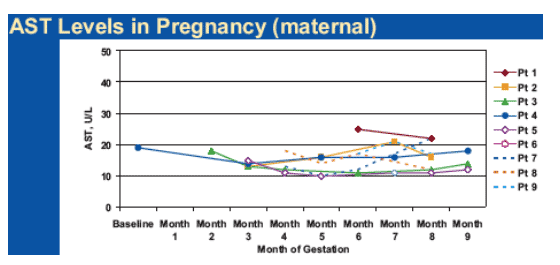
--No hepatic, renal, or pancreatic abnormalities were seen in the mothers during pregnancy.
--The regimens were well tolerated in all women.
--All pregnancies were full term. There were 8 singleton and one twin pregnancies for a total of 10 infants.
--The highest infant bilirubin was 11.5 mg/dL; three infants were treated briefly with phototherapy.
--Mean birth weight and height were 3091.2 grams and 50.17 cm, respectively.
The PCRs on 8 infants are negative at ≥ 4 months. The initial PCRs of the twins at one week of age are negative.
Discussion
The immunologic and virologic response to atazanavir based regimens during pregnancy in this cohort has been excellent and is comparable to reports of other PI based regimens in pregnancy. Women tolerated the medications well and no
adverse clinical or laboratory events were noted.
In the first eight pregnancies, no transmission has occurred. The twins delivered to the ninth woman are also HIV negative thus far, however, they are too young for these results to be definitive.
Neonatal hyperbilirubinemia, defined as total serum hyperbilirubin of 5 or more mg/dL, occurs frequently in the general population with up to 60% of infants experiencing clinical jaundice. This jaundice typically results from deposition of
unconjugated bilirubin in the skin & mucus membranes.
Common causes include fetal-maternal blood group incompatibility, prematurity, previously affected sibling, bruising / trauma from instrumented delivery, breastfeeding, and medications, including oxytocin (Pitocin). The incidence of hyperbilirubinemia in this cohort is within the range of reported rates in the general population.
Phototherapy is typically instituted when an infant's total serum bilirubin is 15 or more mg/dL. In a healthy full-term infant without hemolysis, the provider should be concerned for potential neurologic consequences (kernicterus) when the
total serum bilirubin is >25 mg/dL. The highest total serum bilirubin in this cohort of infants was 11.5 mg/dL.
Conclusion
Atazanavir based regimens in this cohort of pregnant women were very well tolerated in both the mothers and infants.
To date all have had good responses immunologically and virologically and no infant has been infected.
Infants' serology will continue to be followed 1 year post delivery to confirm HIV status.
Until prospective data are available, this series helps to inform the field.

References
Antiretroviral Pregnancy Registry, Pharmaresearch Corporation, Wilmington, NC, 1997.
Atazanavir, Package Insert. Bristol Myers Squibb. July, 2004. 1168954A2.
Connor EM, Sperling RS, Gelber R, et al. Reduction of maternal-infant transmission of human immunodeficiency virus type 1 with zidovudine treatment. N Engl J Med. 1994;331:1173-1180.
Cooper ER, Charurat M, Mofenson L et al, Combination antiretroviral strategies for the treatment of pregnant HIV-1 infected women and prevention of perinatal HIV-1 transmission. J Acquir Immune Defic Syndr. 2002;29:484-94.
Morris AB, Cu-Uvin S, Harwell JI, Garb J, Zorrilla C, Vajaranant M, Dobles AR, Jones TB, Carlan S, Allen DY. Multicenter review of protease inhibitors in 89 pregnancies. J Acquir Immune Defic Syndr. 2000;25:306-311.
Morris AB, Dobles AR, Cu-Uvin S, Zorrilla C, Anderson J, Harwell JI, Keller J, Garb J. Protease inhibitor use in 233 pregnancies. J Acquir Immune Defic Syndr. 2005; in press.
Porter ML, Dennis BL. Hyperbilirubinemia in the term newborn. Am Fam Physician. 2002;65:599-606.
Sarici SU, Serdar MA, Korkmaz A et al. Incidence, course, and prediction of
hyperbilirubinemia in near-term and term newborns. Pediatrics. 2004; 113:775-80.
Tuomala RE, Shapiro DE, Mofensen LM, et al. Antiretroviral therapy during pregnancy
and the risk of an adverse outcome. N Engl J Med. Vol. 346, N 24, June 13, 2002. 1863-1870.
Watts DH. Management of human immunodeficiency virus in pregnancy. N Engl J Med. Vol. 346, N 24, June 13, 2002. 1879-1891.
|
|
| |
|
 |
 |
|
|