 |
 |
 |
| |
Entecavir (ETV) Demonstrates Consistent Responses Throughout Baseline Disease and Demographic Subgroups for the Treatment of Lamivudine-Refractory (LVD-ref) HBeAg(+) Patients with Chronic Hepatitis B
|
| |
| |
Reported by Jules Levin
EASL, April 26-30, 2006, Vienna, Austria
C Yurdaydin1, H Senturk2, A Boron-Kaczmarska3, M Raptopoulou-Gigi4, Y Batur5, Z Goodman6, J Vaughan7, H Brett-Smith7, R Hindes7
1Department of Gastroenterology and the Hepatology Institute, University of Ankara Medical School, Ankara, Turkey; 2Department of Internal Medicine, Istanbul University Cerrahpasa Medical School, Istanbul, Turkey; 3Department of Infectious Diseases, Medical University, Szczecin, Poland;
4Fourth Medical Department, Aristotle University of Thessaloniki, Greece; 5Ege University, Faculty of Medicine, Gastroenterology Department, Izmir, Turkey; 6Department of Hepatic and Gastrointestinal Pathology, Armed Forces Institute of Pathology, Washington, DC, USA;
7Bristol-Myers Squibb Company, Wallingford, CT, USA
Introduction
- Entecavir (ETV) is a potent and selective inhibitor of hepatitis B virus (HBV) polymerase; it inhibits priming, reverse transcription and DNA synthesis1,2
- In this Phase III trial (ETV-026) LVD-ref patients switched to ETV demonstrated superior virologic, histologic and biochemical benefit compared with continued lamivudine (LVD) treatment in HBeAg(+), LVD-ref chronic hepatitis B (CHB) patients after 48 weeks of therapy
- ETV was well tolerated and the safety profile was comparable to that of LVD3,4
- In concurrent trials of HBeAg(+) and HBeAg(-) nucleosidenaïve CHB patients, ETV responses were consistent across baseline disease and demographic subgroups
Objective
- The objective of this subgroup analysis was to assess the influence of demographic subgroups and baseline disease characteristics on efficacy outcomes demonstrated in HBeAg(+) LVD-ref patients
Methods
Phase III Study Design: Study-026
- Randomized, double-blind study in lamivudine-refractory HBeAg(+) patients with chronic hepatitis B (CHB)
- ETV 1.0 mg once daily (QD; n=141) versus LVD 100 mg QD (n=145) for at least 52 weeks of treatment
- LVD-ref was defined as evidence of viremia while on LVD or the presence of LVD resistance mutations
- Primary endpoint:
- proportion of patients at week 48 with histologic improvement (≥2-point decrease in Knodell necroinflammatory score and no worsening of fibrosis [≥1-point increase from baseline in Knodell fibrosis score])
Subgroup Analysis
- The subgroup analyses assessed the relationship between efficacy endpoints and baseline disease, and demographic subgroups in lamivudine-ref CHB patients:
- demographic subgroups:
- geography
- race
- gender
- disease subgroups:
- HBV genotype
- baseline ALT level
- baseline HBV DNA level
- The predictive value of these subgroups was investigated by examining the following across each subgroup:
- proportion of patients achieving histologic improvement at week 48
- Mean HBV DNA change from baseline at week 48
Baseline Characteristics
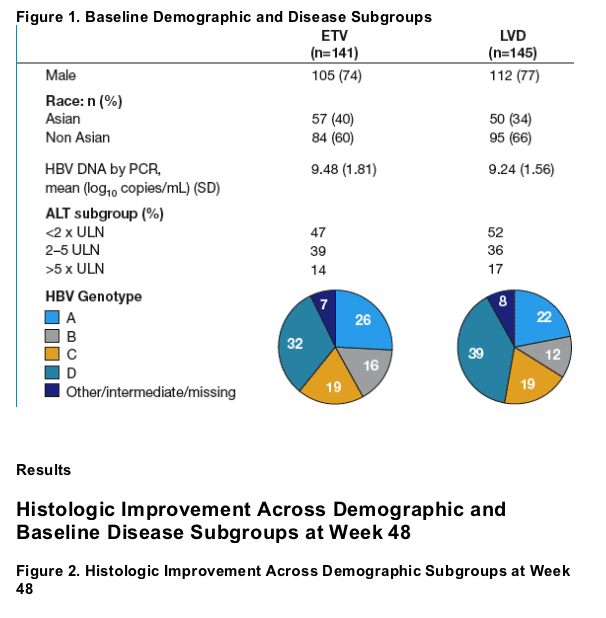
- The distribution of baseline demographic and disease subgroups was well balanced between treatment groups (Figures 1 and 2)
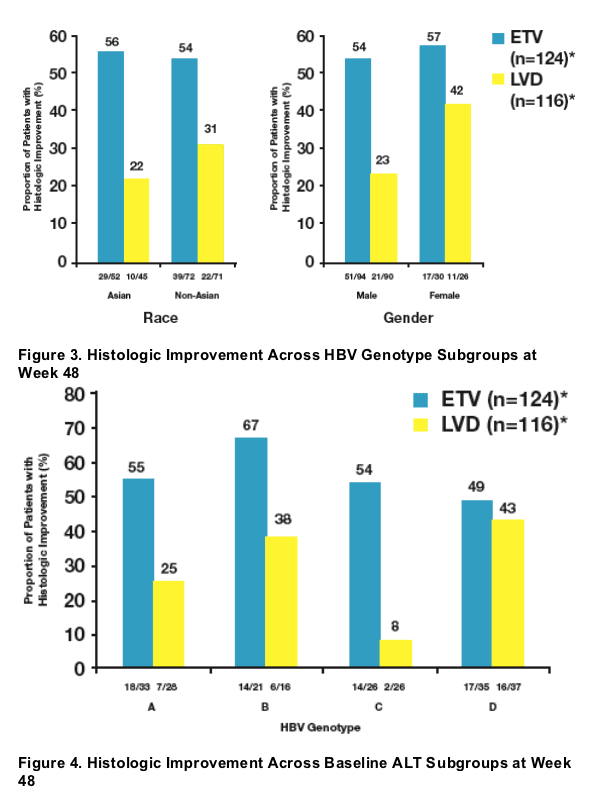
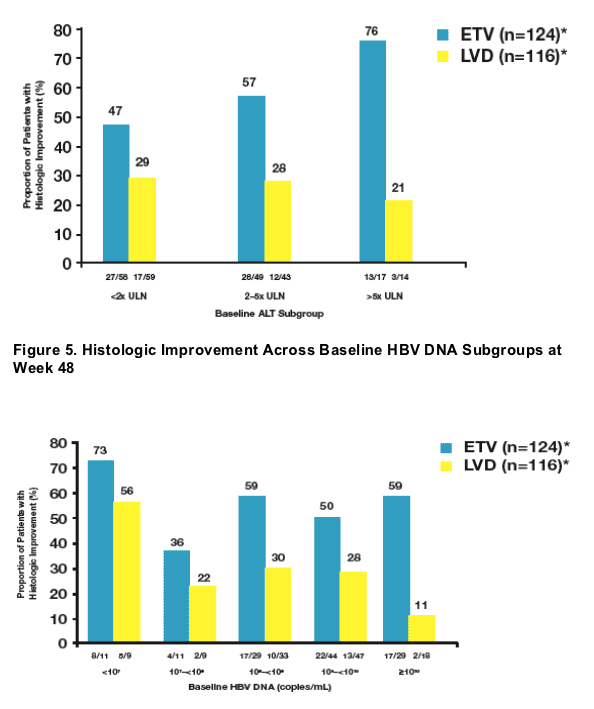
Baseline ALT levels >/= 2 x ULN were associated with higher rates of histologic improvement among ETV-treated patients at week 48
- Regardless of baseline characteristics, the majority of ETV-treated patients showed histologic improvement at week 48
- Analysis of subsets supported the principal analysis for superiority of ETV over LVD for histologic improvement
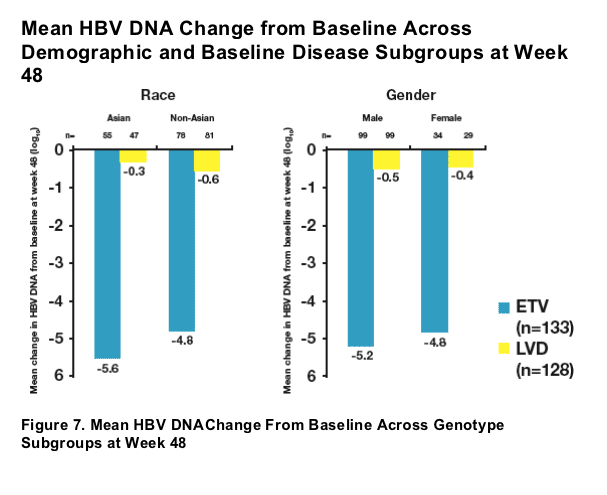
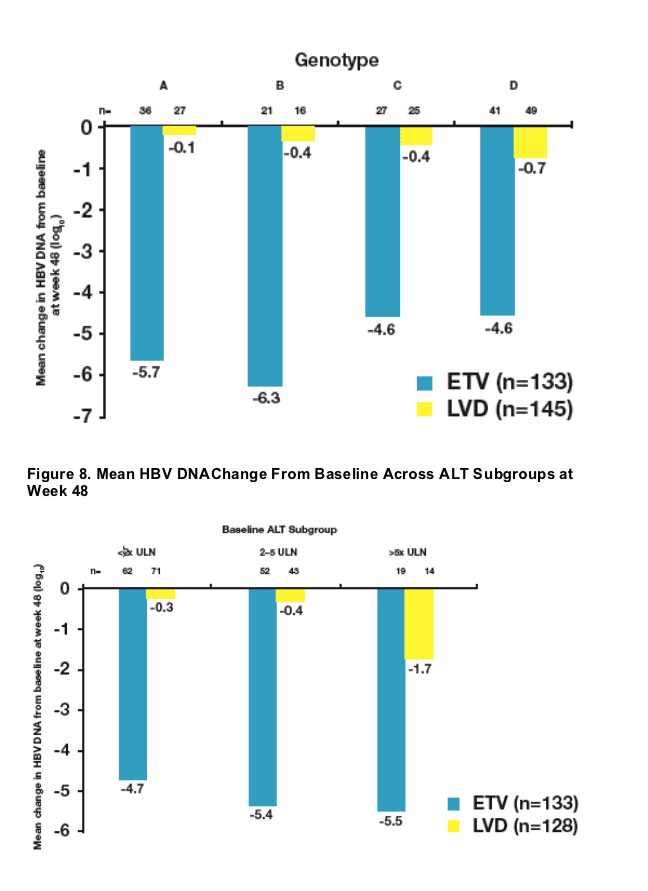
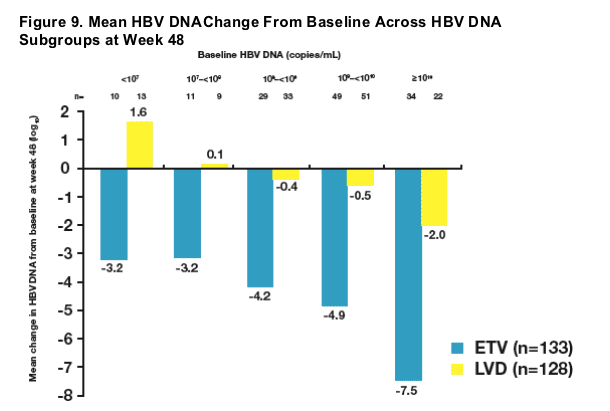
- Baseline HBV DNA levels ≥108 were associated with a higher observed mean HBV DNA change from baseline among ETV-treated patients at week 48
- Regardless of baseline characteristics, mean HBV DNA change from baseline was substantial among the majority of ETV-treated patients at week 48
- Analysis of subsets supported the principal analysis for superiority of ETV over LVD for mean HBV DNA change
|
| |
|
 |
 |
|
|